Volume 70 - Issue 1 - February 2022
Original Paper
Extraction of 40Ar-39Ar Ages from a Multicomponent Mixture: A Case Study From the Tatra Mountains, Poland
- Artur Kuligiewicz, Michał Skiba, Marek Szczerba, Chris M. Hall, Dorota Bakowska
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-19
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Extraction of meaningful information on the timing of fault activity from clay gouges using radiometric dating methods, such as those based on the K-Ar system, can be challenging. One of the factors complicating interpretation of the radiometric dating results is the presence of multiple K-bearing components in the gouge material. In the current study, an attempt was made to develop a new interpretative method for K-Ar and 40Ar-39Ar dating, capable of handling a three-component mixture. In addition, the mineral composition of clay gouges from the Tatra Mountains (Poland), which has not been investigated before, is reported. The mineral compositions of the bulk clay gouge material and separated size fractions were determined by X-ray diffractometry and Fourier-transform infrared spectroscopy. The gouge samples were composed of quartz, dioctahedral mica (as a discrete phase and as a component of mixed-layered illite-smectite), and chlorite, commonly with plagioclase and more rarely with K-feldspar, dioctahedral smectite, calcite, anatase, or trace kaolinite. One feldspar-free sample containing three mica polytypes (1Md, 1M, and 2M1) was chosen for dating with the 40Ar-39Ar method. The results of 40Ar-39Ar dating were interpreted using three concepts: Illite Age Analysis (IAA), a method based on the MODELAGE software, and a newly developed three-component concept. The age values obtained with IAA were −14 Ma ± 31 Ma and 180 ± 91 Ma for authigenic (1Md) and inherited (1M + 2M1) components, respectively. The MODELAGE-based approach returned –4 ± 40 Ma and 165 ± 62 Ma. The three-component approach returned age values of polytypes as follows: 1Md, 15 ± 37 Ma; 1M, 135 ± 57 Ma; 2M1, 121 ± 56 Ma based on the medians and the interquartile ranges of non-normal distributions of Monte Carlo-simulated age values. The results obtained indicated that the 1Md polytype was probably formed during the most recent stage of fault activity, while 1M and 2M1 polytypes are of equal age, roughly.
Evaluation of the Hydraulic Conductivity of Geosynthetic Clay Liners
- Juan Hou, Yuyang Teng, Shifen Bao, Hao Li, Lei Liu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 20-33
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The hydraulic conductivity of geosynthetic clay liners (GCLs) is not fully understood and certain gaps in knowledge are still present, such as the effect of coupled mechanical and chemical processes. The current study aimed to develop a simplified mathematical model to predict the hydraulic conductivity of GCLs, particularly regarding the coupled effects of mechanical and chemical processes. Based on Darcy's Law and Poiseuille’s Law, the method combines diffuse double layer (DDL) theory and fractal theory. External factors such as confining pressure and the concentration of the permeating solution, and inherent properties such as exchangeable cations, ionic radius, montmorillonite surface fractal dimension, the distance between two montmorillonite layers (m) after swelling at the exchangeable cation i (i denotes the primary exchangeable cations, such as Na+, Ca2+, K+, and Mg2+ in bentonite), density, and coefficient of viscosity of interlayer water between two montmorillonite layers, were considered. The proposed theoretical model gave relatively accurate predictions. A practical estimate of GCL hydraulic conductivity was also derived. The predictions were compared with experimental results and good qualitative agreement was found. From the experimental results, the proposed prediction model has a maximum deviation of ~1:10–10:1, and the empirical model has a mean deviation of ~1:15–15:1.
Aqueous Solubility of Zn Incorporated into Mg-Al-Layered Double Hydroxides
- Anna Dabizha, Michael Kersten
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 34-47
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Hydrotalcite minerals are layered double hydroxides (LDH) which play an important role in immobilizing hazardous compounds to decontaminate industrial wastewaters. The stability of an LDH is mostly evaluated in terms of its low solubility in water. However, the solubility of divalent trace metals immobilized by Mg-Al-type LDHs is not well known. Hydrotalcites containing Zn in solid solution, (Mg+Zn)3-Al-LDH, were synthesized by alkaline co-precipitation. A series of eleven LDH phases with Zn mole fractions XZn = Zn/(Mg+Zn) of 0–1 were characterized by powder X-ray diffractometry (XRD), Fourier-transform infrared (FTIR) spectroscopy, thermogravimetry (TGA), scanning and transmission electron microscopy (SEM/TEM), Brunauer-Emmett-Teller (BET) surface area analysis, and inductively coupled plasma mass (ICP-MS) spectrometry. The XRD analysis provided sharp characteristic spacings for d003 and d006 which occurred for all samples, confirming a layered LDH structure. Cell parameters (a, c) obeyed Vegard’s law and confirmed the formation of a regular solid-solution series without a mixing gap. An aqueous equilibrium time was determined by kinetic dissolution experiments. Steady-state solubility occurred after 120 days, but the experiments continued up to 240 days. The XRD and SEM/TEM analyses indicated no phase changes during the long-term dissolution experiments; neither were phase impurities detected after 240 days. The solubility products of the Mg- and Zn-bearing endmember compositions were calculated from experimentally determined total cation and anion concentrations using the Visual Minteq code for considering element speciation and ion pairing. The solubility product decreased as the Zn mole fraction increased, suggesting that the Zn-bearing LDH phases were more stable than the pure Mg3-Al-LDHs. Solid-solution aqueous-solution thermodynamic equilibrium modeling using the Lippmann “total solubility product” approach and applying Lippmann diagrams with logarithmic x-axes revealed a log-linear decrease in aqueous Zn solubility. The results are promising for remediation of metal-bearing liquid wastes because the metals that co-precipitated with the LDH were more strongly retained and, therefore, less soluble than the hydroxides or carbonates of the trace metal.
The Composition and Growth Mechanism of Coexisting 4M2 and 4A8 Biotite Polytypes from Rhyolite of Long Valley Caldera, California
- Jiaxin Xi, Yiping Yang, Lingya Ma, Hongping He, Huifang Xu, Jianxi Zhu, Jingming Wei
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 48-61
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Polytypism is common in micas, and the frequency of polytype occurrence is believed to be related closely to the crystallization conditions and chemical compositions of the corresponding fluids and melts. Coexisting multiple standard and complex/disordered polytypes in igneous rocks generally reflect a complicated magma evolution history. The purpose of the current study was to clarify the origin of coexisting biotite polytypes and their growth mechanism. Micro-X-ray diffraction (μXRD) and transmission electron microscopy (TEM) were used to investigate Fe-rich biotite phenocrysts in rhyolite from the Long Valley Caldera, California, USA. The μXRD analyses characterized various polytypes, and TEM observations revealed that common polytypes (e.g. 1M, 2M1, and 3T) and rare polytypes (e.g. 4M2 and 4A8) coexist within biotite monocrystals. The two 4-layer polytypes of Fe-rich biotite, 4M2 and 4A8, were identified via selected-area electron diffraction (SAED) and high-resolution scanning transmission electron microscopy (HRSTEM) at the atomic resolution, with unique stacking sequences ([0222] for 4M2 and [002
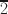 ] for 4A8). Energy-dispersive X-ray spectroscopy (EDS) results showed differences in their chemical compositions, especially Fe and K. The 4A8 polytype is reported for the first time. The present study suggested that environmental changes, such as rapid cooling and inhomogeneous compositional distribution, led to chemical and structural oscillations and complex nucleation of the two 4-layer polytypes. Screw dislocations producing spiral growth enhance polytype stability and form ordered long-period/complex polytypes. These results are useful to understand the origin of long-period/complex polytypes and the intergrowths of diverse polytypes formed in non-equilibrium crystallization environments.
] for 4A8). Energy-dispersive X-ray spectroscopy (EDS) results showed differences in their chemical compositions, especially Fe and K. The 4A8 polytype is reported for the first time. The present study suggested that environmental changes, such as rapid cooling and inhomogeneous compositional distribution, led to chemical and structural oscillations and complex nucleation of the two 4-layer polytypes. Screw dislocations producing spiral growth enhance polytype stability and form ordered long-period/complex polytypes. These results are useful to understand the origin of long-period/complex polytypes and the intergrowths of diverse polytypes formed in non-equilibrium crystallization environments.
Sustained-Release Drug Delivery Potential of Antibiotic–Montmorillonite Composites Prepared Using Montmorillonite Mined in Gampo, Republic of Korea
- Myungjae Kim, Ki-Min Roh, Jiwoo Kim, Minkyeong Seo, Il-Mo Kang, Jiwoong Kim
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 62-71
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Biocompatible drug-delivery materials are important because they provide controlled release of biologically active agents to enhance the effectiveness of medical treatments. Montmorillonite (Mnt) has been utilized in drug-delivery systems for delayed-release application because it can safely encapsulate drug molecules via intercalation reactions. The objective of the present study was to evaluate the delivery characteristics of the drug ciprofloxacin (CIP) from a composite with Mnt (Mnt-CIP) in which the Mnt was first prepared by acid treatment and vibration ball milling. The surfaces of Mnt were modified by reacting the Mnt suspension in 1.0 M HCl acid and by dispersing the powder with a vibration ball mill, then the CIP drug was added at pH 4 and stirred. The goal was to improve the sustained-release performance of the CIP. This Mnt-CIP drug-release system was characterized by X-ray diffraction, X-ray fluorescence analysis, Fourier-transform infrared spectroscopy, surface area measurement using the Brunauer-Emmett-Teller (BET) method, and ultraviolet spectroscopy. The X-ray diffraction results confirmed the intercalation of CIP into the interlayer space of Mnt. The in vitro release properties of the intercalated CIP were investigated using a simulated phosphate-buffered saline solution (pH 7.4) at 36±0.5°C. The CIP drug exhibited a continued release for 3 h. Moreover, Mnt prepared by HCl acid treatment and dispersion in the vibration ball mill delayed the drug dissolution rate. In summary, the Mnt-CIP composite prepared in this study exhibited slow and sustained release characteristics, indicating that Mnt mined from the Gampo-40 mining area in Gyeongju can be used in various drug-delivery applications.
Boron and Lithium Isotopic Signatures of Nanometer-Sized Smectite-Rich Mixed-Layers of Bentonite Beds From Campos Basin (Brazil)
- Norbert Clauer, Lynda B. Williams, I. Tonguç Uysal
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 72-83
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Boron and lithium were analyzed in three nanometer-sized (<20, 20-50 and 50-100 nm) separates of two Santonian (85.8-83.5 Ma) bentonite samples collected closely in the Campos Basin along the southeastern Atlantic coast (Brazil). The B and Li data give various trends that suggest varied crystallization conditions for separates that consist of overwhelming smectite with less than 9% illitic layers. The δ11B of the few illitic tetrahedral sites from one of the samples remains quite constant, while its contents are strictly correlated with those of K, which suggests that illitization proceeded by interaction with pore fluids of the host sediments that supplied the K. In the second sample, the δ11B of the illite layers from the two coarser fractions is indicative of an early volcanic origin, while the smaller size fraction also interacted with sedimentary fluids. Favored by octahedral substitutions of the smectite layers, the δ7Li is more strictly regulated by a volcanic link. In turn, the information of the B and Li isotopic compositions and contents from studied mixed-layers suggests a various origin for the few illite layers of the smectite-rich I-S that contain more B than the smectite layers that host more Li. The difference appears to be sample-site and crystal-size dependent, fueled by pore fluids of the hosting turbidites.
A FIB-SEM Study of Illite Morphology in Aeolian Rotliegend Sandstones: Implications for Understanding the Petrophysical Properties of Reservoir Rocks
- Markus Peltz, Arne Jacob, Georg H. Grathoff, Frieder Enzmann, Michael Kersten, Laurence N. Warr
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 84-105
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Diagenetic illite growth in porous sandstones leads to significant modifications of the initial pore system which result in tight reservoirs. Understanding and quantifying these changes provides insight into the porosity-permeability history of the reservoir and improves predictions on petrophysical behavior. To characterize the various stages of diagenetic alteration, a focused ion beam – scanning electron microscopy (FIB-SEM) study was undertaken on aeolian sandstones from the Bebertal outcrop of the Parchim Formation (Early Permian Upper Rotliegend group). Based on 3D microscopic reconstructions, three different textural types of illite crystals occur, common to many tight Rotliegend sandstones, namely (1) feldspar grain alterations and associated illite meshworks, (2) tangential grain coats, and (3) pore-filling laths and fibers. Reaction textures, pore structure quantifications, and numerical simulations of fluid transport have revealed that different generations of nano-porosity are connected to the diagenetic alteration of feldspars and the authigenic growth of pore-filling illites. The latter leads to the formation of microstructures that range from authigenic compact tangential grain coatings to highly porous, pore-filling structures. K-feldspar replacement and initial grain coatings of illite are composed primarily of disordered 1Md illite whereas the epitaxially grown illite lath- and fiber-shaped crystals occurring as pore-filling structures are of the trans-vacant 1Mtv polytype. Although all analyzed 3D structures offer connected pathways, the largest reduction in sandstone permeability occurred during the initial formation of the tangential illite coatings that sealed altered feldspars and the subsequent growth of pore-filling laths and fibrous illites. Analyses of both illite pore-size and crystallite-size distributions indicate that crystal growth occurred by a continuous nucleation and growth mechanism probably controlled by the multiple influx of potassium-rich fluids during late Triassic and Jurassic times. The detailed insight into the textural varieties of illite crystal growth and its calculated permeabilities provides important constraints for understanding the complexities of fluid-flow in tight reservoir sandstones.
Development of Kinetic Parameters for Nitric Acid Leaching of Phlogopite and the Characterization of Solid Products
- Cheri M. Favel, Barend J. du Plessis
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 106-119
-
- Article
-
- You have access Access
- HTML
- Export citation
-
South Africa is a net importer of fertilizer products, importing all of its potassium, as well as 60–70% of its nitrogen requirements. Thus, domestic prices are impacted significantly by international prices, shipping costs, and exchange rates. Producing these fertilizers locally would be far more economical. Phlogopite, a rich source of potassium, is discarded in large quantities during mining operations; the objective of the present study, therefore, was to determine the acid-leaching characteristics and behavior of phlogopite as a means of releasing potassium. Phlogopite samples were leached with nitric acid (source of nitrogen for fertilizers) at various concentrations, temperatures, and reaction times. The feed phlogopite and leached residue samples corresponding to conversions of 14% (LP1), 44% (LP2), and 100% (LP3) were collected and analyzed using X-ray fluorescence spectroscopy (XRF), X-ray diffractometry (XRD), attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR), Brunauer–Emmett–Teller surface area and porosity analysis (BET), thermogravimetric analysis (TGA), and field emission gun-scanning electron microscopy (FEG-SEM). The feed phlogopite was highly crystalline. The absence of defects in the lattice meant that the motion of H+ atoms penetrating into the lattice was restricted, suggesting internal diffusion-controlled leaching. Furthermore, results obtained from the various analytical techniques corroborated each other in terms of the release of cations during leaching. All leaching experiments were conducted batchwise, in a closed system. The gravimetric data from the experiments were used to identify a suitable model which predicts accurately the leaching behavior. The reaction was found to be internal diffusion-controlled, and the D1 model, which represents one-dimensional diffusion through a flat plate, predicts the leaching behavior most accurately. The observed activation energies (Ea) and pre-exponential constants (k0) varied with initial nitric acid concentration ([H+]0).
Adsorption Behavior of Asphaltene on Clay Minerals and Quartz in a Heavy Oil Sandstone Reservoir with Thermal Damage
- Yanlong He, Weizhe Niu, Zhanwu Gao, Hao Dong, Shizi An, Chunchun Han, Liang Zhao
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 120-134
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Differences in the properties of clay minerals cause formation damage under the condition of thermal production in heavy-oil reservoirs; asphaltenes adsorbed on clay minerals exacerbate the formation damage. The purpose of the present study was to reveal the variation in clay minerals and the adsorption behavior of asphaltenes on clay mineral surfaces under thermal recovery conditions. Volume changes and transformations of typical clay minerals were studied under various conditions (80 and 180°C, pH 9 and 11, aqueous and oven-dry conditions). On this basis, the adsorption behavior and mechanism of asphaltenes on the surfaces of clay minerals in various simulated conditions were investigated. The adsorption mechanism was revealed using kinetics and isothermal adsorption models. The results showed that the volume of montmorillonite expanded by up to 159.13% after water–rock interaction at 180°C with pH 11; meanwhile, the conversion rates of kaolinite and illite to montmorillonite were 6.6 and 7.8%, respectively. The water–rock interaction intensified the volume changes and transformations of clay minerals under thermal conditions. The amounts of asphaltene adsorbed on clay minerals at 180°C were greater than those at 80°C. The adsorption process of asphaltenes was inhibited under aqueous conditions. The abilities of the constituent minerals to bind asphaltenes was in order: montmorillonite > chlorite > kaolinite > illite > quartz sand. The adsorption process of asphaltenes yielded high coefficients of regression with both the Freundlich and Langmuir models under oven-dry (>0.99) and aqueous (>0.98) conditions. At 180°C under aqueous conditions, the water film significantly inhibited the adsorption of asphaltene on the clay minerals. The adsorption process of asphaltenes, therefore, could be regarded as the adsorption occurring at lower concentrations under oven-dry conditions.
Petro-Mineralogical and Geochemical Evaluation of Glauconitic Rocks of the Ukra Member (Bhuj Formation), Kutch Basin, India
- Saurabh Shekhar, V. Kumari, S. Sinha, D. Mishra, A. Agrawal, K. K. Sahu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 135-153
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Glauconites occurring within the Ukra Member of Kutch Basin have remained unexplored in terms of their economic significance. The present study aimed to present a detailed physicochemical characterization of glauconite occurring in the siliciclastic rocks of Guneri and Umarsar area of the Kutch district, Gujarat, India to explore their economic potential. The study involved an integrated petrographical, mineralogical, and geochemical investigation of glauconitic rocks to highlight the occurrence, nature, and maturity of glauconite. The characterization was carried out using X-ray diffraction (XRD), X-ray fluorescence (XRF), and electron probe microanalysis (EPMA) combined with energy dispersive X-ray (EDX), Field emission gun scanning electron microscopy (FEG-SEM), Fourier-transform infrared spectroscopy (FTIR), and inductively coupled plasma mass spectroscopy (ICP-MS). Petrographic and bulk XRD analysis revealed that the glauconite occurs as green pellets constituting ~30 and 40% of the glauconitic sandstone and shale, respectively. Whole-rock analysis showed that the value of K2O varies considerably from 3.93 wt.% (sandstone) to 5.63 wt.% (shale). Mineral chemistry indicated the distinctive chemical composition of glauconite pellets containing 7.4–8.4 wt.% of K2O. The parameters, such as the distance between the (001) and (020) peaks and the large K2O content (~8 wt.%) of the glauconite fraction reflect an evolved to highly evolved stage of maturation. The morphological and spectral signatures further support the high degree of maturation in glauconites. Trace-element analysis implied that the glauconitic sandstone and shale contain elements such as Zn, Mn, Cu, Co, Mo, and Ni, which serve as essential micronutrients for plants. These data sets collectively constitute part of a preliminary study which is prerequisite to beneficiation, but further evaluation of its potential as a potash fertilizer also is needed.