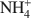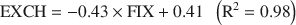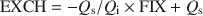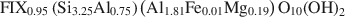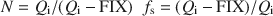Volume 57 - Issue 5 - October 2009
Research Article
Relationship Between Pb2+ Adsorption and Average Mn Oxidation State in Synthetic Birnessites
- Wei Zhao, Haojie Cui, Fan Liu, Wenfeng Tan, Xionghan Feng
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 513-520
-
- Article
- Export citation
-
The relationship between vacant Mn structural sites in birnessites and heavy-metal adsorption is a current and important research topic. In this study, two series of birnessites with different average oxidation states (AOS) of Mn were synthesized. One birnessite series was prepared in acidic media (49.6–53.6 wt.% Mn) and the other in alkaline media (50.0–56.2 wt.% Mn). Correlations between the Pb2+ adsorption capacity and the d110 interlayer spacing, the AOS by titration, and the release of Mn2+, H+, and K+ during adsorption of Pb2+ were investigated. The maximum Pb2+ adsorption by the birnessites synthesized in acidic media ranged from 1320 to 2457 mmol/kg with AOS values that ranged from 3.67 to 3.92. For birnessites synthesized in alkaline media, the maximum Pb2+ adsorption ranged from 524 to 1814 mmol/kg, with AOS values between 3.49 and 3.89. Birnessite AOS values and Pb2+ adsorption increased as the Mn content decreased. The maximum Pb2+ adsorption to the synthetic birnessites calculated from a Langmuir fit of the Pb adsorption data was linearly related to AOS. Birnessite AOS was positively correlated to Pb2+ adsorption, but negatively correlated to the d110 spacing. Vacant Mn structural sites in birnessite increased with AOS and resulted in greater Pb2+ adsorption. Birnessite AOS values apparently reflect the quantity of vacant sites which largely account for Pb2+ adsorption. Therefore, the Pb2+ adsorption capacity of birnessite is mostly determined by the Mn site vacancies, from which Mn2+, H+, and K+ released during adsorption were derived.
Origin and Mineralogy of Sepiolite and Palygorskite From the Tuluanshan Formation, Eastern Taiwan
- Ming Kuang Wang, Pao Chung Tseng, Shyun Sheng Chang, Dah Tong Ray, Yen Hong Shau, Yun Wei Shen, Ruey Chyong Chen, Po Neng Chiang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 521-530
-
- Article
- Export citation
-
The Tuluanshan Formation of the eastern Coastal Range of Taiwan overlies an andesitic core complex presumed to be the source of hydrothermal fluids responsible for the Si- and Mg-rich mineralization of sepiolite and palygorskite (attapulgite) which are found in veins within fissures and in fracture zones of the volcanic rocks of the region. This study was undertaken in order to understand these relationships better by characterizing sepiolite and palygorskite in this Formation and by examining their occurrence and distribution in the Tungho (TH) and Chunjih (CJ) areas. Samples were analyzed using X-ray diffraction (XRD), thermal analysis, Fourier-transform infrared (FTIR) spectroscopy, and petrographic, scanning (SEM), and transmission (TEM) electron microscopic methods. Sepiolite and palygorskite are blocky and earthy-type materials that display fibrous characteristics when viewed using TEM and SEM and occurred alone or with chalcedony in veins. The fibers of blocky sepiolite are commonly intercalated with smectite but the earthy type of sepiolite and palygorskite observed in this study displayed precipitation from fluid enriched in Si, Al, Mg, and minor Fe and depleted in other ions at an earlier stage of offset of the andesitic veins. Continuation of reverse faulting and high shearing stress caused the precipitation of a significant quantity of interlaminated sepiolite. Sepiolite and palygorskite were formed at an earlier stage of fluid interaction relative to smectite in the Tuluanshan Formation.
Partial Dissolution of Glauconitic Samples: Implications for the Methodology of K-Ar and Rb-Sr Dating
- Arkadiusz Derkowski, Jan Środoń, Wojciech Franus, Peter Uhlík, Michał Banaś, Grzegorz Zieliński, Maria Čaplovičová, Małgorzata Franus
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 531-554
-
- Article
- Export citation
-
The K-Ar dating of glauconite has been used as an important stratigraphic tool for many decades. The application of this technique is limited to pure glauconites, free of detrital contamination by K-bearing phases, often not easy to detect. This study extends the application of isotope dating to the contaminated glauconites and offers a precise technique for detecting the detrital contamination of glauconites.
The most common K-bearing detrital contaminants have smaller (K-feldspars, Al-rich dioctahedral micas) or greater (trioctahedral micas) dissolution rates than glauconite in extremely low pH solutions. The differences in the dissolution rates can be applied to evaluate the purity of the glauconite and its crystallization age.
The interlaboratory GLO glauconite standard and grain-size fractions separated from glauconitic sandstones of the Paleogene (sample GL) and Jurassic (sample GW8) ages were treated with acid (3M HCl, at 99±2°C) for different reaction times (0.5–7 h) and measured for their apparent isotopic ages.
Microporous amorphous silica with large specific surface area is the solid product of the reaction and its content increases with reaction time. The K-Ar dates (apparent ages) of the solid residues increase significantly with reaction time: from 44.6 to 107 Ma for the GL sample and from 125.7 to 394.7 Ma for GW8. The increase is negligible in the case of the GLO standard. The Rb-Sr data of the GL sample were modeled using initial 87Sr/Sr ratios of 0.707–0.709, which resulted in a 29.9–35.8 Ma date for the untreated portions of GL, and ∼42.6 Ma after 7 h of treatment.
The increase of isotopic K-Ar date with increasing time of dissolution is interpreted to be a result of increasing concentration of detrital, acid-resistant, K-bearing minerals, observed also with the electron microscope and X-ray diffraction. Probabilistic modeling based on single (K-Ar) or double (K-Ar and Rb-Sr) isotopic systems evaluated the isotopic ages of the detrital and authigenic minerals, and their K2O and Rb concentrations. The crystallization ages computed using these two methods are: 24.0, 26.5, and 32.3 Ma for the GL material, and 117.3–121.8 Ma for the GW8 series.
The proposed method based on partial dissolution is a potential tool for evaluating the reliability of glauconite dating.
Correction
Evidence of Degradation of Triarylmethine Dyes on Texas Vermiculite
- Giora Rytwo, Yotam Gonen, Reuma Huterer-Shveky
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 555-565
-
- Article
- Export citation
-
Synthetic dyes in industrial effluents pose a significant risk to human health and the environment, so much effort has been expended to degrade them using various methods, including the use of clay minerals as catalysts. The purpose of this study was to advance understanding of the mechanisms for clay-catalyzed degradation of crystal violet (CV) and other triarylmethine dyes using three different vermiculite clays (Llano, Texas, VTx-1; Ojen, OV; and Russian, RV), a montmorillonite (SWy-1), and a Spanish sepiolite (SEP). While OV, RV, SWy-1, and SEP showed almost no activity with respect to dye degradation, VTx-1 caused complete removal of the dye from solution up to the equivalent of 200% of the cation exchange capacity of the clay. While large amounts of dye were removed from the solution, no change in basal spacing was observed by X-ray diffraction. The kinetics of removal of CV from solution began after a lag period of >10 days in a process that can be described by pseudo-second order kinetics. By comparison, adsorption of CV onto SWy-1 and SEP was immediate, without any lag period. Sonication treatment of the VTx-1 vermiculite suspension caused the CV removal process to begin immediately. Fourier-transform infrared measurements of adsorption of CV on clays revealed that for the OV and RV vermiculites, SEP sepiolite, and SWy-1 montmorillonite the spectra were similar to the original dye; the spectra of the VTx-1-dye differed considerably, however, exhibiting vibrations of methylene groups (—CH2—) which were not present in the CV molecule. The significant changes in the IR spectrum indicated that CV underwent degradation on the surface of the VTx-1 vermiculite. Carbon-content analysis led to the conclusion that degradation products remained bound to the clay. Similar effects were observed for two other triarylmethine dyes (malachite green and methyl green) added to VTx-1, indicated that it may, therefore, be considered suitable as a sorbent to remove and decompose such dyes from industrial effluents. Pretreatment by sonication would remove the need for long incubation times.
Research Article
Progressive Formation of Halloysite From the Hydrothermal Alteration of Biotite and the Formation Mechanisms of Anatase in Altered Volcanic Rocks From Limnos Island, Northeast Aegean Sea, Greece
- Dimitrios Papoulis, Panagiota Tsolis-Katagas, Angelos G. Kalampounias, Basilios Tsikouras
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 566-577
-
- Article
- Export citation
-
Occurrences of halloysite-rich material in altered volcanic rocks, principally trachyandesites, dacites, and tuffs, extend over an area of ∼1 km2 in the southwestern part of Limnos, Island, northeast Aegean Sea, Greece. The present study was designed to investigate the alteration processes which acted on the biotite in these volcanic rocks, to describe in detail the mechanism of formation of the halloysite, and to specify the mechanisms of formation of anatase during the alteration processes. Samples were examined using polarized-light microscopy, X-ray powder diffraction, scanning electron microscopy, scanning electron microscopy-energy dispersive spectroscopy, and Fourier-transform-Raman techniques. The extensive alteration of the parent rocks, triggered by the circulation of hydrothermal fluids through faults and fractures, resulted in the alteration of biotite to halloysite. Six stages of alteration were recognized. Nanoparticles of halloysite were initially formed on the mica layers, which progressively grew through short-tubular to well formed tubular halloysite, with increasing alteration. In the most altered samples, laths and interconnected laths with the composition (Al3.96Fe0.04)Si4O10(OH)8, were the dominant halloysite morphologies. Anatase was encountered as an alteration product of both ilmenite and biotite. Ilmenite was altered to anatase and Fe oxides. The altered ilmenite crystals constrained most of the newly formed anatase within the space occupied previously by ilmenite, leading to the formation of skeletal anatase. The layered structure of the micas was the main factor governing the morphology of newly formed anatase developed outside ilmenite margins in the form of layers parallel to those of mica. An unusual ring-like structure of anatase was thought to be the result of the uncommon alteration of inner parts of mica folia to tubular halloysite oriented perpendicular to the mica layers. The detachment of the halloysite tubes by circulating hydrothermal fluids was considered to be the reason for the creation of holes which were subsequently surrounded by the anatase ring forms.
Effect of Silica Polymerization on the Oxalate-Promoted Dissolution of Goethite
- Matthew J. Eick, Todd P. Luxton, Holly A. Welsh
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 578-585
-
- Article
- Export citation
-
Numerous studies have investigated the ligand-promoted dissolution of Fe (oxyhydr)oxides. In natural environments, inorganic ligands can compete with organic ligands for surface sites on (oxyhydr)oxides which may influence dissolution rates. Published research of this interaction and its effect on the dissolution of (oxyhydr)oxides is rare. The objective of the present study was to examine the extent to which silica, as a naturally occurring competitive ligand added in the form of silicic acid, impacts the oxalate-promoted dissolution of the common soil Fe (oxyhydr)oxide goethite. Sorbed silica reduced the oxalate-promoted dissolution rate of goethite at all surface coverages investigated. As initial silica solution concentrations increased from 0.50 mM to 5.0 mM, relatively little change in the dissolution rate was observed. Fourier-transform infrared (FTIR) spectra indicated that, as silica-surface coverages increased, the silica underwent polymerization on the goethite surface. Initially, silicate was associated with surface functional groups, but as polymerization occurred some of the silica appeared to desorb from the goethite surface without being released into the bulk solution, suggesting that silica polymers formed discrete islands or surface clusters that grew away from the goethite surface rather than expanding epitaxially across the surface. Minimal changes were observed in the quantity of reactive goethite surface, which is responsible for the observed dissolution rates, as silica-surface coverages increased.
Effect of the Size of Aggregates on Pore Characteristics of Minerals Measured by Mercury Intrusion and Water-Vapor Desorption Techniques
- Grzegorz Jozefaciuk
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 586-601
-
- Article
- Export citation
-
The size, shape, and continuity of pores in mineral solids greatly influence the behavior of percolating liquids and solids in porous media, which has significant practical environmental implications. In order to expand understanding of these properties in soil minerals, the present study was undertaken to analyze the pore characteristics of bentonite, illite, and kaolinite in the forms of powder and aggregates of different dimensions, combining water-vapor desorption and mercury-intrusion techniques. Different granulometric fractions of milled quartz glass were also studied. With increasing aggregate size of the minerals, larger pore volumes (up to 25%), smaller surface areas (down to 15%), larger average radii (up to 15%), and smaller fractal dimensions (down to 6%) were measured using water-vapor adsorption-desorption data. The differences were smallest for bentonite, possibly due to the smallest particle size of this mineral and/or to its very large water-vapor adsorption capacity. The degree of water-vapor adsorption on quartz was too small to rely on the data obtained.
The pore volumes and average radii, measured by mercury-intrusion porosimetry, were up to few times larger for the mineral powders than for their aggregate counterparts. Similar values were noted for aggregates >1 mm in diameter, for which the input of interaggregate spaces into total porosity of the sample bed was negligible. Two pathways of mercury intrusion were detected in porosimetric curves: filling of interaggregate spaces, and penetration into aggregates. Similar penetration thresholds into aggregates of different sizes were calculated. With increasing size of quartz grains, the pore volume of the quartz bed decreased whereas the average pore radius increased. Mercury intrusion detected pore-fractal behavior of bentonite and kaolinite, but for aggregated minerals the calculated values of fractal dimensions were >3, values which increased with increasing aggregate size. Very similar pore parameters were measured for aggregates prepared from a natural deposit of kaolinite and for artificially prepared aggregates from powder of the same mineral, indicating that artificial aggregation can simulate natural processes.
Both water desorption and mercury intrusion detected fractal behavior in the limited range of pores. A test to find fractal build up of the aggregates in extended scales based on a dependence of surface area of unit volume of aggregate bed on aggregate size showed no fractal-aggregate build-up.
Clay Mineralogy Along the Laterite Profile in Hubei, South China: Mineral Evolution and Evidence for Eolian Origin
- Hanlie Hong, Zhaohui Li, Ping Xiao
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 602-615
-
- Article
- Export citation
-
In spite of many studies of laterite origins in various parts of the world, the origin of laterite in the middle to lower reaches of the Yangtze River is still a topic of debate, thus leaving doubts about the prevailing environmental and climatic conditions at the time. The purpose of this study was to provide greater understanding of this subject by examining in more detail the associated mineralogical evolution, i.e. clay mineral composition, structural characteristics of clays in various beds with different degrees of weathering along the laterite profile, and the alteration mechanisms during the pedogenic process in tropical to semitropical climate conditions. High-resolution transmission electron microscopy (HTEM), X-ray diffraction, and wavelength dispersive X-ray fluorescence spectrometry were used to characterize the samples in order to link clay mineralogy in various beds with different degrees of weathering along the laterite profile to the formation and origin of laterites in the region. The laterite profile displayed a distinct layered structure and was divided into a saprolite (B4), a light colored net-like clay bed (B3), a brown-red gravelly clay bed (B2), and a dark-brown topsoil (B1), respectively, from bottom to top. The clay mineral assemblage of beds B1 and B2 was illite, kaolinite, and chlorite, while that of beds B3 and B4 was mainly kaolinite with minor illite. The bimodal particle-size distribution of clay minerals in the laterite profile indicated that fine-grained particles could have been produced by partial dissolution and decomposition of coarse-grained ones. Examination by HRTEM revealed that fine-grained particles usually occurred as X-ray amorphous materials in the upper soil beds, but with euhedral morphology in the lower portions, suggesting that the fine-grained particles in the lower soil beds might be partially neoformed during the weathering process. Amorphous spots occurred frequently in kaolinite crystals in the upper soil beds, while the structure of kaolinite was well preserved, with a well defined lattice-fringe image. The illite crystallinity index exhibited a trend of downward decrease, while the values of the chemical index of alteration (CIA = Al2O3/(Al2O3+ CaO+ K2O+ Na2O) × 100%) of the soil profile showed a trend of downward increase. Samples of the upper soil beds, B1 and B2, had comparable SiO2/Al2O3 ratios of 5.37–6.22, while those of the lower beds, B3 and B4, had significantly smaller SiO2/Al2O3 ratios of 1.92–3.98, suggesting that the latter had a greater degree of weathering than the former, in reasonable agreement with the results of the illite crystallinity and CIA index. In addition, samples from B1 and B2 had similar TiO2/Al2O3 ratios of 0.042–0.053, while those from B3 and B4 had comparable TiO2/Al2O3 ratios of 0.021–0.033, comparable to the value 0.020 of the bedrock, and were notably smaller than the upper soil beds, indicating that materials of the upper soil beds, B1 and B2, had a different origin from the lower soil beds. The upper bed was probably derived from eolian accumulation due to intensification of the winter monsoon and aridity in central Asia and was modified by intense chemical weathering since the late Pleistocene, while the lower bed originated from an in situ weathering of the underlying argillaceous limestone.
Clayey Cap-Rock Behavior in H2O-CO2 Media at Low Pressure and Temperature Conditions: An Experimental Approach
- Eric Kohler, Teddy Parra, Olivier Vidal
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 616-637
-
- Article
- Export citation
-
The storage of CO2 in geological reservoirs requires an understanding of the impact of CO2 on clay-rich sealing cap-rocks to identify and explore critical parameters that modify petrophysical properties such as permeability and fracturing. The purpose of this study was to investigate the effect of heating, under different hydrated-CO2 partial pressures, on the chemical compositions and relative amounts of mineral phases in the Saint Martin de Bossenay (SMB, Paris Basin, France) cap-rock in order to identify possible mineral-phase transitions and to estimate reaction kinetics induced by the presence of excess dissolved CO2.
X-ray diffraction, transmission electron microscopy, and electron microprobe analyses were employed to study mineral alteration, with particular attention given to visualization and quantification of the mineral evolution of clay minerals. In all the altered mixtures investigated, the illitization of clays was combined with the formation of anhydrite. These changes were accompanied by a dolomitization and a slight increase in the quartz content. The CO2-rich samples crystallized Fe2+-and K+-enriched illites, whereas the CO2-free experiments precipitated Al3+-deprived and Mg2+-enriched illites. Advanced characterizations of cap-rock material allowed reaction paths, induced by the increase in dissolved CO2 in the porous media, to be determined precisely. The results place strong constraints on numerical models aimed at evaluating the safety of an SMB site.
Functionalization of the Interlayer Surfaces of Kaolinite by Alkylammonium Groups From Ionic Liquids
- Sadok Letaief, Christian Detellier
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 638-648
-
- Article
- Export citation
-
The objective of this study was to design new, functional, nanostructured materials from the abundant clay mineral kaolinite, in spite of development problems with the interlayer chemistry of kaolinite because of its non-swelling properties. A particular goal of this work was to graft, in a controlled way, alkylammonium groups onto the aluminol interlayer surfaces of kaolinite. This was successfully achieved by soft-chemical approaches, more specifically by the melting intercalation process of alkylammonium ionic liquids which were synthesized for this purpose. The resulting nanohybrid materials were characterized chemically and structurally by X-ray diffraction analysis, thermal analysis (TG/DTA), 13C cross polarization magic angle spinning nuclear magnetic resonance spectroscopy, and Fourier-transform infrared spectroscopy. The amount of grafted organic material was quantified from TGA results. Alkylammonium salts with a short alkyl chain were grafted directly using a melting reaction at 180°C under N2 involving the in situ displacement of dimethylsulfoxide (DMSO) from a DMSO-kaolinite pre-intercalate; for longer alkyl chains, the grafting was done in two steps. In the first step, the corresponding amino-alcohol was grafted into the kaolinite by displacement of DMSO from the interlayer space. The second step consisted of quaternarization of the grafted material by reaction with iodomethane or iodoethane.
The Charge of Component Layers of Illite-Smectite in Bentonites and the Nature of End-Member Illite
- Jan Środoń, Edwin Zeelmaekers, Arkadiusz Derkowski
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 649-671
-
- Article
- Export citation
-
The nature of component layers of mixed-layer illite-smectite and their possible evolution in the course of illitization have been debated since the 1960s. The present study is a new attempt to solve these problems, using samples collected from diverse geological formations around the world. Twenty three purified illite-smectites from bentonites and hydrothermal rocks, covering the complete range of expandability, were analyzed chemically, including \$\end{document}
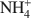 determination, and their structural formulae were calculated. The exchangeable cations (EXCH) were plotted vs. the fixed cations (FIX) yielding the following experimental regression:\$\end{document}
determination, and their structural formulae were calculated. The exchangeable cations (EXCH) were plotted vs. the fixed cations (FIX) yielding the following experimental regression:\$\end{document}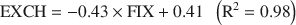
FIX and EXCH depend on the charge of the illite (Qi) and smectite (Qs) interlayers, and the fractions of these interlayers in the bulk clay leading to: \$\end{document}
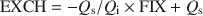
Analysis of these relations and independent measurements of the total specific surface area (TSSA) indicate that the layer charges of both types do not change in the course of illitization. The smectite layer charge is equal to 0.41 and the illite layer charge is equal to 0.95 per O10(OH)2. End-member illite has a well defined composition that is close to intermediate between muscovite and phengite, with fixed cations content greater than that specified in the AIPEA classification of layer silicates:\$\end{document}
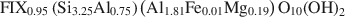
The established relationship allows the calculation of the mean number (N) of 2:1 layers in all fundamental particles and also the fraction of smectitic layers (fs) from FIX:\$\end{document}
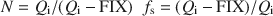
N and fs can be used to calculate TSSA, and all three parameters can also be calculated from cation exchange capacity and from X-ray diffraction peak positions, utilizing the regressions established here.