Volume 49 - February 2001
Research Article
Baseline Studies of the Clay Minerals Society Source Clays: Preface
- P. A. Costanzo, S. Guggenheim
-
- Published online by Cambridge University Press:
- 28 February 2024, p. 371
-
- Article
- Export citation
-
The Source Clays Program of The Clay Minerals Society was initiated in 1972 to distribute a set of reference clays, so that distributed clays could be identical for all recipients. Because most clays do not consist of a single phase, the immediate objective was not to produce a pure product consisting of one clay mineral, but to provide a uniform product. These materials were collected and processed carefully, and sufficient amounts were collected so that material was available for researchers for many years, Large numbers of researchers were thereby assured of working on identical material. Initial descriptions of these materials were presented in the Data Handbook (van Olphen and Fripiat, 1979). An updated version of this book was suggested several years ago because of the availability of new analytical techniques and to provide descriptions of material added to the reference set since 1979.
Mica Structure and Fibrous Growth of Illite
- Necip Güven
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 189-196
-
- Article
- Export citation
-
The relative growth rates of the three joint chains of silica tetrahedra and metal octahedra in the [100], [1̅10] and [1̅1̅0] directions within the mica layer (referring to the 1M unit-cell) seem to control the morphology of mica crystallites. Laths and fibers are the products of relatively fast growth along the [100] direction compared to growth along the [1̅10] and [1̅1̅0] directions. The (010) growth front in 1M micas with trans-octahedral vacancies exposes a pair of reactive OH ions that can form organic or inorganic complexes and ‘poison’ the growth on the (010) face.
Authigenic illite fibers in two sandstones with contrasting lithologies are found to have grown on mica or kaolinite cores. Illite fibers appear in single sets or in multiple sets, 120° apart. This texture seems to be related to the stacking sequence of the layers in mica or kaolinite in the core of these fibers.
Perturbative Theory of Mica Polytypism. Role of the M2 Layer in the Formation of Inhomogeneous Polytypes
- Massimo Nespolo
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 1-23
-
- Article
- Export citation
-
A new model is proposed to explain, within the framework of the theory of spiral growth of Frank, the formation on inhomogeneous mica polytypes. This model relates the interaction and cooperative growth of two components (spirals and/or crystals) to produce a new stacking sequence. Depending on the relative orientation between the two components, a mismatch of the interlayer positions occurs, which is compensated through either a growth defect or a crystallographic slip at the octahedral (O) sheet. Both these adjustments transform the Ml layer into the M2 layer. These two types of layers have the same chemical composition but differ in cation distribution in the O sheet. The coalescence and cooperative growth of crystals occurs in fluid-rich environments and is most frequent in druses and volcanic fumaroles. These environments favor the inhomogeneous polytypes, especially those with complex stacking sequenc¬es. In addition, the Ml → M2 transformation is most probable in micas with an oxybiotitic composition, where the removal of the OH dipole strengthens the interlayer bonding and the presence of high-charge cations destabilizes the O sheet. Three examples of inhomogeneous polytypes of titaniferous oxybiotite from Ruiz Peak (a volcanic environment where many inhomogeneous polytypes have been reported) are presented.
Thickness and Surface Characteristics of Colloidal 2:1 Aluminosilicates Using an Indirect Fourier Transform of Small-Angle X-Ray Scattering Data
- Chao Shang, James A. Rice, Jar-Shyong Lin
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 277-285
-
- Article
- Export citation
-
An indirect Fourier transformation applied to small-angle X-ray scattering data has been used to determine the thickness and surface properties of two common clay minerals. For an illite system, the particle density distribution function (PDDF) generated by the analysis gave a correct description of particle geometry, and the calculated electron density profile was in accordance with the theoretical electron density distribution for this mineral. This approach provides the opportunity to determine the thickness of fundamental particles of illite while avoiding the difficulties encountered in other methods. Both the PDDF and the electron density profile accurately predict the thickness of Na-montmorillonite layers, and the results suggest that an electron inhomogeneity exists at the interface of this mineral.
Transmission Electron Microscopy Study of Conversion of Smectite to Illite in Mudstones of the Nankai Trough: Contrast with Coeval Bentonites
- Harue Masuda, Donald R. Peacor, Hailiang Dong
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 109-118
-
- Article
- Export citation
-
Clay minerals in shales from cores at Site 808, Nankai Trough, have been studied using X-ray diffraction (XRD), scanning transmission electron microscopy (STEM), and analytical electron microscopy (AEM) to compare the rates and mechanisms of illitization with those of coeval bentonites, which were described previously. Authigenic K-rich smectite having a high Fe content (∼7 wt. %) was observed to form directly as an alteration product of volcanic glass at a depth of ∼500 meters below seafloor (mbsf) with no intermediate precursor. Smectite is then largely replaced by Reichweite, R, (R = 1) illite-smectite (I-S) and minor illite and chlorite over depths from ∼550 to ∼700 mbsf. No further mineralogical changes occur to the maximum depth cored, ∼1300 m. Most smectite and I-S in shales are derived from alteration of glass, rather than being detrital, as is usually assumed. Discrete layer sequences of smectite, I-S, or illite coexist, indicating discontinuities of the transformation from smectite to (R = 1) I-S to illite. Authigenic Fe-rich chlorite forms concomitantly with I-S and illite, with the source of Fe from reactant smectite.
Smectite forms from glass with an intermediate precursor in coeval bentonites at approximately the same depth as in shales, but the smectite remains largely unchanged, with the exception of exchange of interlayer cations (K → Na → Ca) in response to formation of zeolites, to the bottom of the core. Differences in rates of illitization reflect the metastability of the clays. Temperature, structure-state, and composition of reactant smectite are ruled out as determining factors that increase reaction rates here, whereas differences in water/rock ratio (porosity/permeability), Si and K activities, and organic acid content are likely candidates.
Mechanisms and Crystal Chemistry of Oxidation in Annite: Resolving the Hydrogen-Loss and Vacancy Reactions
- D. G. Rancourt, P. H. J. Mercier, D. J. Cherniak, S. Desgreniers, H. Kodama, J.-L. Robert, E. Murad
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 455-491
-
- Article
- Export citation
-
A synthetic octahedral-site-vacancy-free annite sample and its progressive oxidation, induced by heating in air, were studied by powder X-ray diffraction (pXRD), Mössbauer spectroscopy, nuclear reaction analysis (NRA), Raman spectroscopy, X-ray fluorescence (XRF) spectroscopy, gas chromatography (GC), thermogravimetric analysis (TGA), differential thermal analysis (DTA), scanning electron microscopy (SEM), and size-fraction separation methods. For a set heating time and as temperature is increased, the sample first evolves along an annite-oxyannite join, until all H is lost via the oxybiotite reaction (Fe2+ + OH− ⇌ Fe3+ + O2− + H↑). It then evolves along an oxyannite-ferrioxyannite join, where ideal ferrioxyannite, KFe3+8/3□1/3AlSi3O12, is defined as the product resulting from complete oxidation of ideal oxyannite, KFe3+2Fe2+AlSi3O12, via the vacancy mechanism (3 Fe2+ ⇌ 2 Fe3+ + [6]□ + Fe↑). A pillaring collapse transition is observed as a collapse of c near the point where
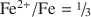 and all OH groups are predicted and observed to be lost. Quantitative analyses of H, using NRA, GC, and Raman spectroscopy, corroborate this interpretation and, in combination with accurate ferric/ferrous ratios from Mössbauer spectroscopy and lattice parameter determinations, allow a clear distinction to be made between vacancy-free and vacancy-bearing annite. The amount of Fe in ancillary Fe oxide phases produced by the vacancy mechanism is measured by Mössbauer spectroscopy to be 11.3(5)% of total Fe, in agreement with both the theoretical prediction of 1/9 = 11.1% and the observed TGA weight gain. The initiation of Fe oxide formation near the point of completion of the oxybiotite reaction (
and all OH groups are predicted and observed to be lost. Quantitative analyses of H, using NRA, GC, and Raman spectroscopy, corroborate this interpretation and, in combination with accurate ferric/ferrous ratios from Mössbauer spectroscopy and lattice parameter determinations, allow a clear distinction to be made between vacancy-free and vacancy-bearing annite. The amount of Fe in ancillary Fe oxide phases produced by the vacancy mechanism is measured by Mössbauer spectroscopy to be 11.3(5)% of total Fe, in agreement with both the theoretical prediction of 1/9 = 11.1% and the observed TGA weight gain. The initiation of Fe oxide formation near the point of completion of the oxybiotite reaction (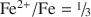 ) is corroborated by pXRD, TGA, Raman spectroscopy, and appearance of an Fe oxide hyperfine field sextet in the Mössbauer spectra. The region of Fe oxide formation is shown to coincide with a region of octahedral site vacancy formation, using a new Mössbauer spectral signature of vacancies that consists of a component at 2.2 mm/s in the [6]Fe3+ quadrupole splitting distribution (QSD). The crystal chemical behaviors of annite-oxyannite and of oxyannite-ferrioxyannite are best contrasted and compared to the behaviors of other layer-silicate series in terms of b vs. [D] (average octahedral cation to O bond length). This also leads to a diagnostic test for the presence of octahedral site vacancies in hydrothermally synthesized annite, based on a graph of b vs. Fe2+/Fe. The implications of the observed sequence of thermal oxidation reactions for the thermodynamic relevance of the oxybiotite and vacancy reactions in hydrothermal syntheses are examined and it is concluded that the oxybiotite reaction is the relevant reaction in the single-phase stability field of annite, at high hydrogen fugacity and using ideal starting cation stoichiometry. The vacancy reaction is only relevant in a multi-phase field, at lower hydrogen fugacity, that includes an Fe oxide equilibrium phase (magnetite) that can effectively compete for Fe, or when using non-ideal starting cation stoichiometries.
) is corroborated by pXRD, TGA, Raman spectroscopy, and appearance of an Fe oxide hyperfine field sextet in the Mössbauer spectra. The region of Fe oxide formation is shown to coincide with a region of octahedral site vacancy formation, using a new Mössbauer spectral signature of vacancies that consists of a component at 2.2 mm/s in the [6]Fe3+ quadrupole splitting distribution (QSD). The crystal chemical behaviors of annite-oxyannite and of oxyannite-ferrioxyannite are best contrasted and compared to the behaviors of other layer-silicate series in terms of b vs. [D] (average octahedral cation to O bond length). This also leads to a diagnostic test for the presence of octahedral site vacancies in hydrothermally synthesized annite, based on a graph of b vs. Fe2+/Fe. The implications of the observed sequence of thermal oxidation reactions for the thermodynamic relevance of the oxybiotite and vacancy reactions in hydrothermal syntheses are examined and it is concluded that the oxybiotite reaction is the relevant reaction in the single-phase stability field of annite, at high hydrogen fugacity and using ideal starting cation stoichiometry. The vacancy reaction is only relevant in a multi-phase field, at lower hydrogen fugacity, that includes an Fe oxide equilibrium phase (magnetite) that can effectively compete for Fe, or when using non-ideal starting cation stoichiometries.
Current Problems with the Nomenclature of Phyllosilicates
- B. B. Zvyagin
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 492-499
-
- Article
- Export citation
-
The nomenclature of phyllosilicates is discussed in relation to structural variations of layers and interlayers. Some discrepancies in the nomenclature which arose due to historical reasons and different viewpoints may be reconciled if the choice of the construction units (layers or their parts) is related to definite sets of structures for which these units are common. Features of idealized models approximating real structures should be considered for classification and derivation of nomenclature sets, whereas priority is given to structural variations before lattice and symmetry characteristics. Layer polymorphs, pseudo-polytypes, polytypes (both simple and complex), and OD structures are distinguished. Particular results obtained for micas as a model example of phyllosilicates in relation to modular structures in general are considered in an Appendix.
Three-Dimensional Crystal Structures of Illite-Smectite Minerals in Paleozoic K-Bentonites from the Appalachian Basin
- Douglas K. McCarty, Robert C. Reynolds, Jr.
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 24-35
-
- Article
- Export citation
-
The three-dimensional crystal structures of illite-smectite (I-S) in K-bentonite samples from the Appalachian Basin are characterized by rotational disorder in the stacking sequence of 2:1 illite layers, different proportions of n60° rotations (as opposed to n120°) in the rotated layers, and layers with centrosymmetric trans-vacant (tv) octahedral sites that are randomly interstratified with noncentric cis-vacant (cv) layers. The proportion of cv interstratification in the I-S increases with tetrahedral A1 and decreases with octahedral Mg and Fe content. The I-S minerals in the northern Appalachian basin K-bentonites are characterized by high (79% average) proportions of cv (Pcv) layers. In contrast, I-S from equivalent K- bentonites from the southern Appalachian basin has low Pcv values (38% average). These values do not correlate with expandability or rotational disorder. The geographic distribution of these I-S structural parameters may have resulted from possibly short-term, hot, and advective fluid migrations that differed in Mg concentrations and/or other physical and chemical parameters.
Rheology of Na-Rich Montmorillonite Suspension as Affected by Electrolyte Concentration and Shear Rate
- Hadar Heller, Rami Keren
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 286-291
-
- Article
- Export citation
-
Rheological measurements were used to evaluate the particle-particle associations of Na-rich montmorillonite in suspensions, under various electrolyte concentrations. A 2% free electrolyte clay suspension showed pseudoplastic flow behavior and had a high apparent viscosity, attributed at low shear rates to the high volume fraction of the suspended clay platelets, the flexibility of the platelets, and the presence of edge-to-edge association. The breaking of edge-to-edge associations and the progressive orientation of the individual platelets in the direction of flow contribute to the reduction in viscosity with increasing shear rate.
The compression of the diffuse double layer at a NaCl concentration of 10 mEq L-1 contributes to the free movement of the individual platelets, even at low shear rates. The flow behavior changed from pseudoplastic to plastic at an NaCl concentration of 100 mEq L-1. At this electrolyte concentration, face-to-face associations of specific junction points at certain areas of the planar surface are probably occurring.
The apparent viscosity of the clay suspension for the two particle-size ranges (<2 and <0.02 μm) at all shear rates converged to a minimum value of 4.5 mPa s at NaCl concentrations of 10–20 mEq L-1. On both sides of the minimum, the lower the shear rate, the greater the slope. The apparent viscosity of a 2% suspension of Na-rich montmorillonite <0.02 μm particles, however, was significantly greater than that observed for a suspension of <2 u,m particles. This high apparent viscosity is attributed to the increase in edge surface area and the number of clay particles in a unit volume.
We suggest that edge-to-edge association between Na-rich montmorillonite platelets prevails when the NaCl concentration is below the electrolyte critical concentration, for which the apparent viscosity of the suspension is at its minimum value, whereas face-to-face association prevails at NaCl concentrations above this critical value.
Unusual X-Ray Characteristics of Vermiculite from Wiry, Lower Silesia, Poland
- Boris A. Sakharov, Elżbieta Dubińska, Paweł Bylina, Grzegorz Kaproń
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 197-203
-
- Article
- Export citation
-
Coarse-grained vermiculite from a serpentinite-pegmatite thermal zone displays a rational series of narrow 14.4 Å basal reflections and an unusual broad 28 Å peak. X-ray diffraction simulations and fitting techniques show that the 28 Å peak is related to 28 Å domains consisting of elongated 2:1 layers of different lengths. The domains are located at the crystal edges of the vermiculite.
Baseline Studies of the Clay Minerals Society Source Clays: Introduction
- Patricia M. Costanzo
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 372-373
-
- Article
- Export citation
-
Clay minerals share a basic set of structural and chemical characteristics (e.g. they are largely aluminosilicates with layer structures) and yet each clay mineral has its own unique set of properties that determine how it will interact with other chemical species. The variation, in both chemistry and structure, among the clays leads to their applications in extremely diverse fields. Common and important industrial applications of clays are in the manufacture of paper, paint, plastics and rubber. One of their more recent and most economically important applications is in the petlitter industry where their adsorptive and deodorizing properties are used. Specialty uses include clay additive to chicken feed to boost nutritional uptake by the chicken, and in using clay as fillers and major ingredients in pharmaceuticals and cosmetics. Clays are used for their catalytic properties and for their ability to adsorb greases, fats and other organic materials. Those who exist with scarce resources frequently collect clays from local deposits and ingest them as a source of dietary minerals. It is difficult for a day to go by without using a product incorporating clay minerals, as we all use ceramics such as dinnerware and sanitaryware.
Characterization of Montmorillonite Surfaces After Modification by Organosilane
- Kang Song, Giselle Sandí
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 119-125
-
- Article
- Export citation
-
X-ray powder diffraction (XRD), thermal gravimetric analysis (TGA), surface area measurements, and near-edge X-ray absorption fine structure (NEXAFS) spectroscopy were used to examine the surface properties of organosilane-modified smectite-type aluminosilicate clays. Organic modified clays derived from the reactions of montmorillonite (containing 93–95% montmorillonite from a bentonite, <1% quartz, and 4–6% opal CT) with octadecyltrichlorosilane (C18H37SiCl3) and octadecyltrimethoxysilane [C18H37Si(OMe)3] are highly hydrophobic. Surface loadings of the modified clays depend on the organosilane and the solvent, and they range from 10 to 25 wt. %. The organic species are probably adsorbed to the outer surfaces and bound to the edges of the clay via condensation with edge-OH groups. Encapsulation of montmorillonite with C18H37SiCl3 and C18H37Si(OMe)3 resulted in a hydrophobic coating that acts like a “cage” around the clay particles to limit diffusion. Basal spacings of the organic modified clays remain at ∼15 Å upon heating to 400°C in N2, whereas those of unmodified clays collapse to ∼10 Å. A considerable reduction in surface area (by 75–90%) for organic modified clays is observed, which is consistent with the existence of a surface coating. The solvent used can affect the amount of organic silane coated on the clay particles, whereas the difference between the products prepared using C18H37SiCl3 and C18H37Si(OMe)3 in the same solvent is relatively small. The carbon and oxygen K-edge NEXAFS spectroscopy of the modified montmorillonite surfaces showed that surface coatings on the outside of the clay particles exist. The encapsulating system may allow for economical remediation and storage of hazardous materials.
Baseline Studies of the Clay Minerals Society Source Clays: Geological Origin
- William F. Moll, Jr.
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 374-380
-
- Article
- Export citation
-
In any clay deposit, the nature of the mineral assemblage and the composition of individual clay minerals can change radically in a few em. Consequently, any given locality can contain many subtly different types of clay minerals. Results from different laboratories on ostensibly the same clay material may not always be comparable because the samples may indeed not contain an assembly of identical clay minerals. Such confusion slows the understanding of this important group of minerals. Several attempts were made to provide investigators with reasonably constant clay materials, the first being that of the American Petroleum Institute Project 49 (Kerr, 1949). The Clay Minerals Society Source Clays project proposed to provide investigators with gently homogenized clay materials, carefully collected and processed under the supervision of clay specialists. The collection would include metric ton amounts to ensure a long-lasting collection. The program began in 1972, with the introduction of the materials described in this paper. Later the program expanded to include the Special Clays. These samples are materials not amenable to homogenization, or they are available in very small amounts.
Possible Role of Microbial Polysaccharides in Nontronite Formation
- Masato Ueshima, Kazue Tazaki
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 292-299
-
- Article
- Export citation
-
Nontronite and microbes were detected in the surface layers of deep-sea sediments from Iheya Basin, Okinawa Trough, Japan. Nontronite, an Fe-rich smectite mineral, was embedded in acidic polysaccharides that were exuded by microbial cells and electron microscopy showed that the nontronite layers were apparently oriented in the polysaccharide materials. We propose that the formation of nontronite was induced by the accumulation of Si and Fe ions from the ambient seawater and that extracellular polymeric substances (EPS) served as a template for layer-silicate synthesis. Experimental evidence for this hypothesis was obtained by mixing a solution of polysaccharides (dextrin and pectin) with ferrosiliceous groundwater. After stirring the mixture in a sealed vessel for two days, and centrifuging, Fe-rich layer silicates were identified within the precipitate of both the dextrin and pectin aggregates, whereas rod-shaped or spheroidal Si-bearing iron hydroxides were found in the external solution. Microbial polysaccharides would appear to have affected layer-silicate formation.
Kübler Illite “Crystallinity” Index of the Cretaceous Gyeongsang Basin, Korea: Implications for Basin Evolution
- Jae Il Lee, Yong Il Lee
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 36-43
-
- Article
- Export citation
-
Thermal maturity of the Lower Cretaceous Sindong and Hayang groups in the Gyeongsang Basin, Korea, was investigated using the Kübler illite “crystallinity” index (KI) which is based on the numerical expression of the 10-Å peak width after calibration to the Crystallinity Index Standard scale. The metamorphic grade of the Sindong and Hayang groups ranges from late diagenetic zone to high anchizone. Depth of burial was not a major factor controlling KI variation in the basin because Hayang mudrocks have higher thermal maturity than the underlying Sindong mudrocks. Short-lived heating by the emplacement of the Upper Cretaceous plutonic rocks is responsible for the higher thermal maturity in the Hayang mudrocks. A linear NNE-SSW trending belt is drawn by connecting the lowest KI values, and it seems to reflect a hidden fault trace beneath the Hayang Group. Emplacement of Upper Cretaceous plutonic rocks and introduction of hydrothermal fluids along the inferred fault may have been the main cause of the lowering of KI values along this belt. This inferred fault is interpreted to represent the buried eastern boundary of the Nagdong Trough, which was the main depositional site for the Sindong Group in the western part of the Gyeongsang Basin. Deposition of the Hayang Group occurred in the enlarged basin when the basin extended eastward. This study provides an example that KI values can be used in the reconstruction of an early history of basin evolution.
The CS/K Exchange in Muscovite Interlayers: An Ab Initio Treatment
- Kevin M. Rosso, James R. Rustad, Eric J. Bylaska
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 500-513
-
- Article
- Export citation
-
Plane-wave pseudopotential total energy calculations have been applied to investigate the structure and energetics of the Cs/K exchange into interlayer sites in muscovite mica. Novel muscovite structures were designed to isolate the effects of 2:1 layer charge, cation size/interlayer site shape, and tetrahedral Al/Si substitutions on the exchange. All atom and cell-parameter optimizations were performed with the intention to mimic the constant pressure, non-isovolumetric exchange conditions thought to be found at frayed-edge sites. Under conditions where the cell parameters are allowed to relax, the overall Cs/K exchange reaction is surprisingly close to isoenergetic. The forward reaction is more strongly favored with increasing layer charge. For the condition of zero layer charge and no interlayer site distortion, the difference in the optimal interlayer spacing for Cs relative to K is very small, indicating a baseline indifference of the muscovite structure to cation size. The presence of 2:1 layer charge or tetrahedral rotations arising from Al/Si substitutions clearly change this outcome. Analysis of the dependence of the interlayer spacing on layer charge shows that while the spacing collapses with increasing layer charge for K as the interlayer cation, the reverse is true for Cs. We attribute the contrasting behavior to inherent differences in the ability of these cations to screen 2:1 layer-layer repulsions. Such effects might be involved during exchange at frayed-edge sites where interlayer spacings are increased. This is known, from experiment, to be very selective for Cs. Overall, the exchange energetics are so low that the Cs/K exchange rate and degree of irreversibility are likely to be dominated by diffusion kinetics.
Formation and Growth of Smectites In Bentonites: A Case Study From Kimolos Island, Aegean, Greece
- George E. Christidis
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 204-215
-
- Article
- Export citation
-
The low-temperature alteration of a rhyolitic rock from Kimolos Island, Aegean, Greece, yielded an alteration profile characterized by gradual transition from fresh glass to bentonite containing homogeneous Chambers-type montmorillonite and then to a mordenite-bearing bentonite. Both mordenite and smectite were formed from poorly crystalline precursors, which probably had compositions comparable to that of the crystalline end-product. However, their composition may have been modified to some degree after reaction with the fluid phase. Particle length and width measurements of smectite crystals yielded lognormal profiles, which suggest supply-controlled crystal growth in an open system or random ripening in a closed system. The former mechanism is in accordance with the observed sustained supply of Mg and Fe by the fluid phase throughout the alteration profile and is believed to be the dominant formation mechanism of smectites in bentonites in general. In the mordenite-bearing zone, random ripening is expected in domains with low permeability, in which the system was essentially closed, favoring the formation of mordenite. The level of supersaturation with respect to smectite was probably lower in the mordenite-bearing zone. Smectite probably affected pore-fluid chemistry either through ion exchange or via dissolution of initially formed K-bearing smectite. The latter process raised the K+/(Na+ + Ca2+) activity ratio of the pore-fluid favoring K-bearing mordenite.
Genesis of Dioctahedral Phyllosilicates During Hydrothermal Alteration of Volcanic Rocks: I. The Golden Cross Epithermal Ore Deposit, New Zealand
- David A. Tillick, Donald R. Peacor, Jeffrey L. Mauk
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 126-140
-
- Article
- Export citation
-
To characterize the evolution of dioctahedral interstratified clay minerals in the Golden Cross epithermal deposit, New Zealand, hydrothermally altered volcanic rocks containing the sequence smectite through illite-smectite (I-S) to muscovite were examined by optical microscopy, X-ray diffraction (XRD), scanning electron microscopy (SEM), and transmission and analytical electron microscopies (TEM/AEM).
XRD analyses of 30 oriented clay samples show a broad deposit-wide trend of increasing illite content in I-S with increasing depth and proximity to the central vein system. Six representative samples were selected for SEM/TEM study on the basis of petrographic observations and XRD estimates of I-S interstratification. Ca and Na are the dominant interlayer cations in smectite, but as the proportion of illite layers in I-S increases, so does the K content and (IVAl + VIAl)/Si ratio. Layers and packets tend to flatten and form larger arrays, reducing the amount of pore space. Smectite coexists with (R = 1) I-S, rather than being (R = 0) I-S where R is the Reichweite parameter. The highest alteration rank samples contain discrete packets of mica to ∼300 Å thick, but a limited chemical and structural gap exists between illite, which is intermediate in composition between common illite and muscovite, and illite-rich I-S. Selected-area electron diffraction (SAED) patterns of mica show that the 1M polytype dominates, rather than the common 2M1 polytype.
Petrographic, SEM, and TEM data imply that all phyllosilicates formed via neoformation directly from fluids. Relatively mature I-S and micas form simultaneously, without progressing through the series of transformations that are commonly assumed to characterize diagenetic sequences during burial metamorphism in mud-dominated basins. Although the overall distribution of clay minerals is consistent with temperature as a controlling variable, local heterogeneities in the distribution of clay minerals were controlled by water/rock ratio, which varied widely owing to different rock types and fracture control.
The Relationships between Kaolinite Crystal Properties and the Origin of Materials for a Brazilian Kaolin Deposit
- Angélica F. Drummond C. Varajāo, Robert J. Gilkes, Robert D. Hart
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 44-59
-
- Article
- Export citation
-
The clay particles in a kaolin deposit from Brazil were investigated by X-ray diffraction (XRD), differential thermal analysis (DTA), analytical transmission electron microscopy (ATEM), and electron paramagnetic resonance (EPR) to examine the relationships between morphological and chemical properties of the crystals and to relate these properties to formation conditions. The XRD patterns show the dominant presence of kaolinite with minor amounts of gibbsite, illite, quartz, goethite, hematite, and anatase. ATEM observations show two discontinuities in the deposit as indicated by changes in morphology and size of the kaolinite crystals. At the base of the deposit, hexagonal platy and lath-shaped particles (mean area of 001 face = 0.26 μm2) maintain the original fabric of the parent rock which characterizes an in situ evolution. In the middle of the deposit a bimodal population of large (mean area of 001 face > 0.05 μm2) and small (mean area of 001 face < 0.05 μm2) sub-hexagonal platy kaolinite crystals occurs. This zone defines the boundary between the saprolitic kaolinite and the pedogenic kaolinite. Near the top of the profile, laths and irregular plates of kaolinite, together with sub-hexagonal particles, define two different depositional sources in the history of formation of the deposit. Crystal thickness as derived from the width of basal reflections and the Hinckley index are compatible with the morphological results, but show only one discontinuity. At the base of the deposit, kaolinite has a low- defect density whereas in the middle and at the top of the profile, kaolinite has a high-defect density. Likewise, EPR spectroscopy shows typical spectra of low-defect kaolinite for the bottom of the deposit and typical spectra of high-defect kaolinite for the other portions of the deposit. Despite the morphological changes observed through the profile, the elemental composition of individual kaolinite crystals did not show systematic variations. These results are consistent with the deposit consisting of a transported pedogenic kaolinite over saprolite consisting of in situ kaolinized phyllite.
Genesis of Dioctahedral Phyllosilicates During Hydrothermal Alteration of Volcanic Rocks: II. The Broadlands-Ohaaki Hydrothermal System, New Zealand
- Yonghong Yan, David A. Tillick, Donald R. Peacor, Stuart F. Simmons
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 141-155
-
- Article
- Export citation
-
The clay mineral textures, assemblages, formation mechanisms, and controlling geological parameters relating to alteration of silicic volcanic rocks by hydrothermal solutions, in core samples from the Broadlands-Ohaaki hydrothermal system, New Zealand, were investigated using X-ray diffraction (XRD), scanning electron microscopy (SEM), and transmission and analytical electron microscopy (TEM/AEM). Mineralogical and textural relations of this active hydrothermal system, for which temperatures and fluid relations are well known, are equivalent to those in the Golden Cross hydrothermal gold deposit as described in Part 1.
XRD data show a sequence of clay minerals from smectite to a range of interstratified I-S to mica with increasing depth and temperature, on average. TEM observations are in general agreement with XRD data, especially with respect to relative proportions of illite (I)- and smectite (S)- like layers. TEM data also show that: (1) Smectite packets contain no discrete illite-like layers in samples identified as (Reichweite, R = 0) I-S by XRD. They coexist with separate packets of (R = 1) I-S. (2) A continuous range in I-S occurs from (R = 1) I-S with increasing proportion of illite-like layers, but at high illite-like layer contents there is a gap between I-S and illite. (3) 1M and 2M1 polytypes of mica coexist in separate packets, but the rare 1M polytype has a larger VIMg content.
The data imply that clay minerals formed by dissolution and neocrystallization directly from volcanic phases, although multiple reaction events can not be ruled out. Such “episodic” alteration produces a sequence of clay minerals identical to those of prograde diagenesis of pelitic sediments. This result implies that the presence of a continuous sequence is not definitive proof of continuous sequences of transformation as a function of time and continuous burial. Reaction progress of the clay-mineral sequence is in general accord with the known temperature gradient, but with significant and common exceptions. High porosity and permeability, both inherent in rock texture and local structure, are inferred to foster local reaction progress, as consistent with metastability of phases and the Ostwald step rule.