Research Article
The evaluation of bactericides
- E. R. Withell
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 339-353
-
- Article
-
- You have access Access
- Export citation
-
The Rideal-Walker and Chick-Martin tests depend on end-point methods for comparing bactericides. End-point methods have a number of disadvantages: (1) It is difficult, if not impossible, to estimate end-points accurately. (2) The nature of the test demands a standard time. The results at any standard time will be arbitrary. If the sensitivity of the organisms varies in the two solutions tested, the coefficients obtained will vary with the time chosen as standard. It is impossible to estimate the manner in which the survivor times of the organism are distributed, by end-point methods, and therefore the standard tests in current use do not ascertain whether the result is arbitrary or not. This can only be appreciated if the tests are repeated using different times.
Apart from the difficulties associated with end-points, the coefficients obtained in the present tests are calculated from one concentration of phenol and one concentration of test bactericide. This is also an arbitrary value, for the relative activity of two bactericides will depend on relative values of the concentration exponent, n, for each bactericide; if both phenol and the test bactericide have identical values of n the coefficients will be the same at all concentrations which are compared, but if values of n are different then different coefficients will be obtained for every different concentration compared. As a corollary, unless the value of n for the test bactericide is the same as that of phenol, it is illogical to transfer the phenol coefficient obtained at one concentration to the pure substance, for this procedure implies that the phenol coefficient applies to any concentration of the test bactericide. This is only true when the concentration coefficients of phenol and test bactericide are the same. The standard methods offer no value for n.
All these difficulties can be avoided if the rate of death is followed in both phenol and test bactericide, by viable counts. Two methods of interpreting the results are discussed:
(1) The comparison of reaction rates, calculated by the formula
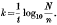 This method is complicated by the occurrence of lag period at the start of the reaction between organism and bactericide. This lag period varies in extent in different experiments (often extending up to 50% of the deaths) and makes the interpretation of the results difficult. The lag period has been shown to be a manifestation of, the distribution of logarithmic survival times (Withell, 1942), and is a function of the standard deviation of the logarithmic survivor times (λ). This fact is taken into account in method (2).
This method is complicated by the occurrence of lag period at the start of the reaction between organism and bactericide. This lag period varies in extent in different experiments (often extending up to 50% of the deaths) and makes the interpretation of the results difficult. The lag period has been shown to be a manifestation of, the distribution of logarithmic survival times (Withell, 1942), and is a function of the standard deviation of the logarithmic survivor times (λ). This fact is taken into account in method (2).(2) The use of the time required for 50% response as a method of comparing bactericides. This time has been symbolized L.t. 50 and is analogous to the lethal dose for the 50% response (l.d. 50) widely used in biological assay. If a logarithmic time scale is used, the logarithmic survival times are found to be approximately normally distributed and consequently probit-logarithm of time lines are straight and in the case of phenol 0·5 % and para-chlor-meta-cresol 0·05% these lines are parallel. In this case a logarithmic time scale means that the comparative times for all percentage responses are equal. In cases where the probit-logarithm-of-time lines are not parallel this would indicate that the standard is unsuitable for the test, for any methods of assay based on a use of a standard to which the organism gives a different response-time relation from that which they give to the test substance or bactericide, are likely to give arbitrary results.
L.T. 50 has been chosen because of the simplicity of the idea underlying it, and because a similar function, L.D. 50, has been used with success in biological assay work. There is also a solid mathematical backing for the use of L.D. 50 in assays involving biological response.
It is suggested that by means of l.t. 50, phenol and the test bactericide can be compared in the following way:
(a) determine the relative speeds of action of the two solutions at any convenient concentrations;
(b) determine the concentration coefficient (ή) of both phenol and test bactericide by means of comparative l.t. 50 values;
(c) determine the temperature coefficient for each substance by similar methods.
From these observations the relative efficiency of the two bactericides can be calculated at any temperature and concentration. If two sets of figures are obtained, one with a vegetative organism, and the other with a sporing organism, a great deal of information can be summarized. It has been shown that an 0·05% solution of parachlor-meta-cresol is approximately one and a half times as efficient as an 0·5% phenol solution (against a Micrococcus). The comparative figure is about 1·0 when Bact. coli is used.
The chemical and physical investigation of germicidal aerosols: II. The aerosol centrifuge
- S. R. Finn, E. O. Powell
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 354-364
-
- Article
-
- You have access Access
- Export citation
-
The principle of the aerosol centrifuge is described, and an elementary mathematicaltreatment is given.
The relations between the behaviour of the centrifuge and its dimensions, speed and airflow have been tested experimentally on three machines. It is shown that the mathe-matical formulae given are an adequate basis for design.
Various applications are mentioned.
Epidemiology of haemolytic streptococcal infection in relation to acute rheumatism: I. Haemolytic streptococcal epidemic and first appearance of rheumatism in a training centre
- C. A. Green
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 365-370
-
- Article
-
- You have access Access
- Export citation
-
1. The circumstances attending an outbreak of haemolytic streptococcal infection in a training centre, followed by acute rheumatism, are described.
2. The incidence of streptococcal infections in trained men and apprentices was 64 and 474 per 1000 respectively.
3. Of streptococcal infections 78·0% occurred in members of two younger classes of apprentices.
4. The eight cases of acute rheumatism were all in apprentices, seven cases being in the same two classes. All but one case had had tonsillitis or scarlatina during the epidemic.
5. The smallest percentage of post-epidemic Dick-positive reactors was found in the youngest class which had suffered most in the outbreak.
Epidemiology of haemolytic streptococcal infection in relation to acute rheumatism: II. Epidemic rheumatism
- C. A. Green
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 371-379
-
- Article
-
- You have access Access
- Export citation
-
Although multiple cases of acute rheumatism have been observed to follow waves of streptococcal infection in institutions (see Green, p. 369) there is doubt as to whether acuter heumatism itself becomes epidemic under such circumstances or whether the element of infectivity lies in the preliminary throat infection as concluded by Sheldon (1931).
Epidemiology of haemolytic streptococcal infection in relation to acute rheumatism: III. Comparative incidence of various infections and acute rheumatism in certain training centres
- C. A. Green
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 380-392
-
- Article
-
- You have access Access
- Export citation
-
The distribution of various infectious diseases in certain training centres is discussed inrelation to the streptococcal aetiology of acute rheumatism, and the relative importance of age and environment are noted.
It gives me real pleasure to acknowledge the interest and support of Vice-Admiral Sir Percival Nicholls, K.C.B., and Vice-Admiral Sir S. F. Dudley, K.C.B., F.R.S., throughout these investigations which were facilitated by the willing co-operation of numerous officers and personnel of the R.N. Medical Service. To Surg. Capt. F. Hitch and the staff of the R.N. Medical School I am greatly indebted, particularly for the efficient technical assistance rendered by Petty-Officer L. Waller. The work was carried out during tenure of the Erederick Pearson-Sir Halley Stewart Naval Research Fellowship of the Empire Rheu-matism Council.
Paratyphoid fever: an epidemiological study
- William Savage
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 393-410
-
- Article
-
- You have access Access
- Export citation
-
The present review furnishes evidence that paratyphoid fever exhibits a good many differences from enteric fever in its epidemiological features.
The incubation period is decidedly variable and, in general, shorter than it is in enteric fever. Not only is the disease usually milder, but the case mortality figure in the present series is particularly low and one more comparable to the case mortality in Salmonella food poisoning than that of enteric fever.
It has to be recognized that acute gastro-intestinal symptoms may be present, very rarely indeed associated with all the cases but more often affecting a proportion of them, which so closely simulate acute food poisoning that that condition may be diagnosed. Usually, however, the same patients later exhibit the normal enteric picture.
The seasonal prevalence, at least as regards the onset of multiple outbreaks as distinct from isolated cases, shows a definite summer prevalence more comparable to the distribution of Salmonella food poisoning than to that of enteric fever.
The vehicles of infection are markedly different and, apart from outbreaks spread by case-to-case infection as in some institution outbreaks, almost invariably the vehicle is some form of food. Whereas milk and ice cream are common agencies in spreading both diseases water and shellfish are absent, or very rare, as vehicles in paratyphoid fever. For this disease cream takes pride of place.
I suggest that these conspicuous differences of vehicle are not associated so much with lack of opportunity to infect as with the need for a vehicle in which multiplication of the paratyphoid bacillus can occur previous to ingestion. These differences of vehicle are explainable on the assumption that the paratyphoid bacillus is less efficient at invading the human body than the typhoid bacillus, and therefore only vehicles which ensure its ingestion in considerable numbers are effective. The few water infections cited in the report bear out this contention, for in all of them water was only effective when massively infected and repeatedly consumed. This explanation is, of course, only a hypothesis, and it may be that other factors, such as preformed toxin, are also required for successful invasion.
The special implication of cream, it may be advanced, is partly due to this factor, and also that this vehicle is one which affords special opportunities for specific infection from an infective food handler.
The vehicle infectivity rate is definitely low, and in many outbreaks a surprisingly small proportion of persons at risk are attacked. The comparative infrequence of definite explosive outbreaks, even with a vehicle from which we should anticipate an outbreak to be explosive in character, is noticeable. Both these facts add support to the view that the paratyphoid bacillus does not readily infect man.
As regards the sources of infection of the vehicle I suggest that the importance of the chronic carrier has been overstressed, and that such carriers are less important in paratyphoid fever than in enteric fever. Our information is still inadequate, but the facts incline one to believe that the true chronic carrier is proportionately rarer than in enteric fever but is likely to be more associated as a causal agent with individual infections where there are opportunities for repeated infections.
On the other hand, my series of outbreaks emphasizes the fundamental importance of the transitory carrier who may be either a mild unrecognized case or a symptomless bacterially infected person. It is evident that in most outbreaks there is in addition to the recognized and so notified cases a reservoir of infected persons in one or both of these groups. Unrecognized by themselves and unknown to the Health Authority they constitute a pool of potential infection, the effectiveness of infection only limited by opportunity, and, to a minor extent, by the difficulty of the paratyphoid bacillus to establish itself as a cause of disease in the human body. No system of control can afford to neglect the potential menace of these two groups of persons.
Much more attention might profitably be given to a detailed study of the extent to which bacillary infection occurs without recognized symptoms and the extent to which these cases clear up within a reasonable period. A comprehensive investigation of isolated single cases of paratyphoid fever would, I am sure, yield valuable epidemiological results.
Influenced no doubt by the prevailing emphasis on the chronic carrier as the commonest source of infection my series shows a tendency to label any person who handled the infected vehicle and whose stools contain the paratyphoid bacillus as the source of infection of that vehicle without steps being taken to prove the contention. So frequently can the presence of the specific bacillus be equally well accounted for on the assumption that the person is a victim of infection in the actual outbreak that some attempt at proof is essential. Modern bacteriological methods are in a position to throw considerable light on the distinction between a potential cause of the outbreak and a victim of that outbreak and ought invariably to be employed.
I am impressed with the extent to which the undoubted fact that Bact. paratyphosum B is a true Salmonella influences the epidemiological picture, and I have suggested that a study of it from this angle furnishes us with a rational explanation of the main differences between its epidemiological behaviour compared with that of the typhoid bacillus.
Immunization against typhoid and paratyphoid with alcohol-killed, alcohol-preserved and heat-killed, phenol-preserved, vaccine
- Hugh Climie
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 411-415
-
- Article
-
- You have access Access
- Export citation
-
1. A comparison of the agglutinogenic response and reactions, both local and general, between a series of sixty-two persons immunized with alcohol-killed, alcohol-preserved, T.A.B.C. vaccine, and forty-four persons immunized with a much older heat-killed, phenol preserved, T.A.B.C. vaccine is described.
2. It was found that there was comparatively little difference between the two series in regard to O agglutinin response.
3. Vi agglutinins were found in 14·4 % of the alcohol-vaccine cases, but none appeared in the heat-killed, phenolized vaccine cases.
4. There was no significant difference in the frequency of local reactions, but the general reactions were markedly fewer in the alcohol-vaccine cases.
5. The occurrence of Vi agglutinin response in six out of fifteen cases given heatkilled, phenol-preserved, T.A.B.C. or T.A.B. vaccines intravenously is recorded.
The Lipoid Antigens of C. Diphtheriae and C. Hofmannii
- L. Hoyle
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 416-422
-
- Article
-
- You have access Access
- Export citation
-
1. Antisera prepared by immunizing rabbits with C. diphtheriae gravis, mitis, and intermedius and C. hofmannii contain antibodies which will give complement-fixation reactions with alcoholic extracts of the organisms.
2. By means of cross-absorption experiments three different lipoid antigens can be demonstrated: (a) a specific antigen h present only in C. hofmannii; (b) a specific antigen d characteristic of C. diphtheriae mitis but probably present also in small amount in C. diphtheriae gravis and intermedius; and (c) a non-specific or group antigen G present in large amount in C. diphtheriae gravis and intermedius and C. hofmannii, and in small amount in C. diphtheriae mitis; this G antigen is probably made up of a number of components.
3. Sera of human diphtheria convalescents may contain antibodies reacting with alcoholic extracts of C. diphtheriae. Sera from mitis cases usually react only with extracts of G. diphtheriae mitis, i.e. they contain only d antibody, while sera from gravis or intermedius cases usually contain only G antibody and react with extracts of C. diphtheriae gravis, intermedius and C. hofmannii, but not with extracts of G. diphtheriae mitis.
Comparative digestibility of wholemeal and white breads and the effect of the degree of fineness of grinding on the former
- T. F. Macrae, J. C. D. Hutchinson, J. O. Irwin, J. S. D. Bacon, E. I. McDougall
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 423-435
-
- Article
-
- You have access Access
- Export citation
-
1. Loaves were baked from three types of wheaten flour made from the same grist; a straight run white flour of 73% extraction, a finely ground wholemeal (100% extraction) and a medium ground wholemeal (100% extraction). Bread from each was consumed by six persons for periods of 11–12 days. The daily consumption was 530–630 g. dry weight of bread and, in addition, 37 g. margarine, 284 c.c. milk, 72 g. marmalade jelly and 284 c.c. of mild ale.
2. The total energy, nitrogen and fibre of the food consumed and of the faeces excreted over a period of 7 days were determined. Assuming that the foods other than bread were wholly digestible, the average percentage absorption of energy, nitrogen and fibre from the white bread was 96·1, 91·1, and 65·8 respectively; from the fine wholemeal bread it was 86·9, 85·3 and 14 respectively and from the coarser ground wholemeal 87·1, 85·7 and 9·7 respectively.
3. The nitrogen intake per 100 g. of the wholemeal bread eaten, was 2·91; of the white bread 2·70 g. This advantage was, however, neutralized by the greater amount of nitrogen lost in the faeces when wholemeal was taken, so that the average net gain of nitrogen to the body was 2·46 and 2·49 respectively per 100 g. dry weight of bread consumed.
4. The loss of 9% more of the energy of the bread in the faeces from wholemeal than from white bread is largely accounted for by the undigested cell envelopes and woody fibre in the bran. The greater utilization of the fibre of the white bread is in accordance with the observations of Rubner who showed that the cell membranes of the endosperm were of different composition from those of the bran cells and were more readily attacked by bacteria in the gut.
5. The range of individual variations in the utilization of the energy of the breads were: for white bread 95·4–96·8% and for wholemeal 85·4–88·7%. In the utilization of nitrogen these were: for white bread 88·2–94·1%, and for wholemeal 78·5–89·6%. The same subjects showed the better utilization of both energy and nitrogen.
6. The fineness of grinding of the wholemeal within the range used made no significant difference to the utilization of either energy or nitrogen.
The disinfectant activity of caustic soda
- Betty C. Hobbs, G. S. Wilson
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. 436-450
-
- Article
-
- You have access Access
- Export citation
-
Quantitative observations were made on the rate of disinfection of Bacterium coli and of the spores of Bacillus subtilis by caustic soda at different temperatures and different concentrations. The tests were carried out in the presence of 1/1000 milk. It was found that:
(1) With Bact. coli the concentration coefficient n was about 2·7, and the temperature coefficient θ10°C. about 2.
(2) With B. subtilis spores the concentration coefficient n was about 1·75 and the temperature coefficient θ10°C. about 1·5.
(3) With Bact. coli the values for the reaction velocity constant k tended to be irregular at 40° C. and with B. subtilis spores at 70° C, suggesting that at these temperatures some additional factor, presumably heat coagulation of the protein, was beginning to affect the results.
(4) With both organisms the value of the reaction velocity constant k was relatively slow at the start and tended to increase progressively during the course of disinfection. Whether it diminished again as sterility was approached could not be ascertained for technical reasons.
(5) With both organisms the presence of 1/1000 milk did not seem to affect the rate of disinfection as compared with distilled water.
(6) One experiment with Bact. coli suggested that the rate of disinfection was affected appreciably by the number of organisms present. Increasing the number of organisms in the suspension 100 times diminished the value of k by about one-third.
(7) The value of k was about 3,000,000 times greater with Bact. coli than with B. subtilis spores.
(8) If the mean value of n is taken as 2·2, it follows that doubling the concentration of caustic soda increases the reaction velocity by 4·6 times. If, for example, with a given concentration of caustic soda sterility was reached in 9 min., then doubling the concentration would reduce this time to 2 min., while halving the concentration would increase it to about 40 min.
(9) If the mean value of θ10°C. is taken as 1·75, it follows that a rise of 10° C. in the temperature increases the reaction velocity 1·75 times. If, for example, with a given temperature sterility was reached in 9 min., then raising the temperature 10° C. (18° F.) would reduce this time to about 5 min., while lowering the temperature 10° C. would increase it to about 15 min. With temperatures, however, of over 40° C. (104° F.) the rate of destruction of yegetative organisms, and with temperatures of over 70° C. (158° F.) the rate of destruction of sporing organisms in the presence of caustic soda is probably increased by the effect of the heat itself.
(10) A comparison of our figures for caustic soda with those of Chick for phenol shows that a 0°05% solution of caustic soda at 20° C. destroyed Bact. coli about five times as fast as a 0°5% solution of phenol at 20° C. destroyed Bact. paratyphosum B, and that a 5% solution of caustic soda at 30° C. destroyed B. subtilis spores nearly three times as fast as a 5 % solution of phenol at 33·3° C. destroyed anthrax spores. The superiority of caustic soda over phenol, particularly at concentrations likely to be used in practice, is manifest.
(11) Discussing the standardization of disinfectants, we conclude that the value of k taken in the middle stage of the reaction, or from the beginning of disinfection to about the end of the middle stage, affords the most suitable measure of comparison. This is essentially the same conclusion as that reached by Withell (1942), who uses as his index the time necessary to destroy 50% of the organisms.
Front matter
HYG volume 42 issue 4 Cover and Front matter
-
- Published online by Cambridge University Press:
- 15 May 2009, pp. f1-f8
-
- Article
-
- You have access Access
- Export citation
Back matter
HYG volume 42 issue 4 Cover and Back matter
-
- Published online by Cambridge University Press:
- 15 May 2009, p. b1
-
- Article
-
- You have access Access
- Export citation



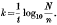 This method is complicated by the occurrence of lag period at the start of the reaction between organism and bactericide. This lag period varies in extent in different experiments (often extending up to 50% of the deaths) and makes the interpretation of the results difficult. The lag period has been shown to be a manifestation of, the distribution of logarithmic survival times (Withell, 1942), and is a function of the standard deviation of the logarithmic survivor times (λ). This fact is taken into account in method (2).
This method is complicated by the occurrence of lag period at the start of the reaction between organism and bactericide. This lag period varies in extent in different experiments (often extending up to 50% of the deaths) and makes the interpretation of the results difficult. The lag period has been shown to be a manifestation of, the distribution of logarithmic survival times (Withell, 1942), and is a function of the standard deviation of the logarithmic survivor times (λ). This fact is taken into account in method (2).