Article contents
Microwaves as a synthetic route for preparing electrochemically active TiO2 nanoparticles
Published online by Cambridge University Press: 24 September 2012
Abstract
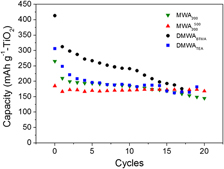
Nanocrystalline anatase was synthesized, using both domestic and laboratory microwave ovens, from different precursors. Nanoparticulate anatase was obtained after microwave irradiation of tetra-butyl orthotitanate solution in benzyl alcohol. As-synthesized samples have orange color due to the presence of organics that were eliminated after annealing at 500 °C, whereas the size of small anatase nanocrystals (around 8 nm) was preserved. Other nanocrystalline anatase samples were obtained from hexafluorotitanate-organic salt ionic liquid-like precursors. In this case, use of a domestic microwave oven and very short processing times (1–3 min irradiation time) were involved. Good specific capacity values and capacity retention at high C rates for insertion/deinsertion of Li+were recorded when testing such nanoparticles as electrode material in lithium cells. The electrochemical performances were found be strongly dependent on the phase composition, which in turn could be tuned through the synthetic procedure.
- Type
- Articles
- Information
- Journal of Materials Research , Volume 28 , Issue 3: Focus Issue: Titanium Dioxide Nanomaterials , 14 February 2013 , pp. 340 - 347
- Copyright
- Copyright © Materials Research Society 2012
References
REFERENCES
- 10
- Cited by


