Volume 78 - Issue 4 - August 2014
Research Article
Mineralogical characterization of calcification in cardiovascular aortic atherosclerotic plaque: A case study
- Yan Li, Xin Wang, Meiqian Zhu, Chong-Qing Yang, Anhuai Lu, Kang Li, Fanlu Meng, Changqiu Wang
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 775-786
-
- Article
- Export citation
-
Calcification in cardiovascular aortic atherosclerotic plaque contains Ca-phosphate minerals. However, most research on cardiovascular calcification has focused on its physiological properties rather than its mineralogical features. In this present study, cardiovascular calcification was characterized by collecting samples from patients’ tissues and applying mineralogical techniques. Synchrotron radiation-based micro-X-ray diffraction showed the calcification had a similar structure to hydroxylapatite (HAp). Transmission electron microscopy showed some structurally HAp-like spherical particles with a diameter of ∼200 nm and acicular crystals ∼100 nm × ∼20 nm in size. Selected-area electron diffraction indicated that these mineral particles belonged to the hexagonal crystal system. Fourier-transform infrared (FTIR) spectroscopy showed three typical peaks at 1469 cm−1, 1455 cm−1 and 1413 cm−1, indicating that the carbonate group in the calcification plaque substituted for a hydroxyl group to form B-type CHAp (Ca10(PO4,CO3)x(OH)y). The FTIR mapping results illustrated the intergrowth of calcification and organic tissues and the inhomogeneous substitution of phosphate by carbonate in the calcification area. X-ray absorption near-edge structure analysis affirmed that the chemical environments of Ca in the calcification were close to those in HAp. Based on these mineralogical characteristics, the calcification in plaque is identified as a mixture phase of HAp and B-type carbonate HAp, which is similar to the composition of bones.
Canutite, NaMn3[AsO4][AsO3(OH)]2, a new protonated alluaudite-group mineral from the Torrecillas mine, Iquique Province, Chile
- A. R. Kampf, S. J. Mills, F. Hatert, B. P. Nash, M. Dini, A. A. Molina Donoso
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 787-795
-
- Article
- Export citation
-
The new mineral canutite (IMA2013-070), NaMn3[AsO4][AsO3(OH)]2, was found at two different locations at the Torrecillas mine, Salar Grande, Iquique Province, Chile, where it occurs as a secondary alteration phase in association with anhydrite, halite, lavendulan, magnesiokoritnigite, pyrite, quartz and scorodite. Canutite is reddish brown in colour. It forms as prisms elongated on [20
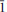 ] and exhibiting the forms {010}, {100}, {10
] and exhibiting the forms {010}, {100}, {10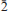 }, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm−3. Optically, canutite is biaxial (+) with α = 1.712(3), β = 1.725(3) and γ = 1.756(3) (measured in white light). The measured 2V is 65.6(4)°, the dispersion is r < v (slight), the optical orientation is Z = b; X ^ a = 18° in obtuse β and pleochroism is imperceptible. The mineral is slowly soluble in cold, dilute HCl. The empirical formula (for tabular crystals from near the mineshaft), determined from electron - microprobe analyses, is (Na1.05Mn2.64Mg0.34Cu0.14Co0.03)∑4.20As3O12H1.62. Canutite is monoclinic, C2/c, a = 12.3282(4), b = 12.6039(5), c = 6.8814(5) Å, β = 113.480(8)°, V = 980.72(10) Å3 and Z = 4. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 6.33(34)(020), 4.12(26)(
}, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm−3. Optically, canutite is biaxial (+) with α = 1.712(3), β = 1.725(3) and γ = 1.756(3) (measured in white light). The measured 2V is 65.6(4)°, the dispersion is r < v (slight), the optical orientation is Z = b; X ^ a = 18° in obtuse β and pleochroism is imperceptible. The mineral is slowly soluble in cold, dilute HCl. The empirical formula (for tabular crystals from near the mineshaft), determined from electron - microprobe analyses, is (Na1.05Mn2.64Mg0.34Cu0.14Co0.03)∑4.20As3O12H1.62. Canutite is monoclinic, C2/c, a = 12.3282(4), b = 12.6039(5), c = 6.8814(5) Å, β = 113.480(8)°, V = 980.72(10) Å3 and Z = 4. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 6.33(34)(020), 4.12(26)(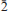 21), 3.608(29)(310,
21), 3.608(29)(310,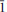 31), 3.296(57)(
31), 3.296(57)(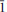 12), 3.150(28)(002,131), 2.819(42)(400,041,330), 2.740(100)(240,
12), 3.150(28)(002,131), 2.819(42)(400,041,330), 2.740(100)(240,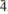 02,112) and 1.5364(31)(multiple). The structure, refined to R1 = 2.33% for 1089 Fo > 4σF reflections, shows canutite to be isostructural with protonated members of the alluaudite group.
02,112) and 1.5364(31)(multiple). The structure, refined to R1 = 2.33% for 1089 Fo > 4σF reflections, shows canutite to be isostructural with protonated members of the alluaudite group.
IMA Commission on New Minerals, Nomenclature and Classification (CNMNC) NewsLetter 21
New minerals and nomenclature modifications approved in 2014
- P. A. Williams, F. Hatert, M. Pasero, S. J. Mills
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 797-804
-
- Article
- Export citation
Research Article
The Rum Igneous Centre, Scotland
- C. H. Emeleus, V. R. Troll
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 805-839
-
- Article
-
- You have access Access
- Open access
- Export citation
-
The publication of the British Geological Survey memoir on Rum and the Small Isles in 1997 was followed by a period of intense petrological and mineralogical research, leading to some 40 papers, books and other publications. The research progress since then is reviewed here and integrated with the information previously available to provide an overview of the current status of understanding of the centre. New data on the acidic and mixed acid/basic magmas of the early Rum caldera demonstrate that frequent mafic replenishments were the main driver for magmatic activity at Rum right from its initial stages. The caldera is bound by the Main Ring Fault, a structure which probably also exercised an influence on the emplacement of the subsequent basic and ultrabasic intrusions. The later emplacement of gabbros and ultrabasic rocks caused only limited thermal metamorphism of the surrounding Torridonian sandstones, contrasting markedly with the crustal isotope signatures of the early intracaldera ignimbrite magmas and the intense alteration of uplifted masses of Lewisian gneiss within the ring fault. Rare picritic dykes provide an indication of the possible parent magma for the mafic and ultrabasic rocks, but these, as with most other magmatic rocks on Rum, have undergone varying degrees of crustal contamination, involving both Lewisian granulite and amphibolite-type crust but, notably, no Moine metasedimentary compositions as is the case at the nearby Ardnamurchan centre. Detailed textural studies on the gabbroic and ultrabasic rocks allow a distinction between intrusive peridotites and peridotite that forms part of the classic layered cumulate units of Rum and, furthermore, this work and that on the chromite seams and veins in these rocks shows that movement of trapped magma and magma derived from later intrusions, may produce textures regarded previously as of primary cumulate origin. Sulfides in the chromitite seams and ultrabasic rocks, in turn, show possible influences from assimilated Mesozoic sediments. Igneous activity on Rum was short-lived, possibly only between 0.5 and 1 m.y. in duration and commenced at ∼60.5 Ma. The Rum Central Complex was extinct by the time the main activity at the nearby Skye Central Complex commenced (∼59 Ma). From recent apatite fission-track studies it seems probable that Rum, in common with other Palaeogene centres, underwent a brief, but significantly later heating event (∼45 Ma).
On the complex H-bonding network in paravauxite, Fe2+Al2(PO4)2(OH)2·8H2O: A single-crystal neutron diffraction study
- G. D. Gatta, P. Vignola, M. Meven
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 841-850
-
- Article
- Export citation
-
The crystal structure and the chemical composition of a paravauxite from the Siglo Veinte Mine, Llallagua, Bustillo Province, Potosi Department, Bolivia [Fe(Fe0.9162+Mn0.0162+Mg0.064Ca0.002)∑0.998Al(1)Al(2)Al2.005P(P1.998Si0.002)∑2O8(OH)2·8H2O, a = 5.242(1) Å, b = 10.569(2) Å, c = 6.970(2) Å, α = 106.78(3)°, β = 110.81(2)° and γ = 72.29(2)°, space group P
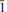 ], was investigated by single-crystal neutron diffraction and electron microprobe analysis in wavelength-dispersive mode. Neutron-intensity data were collected at 293 K and anisotropic structure refinement was performed. At the end of the refinement no peak larger than ±1.3 fm/Å3 was present in the final difference-Fourier map of the nuclear density. The final statistical index was R1 = 0.0495 for 194 refined parameters and 1678 unique reflections with Fo > 4σ(Fo). Eleven independent H sites (i.e. H(1), H(2), H(3), H(4A), H(4B), H(5), H(6), H(7), H(8), H(9A) and H(9B)), all at ∼1 Å from the respective O sites, were located successfully. H(4A) and H(4B) and H(9A) and H(9B) are two mutually exclusive subsite couples only 0.4−0.6 Å apart. The complex H-bonding scheme in the paravauxite structure is now well defined and 12 independent H bonds, with an energetically favourable bonding configuration, are described. A comparison between the previous experimental findings based on Raman and infrared spectroscopy and those obtained in this present study is carried out. Paravauxite provides the rare opportunity to investigate the H-bond configuration of coexisting hydroxyl groups and H2O molecules in minerals by single-crystal neutron diffraction. H2O is present as zeolitic (i.e. lying in the cavities) and non-zeolitic H2O (i.e. bonded to Al or Fe to form Al or Fe octahedra).
], was investigated by single-crystal neutron diffraction and electron microprobe analysis in wavelength-dispersive mode. Neutron-intensity data were collected at 293 K and anisotropic structure refinement was performed. At the end of the refinement no peak larger than ±1.3 fm/Å3 was present in the final difference-Fourier map of the nuclear density. The final statistical index was R1 = 0.0495 for 194 refined parameters and 1678 unique reflections with Fo > 4σ(Fo). Eleven independent H sites (i.e. H(1), H(2), H(3), H(4A), H(4B), H(5), H(6), H(7), H(8), H(9A) and H(9B)), all at ∼1 Å from the respective O sites, were located successfully. H(4A) and H(4B) and H(9A) and H(9B) are two mutually exclusive subsite couples only 0.4−0.6 Å apart. The complex H-bonding scheme in the paravauxite structure is now well defined and 12 independent H bonds, with an energetically favourable bonding configuration, are described. A comparison between the previous experimental findings based on Raman and infrared spectroscopy and those obtained in this present study is carried out. Paravauxite provides the rare opportunity to investigate the H-bond configuration of coexisting hydroxyl groups and H2O molecules in minerals by single-crystal neutron diffraction. H2O is present as zeolitic (i.e. lying in the cavities) and non-zeolitic H2O (i.e. bonded to Al or Fe to form Al or Fe octahedra).
Ba–Cu ordering in bariopharmacoalumite-Q2a2b2c from Cap Garonne, France
- I. E. Grey, W. G. Mumme, J. R. Price, S. J. Mills, C. M. Macrae, G. Favreau
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 851-860
-
- Article
- Export citation
-
Bariopharmacoalumite-Q2a2b2c, Ba0.5(Cu,ZnO)0.1H0.6[Al4(OH)4(As0.9Al0.1O4)3]·5.5H2O, from the south mine of the old copper mine at Cap Garonne, France, has a 2 × 2 × 2 I-centred tetragonal superstructure of the basic pharmacosiderite-type structure. Cell parameters are a = 15.405(2) Å and c = 15.553(3) Å. The structure was determined and refined in I
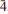 2m to R1= 0.057 for 2697 reflections with I > 2σ(I), using synchrotron X-ray data on a twinned crystal. The origin of the superlattice cell doubling was determined to be due predominantly to the ordering of Ba atoms in half of the [0 0 1] channels, centred at (0, 0, 0) and (½, ½, 0). The other channels, centred at (½, 0, 0) and (0, ½, 0), were found to be occupied by corner-connected chains of Cu/Zn-centred square planar units.
2m to R1= 0.057 for 2697 reflections with I > 2σ(I), using synchrotron X-ray data on a twinned crystal. The origin of the superlattice cell doubling was determined to be due predominantly to the ordering of Ba atoms in half of the [0 0 1] channels, centred at (0, 0, 0) and (½, ½, 0). The other channels, centred at (½, 0, 0) and (0, ½, 0), were found to be occupied by corner-connected chains of Cu/Zn-centred square planar units.
Ferri-fluoro-leakeite: a second occurrence at Bratthagen (Norway), with new data on Zn partitioning and the oxo component in Na amphiboles
- R. Oberti, M. Boiocchi, F. C. Hawthorne, R. Kristiansen
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 861-869
-
- Article
- Export citation
-
A second occurrence of ferri-fluoro-leakeite has been identified in the Bratthagen nepheline syenite pegmatite (Vestfold County, Norway). With respect to the holotype found at the Verkhnee Espe deposit (Akjailyautas Mountains, Kazakhstan; Cámara et al., 2010), it is closer to the ideal composition because of its larger Li and Mg contents and the absence of an oxo-component; however, it has a significant Zn content (0.29 a.p.f.u.). The ideal formula of ferri-fluoro-leakeite is ANaBNa2C(Mg2Fe23+Li)TSi8O22WF2 and the empirical formula derived from electron-microprobe analysis and single-crystal structure refinement for the sample used here is A(Na0.68K0.32)S=1.00BNa2.00 C(Mg1.69Mn0.252+Fe0.242+Zn0.29Al0.23Fe1.503+Ti0.02Li0.78)S=5.00TSi8O22W(F1.59(OH)0.41)S=2.00. Unitcell data are a = 9.788(2), b = 17.826(3), c = 5.282(1) Å, β = 104.195(5)°, V = 893.5 (3) Å3, Z = 2. Crystal-chemical analysis shows that Li is ordered at the M(3) site and Zn is ordered mainly at the M(2) site, confirming previous findings for Li-bearing amphiboles. The new data also make quantification of the oxo component in Na amphiboles possible.
Coquandite, Sb6+xO8+x(SO4)(OH)x·(H2O)1–x (x = 0.3), from the Cetine mine, Tuscany, Italy: crystal structure and revision of the chemical formula
- L. Bindi, C. Biagioni, L. Ceccantini, M. Batoni, S. Menchetti
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 871-888
-
- Article
- Export citation
-
The crystal structure of the mineral coquandite, a rare Sb oxy-sulfate hydrate, was solved using intensity data collected from a crystal from the Cetine mine, Tuscany, Italy. This study revealed that the structure is triclinic, space group P
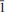 , with a = 11.4292(5), b = 29.772(1), c = 11.2989(5) Å, α = 91.152(3), β = 119.266(4), γ = 92.624(3)° and V = 3346.4(2) Å3. The refinement of an anisotropic model led to an R index of 0.0347 for 21,061 independent reflections. Thirty-two Sb sites, five S sites and 67 oxygen sites occur in the crystal structure of coquandite. Sb atoms display the characteristic SbO3E and SbO4E coordinations whereas S fills (SO4) tetrahedral groups. These atoms are arranged in five symmetry-independent layers perpendicular to b*. Four of them and their centrosymmetrical counterparts form complex modules stacked along b* and bonded through two Sb atoms and H bonds. The complex H bonding system in the structure is discussed. On the basis of information gained from this characterization, the crystal-chemical formula was revised according to the structural results, yielding Sb6+xO8+x(SO4)(OH)x·(H2O)1–x (Z = 10) with x = 0.3 instead of Sb6O8(SO4)·H2O (Z = 12) as reported previously. A recalculation of the chemical data listed in the scientific literature for coquandite according to the structural results obtained here leads to a satisfactory agreement.
, with a = 11.4292(5), b = 29.772(1), c = 11.2989(5) Å, α = 91.152(3), β = 119.266(4), γ = 92.624(3)° and V = 3346.4(2) Å3. The refinement of an anisotropic model led to an R index of 0.0347 for 21,061 independent reflections. Thirty-two Sb sites, five S sites and 67 oxygen sites occur in the crystal structure of coquandite. Sb atoms display the characteristic SbO3E and SbO4E coordinations whereas S fills (SO4) tetrahedral groups. These atoms are arranged in five symmetry-independent layers perpendicular to b*. Four of them and their centrosymmetrical counterparts form complex modules stacked along b* and bonded through two Sb atoms and H bonds. The complex H bonding system in the structure is discussed. On the basis of information gained from this characterization, the crystal-chemical formula was revised according to the structural results, yielding Sb6+xO8+x(SO4)(OH)x·(H2O)1–x (Z = 10) with x = 0.3 instead of Sb6O8(SO4)·H2O (Z = 12) as reported previously. A recalculation of the chemical data listed in the scientific literature for coquandite according to the structural results obtained here leads to a satisfactory agreement.
The crystal structure of arangasite, Al2F(PO4)(SO4)·9H2O determined using low-temperature synchrotron data
- O. V. Yakubovich, I. M. Steele, V. V. Chernyshev, N. V. Zayakina, G. N. Gamyanin, O. V. Karimova
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 889-903
-
- Article
- Export citation
-
The crystal structure of the fibrous mineral arangasite, Al2F(PO4)(SO4)·9H2O from the Alyaskitovoje deposit, Eastern Yakutiya, Russia, was solved using low-temperature single-crystal data from synchrotron radiation and refined against F2 to R = 9.8%. Arangasite crystallizes in the monoclinic space group P2/a, with unit-cell parameters a = 7.073(1), b = 9.634(2), c = 10.827(2) Å, β = 100.40(1)°, V = 725.7(7) Å3 and Z = 2. The positions of all the independent H atoms were obtained by difference- Fourier techniques and refined in an isotropic approximation. The arangasite crystal structure is built from one-dimensional chains of Al octahedra and PO4 tetrahedra sharing vertices, quasi-isolated SO4 tetrahedra and H2O molecules. All O atoms are involved in the system of H bonding, acting as donors and/or acceptors. Hydrogen bonding serves as the only mechanism providing linkage between the main structural fragments, thus maintaining the framework. Chains of corner-sharing Al octahedra and P tetrahedra in the arangasite structure are topologically identical to the chains built from (Fe, Al) octahedra and P tetrahedra in the crystal structure of destinezite, Fe2(OH)(PO4)(SO4)·6H2O. It has been shown that in spite of very similar chemical formulae, arangasite and sanjuanite, Al2(OH)(PO4)(SO4)·9H2O, are not isotypic.
New arsenate minerals from the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. I. Yurmarinite, Na7(Fe3+,Mg,Cu)4(AsO4)6
- I. V. Pekov, N. V. Zubkova, V. O. Yapaskurt, D. I. Belakovskiy, I. S. Lykova, M. F. Vigasina, E. G. Sidorov, D. Yu. Pushcharovsky
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 905-917
-
- Article
- Export citation
-
A new mineral, yurmarinite, Na7(Fe3+,Mg,Cu)4(AsO4)6, occurs in sublimates of the Arsenatnaya fumarole at the Second scoria cone of the Northern Breakthrough of the Great Tolbachik Fissure Eruption, Tolbachik volcano, Kamchatka, Russia. It is associated with hatertite, bradaczekite, johillerite, hematite, tenorite, tilasite and aphthitalite. Yurmarinite occurs as well-shaped, equant crystals up to 0.3 mm in size, their clusters up to 0.5 mm and thin, interrupted crystal crusts up to 3 mm × 3 mm on volcanic scoria. Crystal forms are {101}, {011}, {100}, {110} and {001}. Yurmarinite is transparent, pale green or pale yellowish green to colourless. The lustre is vitreous and the mineral is brittle. The Mohs hardness is ∼4½. One direction of imperfect cleavage was observed, the fracture is uneven. D(calc.) is 4.00 g cm−3. Yurmarinite is optically uniaxial (−), ω = 1.748(5), ε = 1.720(3). The Raman spectrum is given. The chemical composition (wt.%, electron microprobe data) is Na2O 16.85, K2O 0.97, CaO 1.28, MgO 2.33, MnO 0.05, CuO 3.17, ZnO 0.97, Al2O3 0.99, Fe2O3 16.44, TiO2 0.06, P2O5 0.12, V2O5 0.08, As2O5 56.68, total 99.89. The empirical formula, calculated on the basis of 24 O atoms per formula unit, is (Na6.55Ca0.28K0.22)S7.05(Fe2.483+Mg0.70Cu0.48Al0.23Zn0.14Ti0.01Mn0.01)S4.05(As5.94P0.02V0.01)S5.97O24. Yurmarinite is rhombohedral, R
 c, a = 13.7444(2), c = 18.3077(3) Å, V = 2995.13(8) Å3, Z = 6. The strongest reflections in the X-ray powder pattern [d, Å (I)(hkl)] are: 7.28(45)(012); 4.375(33)(211); 3.440(35)(220); 3.217(36)(131,214); 2.999(30)(223); 2.841(100)(125); 2.598(43)(410). The crystal structure was solved from single-crystal X-ray diffraction data to R = 0.0230. The structure is based on a 3D heteropolyhedral framework formed by M4O18 clusters (M = Fe3+ > Mg,Cu) linked with AsO4 tetrahedra. Sodium atoms occupy two octahedrally coordinated sites in the voids of the framework. In terms of structure, yurmarinite is unique among minerals but isotypic with several synthetic compounds with the general formula (Na7–x☐x)(M3+x3+M1–x2+)(T5+O4)2 in which T = As or P, M3+ = Fe or Al, M2+ = Fe and 0 ≤ x ≤ 1. The mineral is named in honour of the Russian mineralogist, petrologist and specialist in studies of ore deposits, Professor Yuriy B. Marin (b. 1939). The paper also contains a description of the Arsenathaya fumarole and an overview of arsenate minerals formed in volcanic exhalations.
c, a = 13.7444(2), c = 18.3077(3) Å, V = 2995.13(8) Å3, Z = 6. The strongest reflections in the X-ray powder pattern [d, Å (I)(hkl)] are: 7.28(45)(012); 4.375(33)(211); 3.440(35)(220); 3.217(36)(131,214); 2.999(30)(223); 2.841(100)(125); 2.598(43)(410). The crystal structure was solved from single-crystal X-ray diffraction data to R = 0.0230. The structure is based on a 3D heteropolyhedral framework formed by M4O18 clusters (M = Fe3+ > Mg,Cu) linked with AsO4 tetrahedra. Sodium atoms occupy two octahedrally coordinated sites in the voids of the framework. In terms of structure, yurmarinite is unique among minerals but isotypic with several synthetic compounds with the general formula (Na7–x☐x)(M3+x3+M1–x2+)(T5+O4)2 in which T = As or P, M3+ = Fe or Al, M2+ = Fe and 0 ≤ x ≤ 1. The mineral is named in honour of the Russian mineralogist, petrologist and specialist in studies of ore deposits, Professor Yuriy B. Marin (b. 1939). The paper also contains a description of the Arsenathaya fumarole and an overview of arsenate minerals formed in volcanic exhalations.
Asbestiform sepiolite coated by aliphatic hydrocarbons from Perletoa, Aosta Valley Region (Western Alps, Italy): characterization, genesis and possible hazards
- R. Giustetto, K. Seenivasan, E. Belluso
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 919-940
-
- Article
- Export citation
-
An atypical asbestiform sepiolite occurrence with exceptionally long fibres wrapped by a sheath of aliphatic hydrocarbons was found in the Gressoney Valley (Italian Western Alps) while monitoring asbestos presence in outcrops of serpentinite rocks. Microscopic and Fourier transform infrared analyses proved that these fibres, apparently up to several cm long, are formed by bundles of thinner fibrils (average length: 150 μm) potentially dispersible in the environment. When observed using transmission electron microscopy these fibrils show a rhomboidal to parallelogram cross section (<1 μm), of which surfaces are covered mostly by an aliphatic hydrocarbon film – an association not reported in the literature. The sepiolite fibrils and their organic coating probably originated in sequential steps from precipitation of Si/Mg rich hydrothermal fluids, resulting from serpentinization of olivine and clinopyroxene and a Fischer-Tropsch-type reaction. The presence of hydrocarbons has serious implications for the sepiolite habit, as the organic wrap interacts with the fibril’s surface reducing the amount of adsorbed water and favouring the fragmentation of thicker units into thinner ones, due to an ‘opening’ process implying separation along z and cleavage on (110). This defibrillation mechanism, coupled with the extraordinary length, further increases the aspect ratio of these fibrils (length/width ≫3) thus amplifying their potential danger for human health when dispersed in air and inhaled.
The formation of ferromanganese nodules in the southwest Indian Ocean; an abiotic process
- B. E. Van Dongen, N. J. Ashton, R. A. D. Pattrick
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 941-955
-
- Article
- Export citation
-
Ferromanganese nodules have been recognized widely as potentially important resources for strategic metals. However it remains unclear if the formation of these nodules is purely an abiotic process or if microorganisms are involved in their formation. To determine the microbial contributions, detailed organic geochemical analyses were performed on ferromanganese nodules collected from across the southwest Indian Ocean. These analyses reveal the presence of specific terrestrial, marine and petroleum derived biomarkers, consistent with formation in a marine setting with a substantial influx of terrestrially derived (higher plant detritus) and naturally occurring petroleum-related organic matter. In contrast, only trace amounts of general bacterial biomarkers, commonly present in these types of depositional environments, were present. This indicates that the formation of these ferromanganese nodules is predominantly an abiotic process although a minor contribution from microbial mediated processes to the growth of these nodules cannot be completely ruled out.
The structure hierarchy hypothesis
- F. C. Hawthorne
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 957-1027
-
- Article
- Export citation
-
The structure hierarchy hypothesis states that structures may be ordered hierarchically according to the polymerization of coordination polyhedra of higher bond valence. A mathematical hierarchy is an ordered set of elements where the ordering reflects a natural hierarchical relation between (or arrangement of) the elements. Here, I review the structure hierarchies for the borate, uranyl oxide, phosphate, sulfate, beryllate and oxide-centred Cu, Pb and Hg minerals (plus synthetics where appropriate). Structure hierarchies have two functions: (1) they serve to organize our knowledge of minerals (crystal structures) in a coherent manner; (2) if the basis of the classification involves factors that are related to the mechanistic details of the stability and behaviour of minerals, then the physical, chemical and paragenetic characteristics of minerals should arise as natural consequences of their crystal structures and the interaction of those structures with the environment in which they occur. We may justify the structure hierarchy hypothesis by considering a hypothetical structure-building process whereby higher bond-valence polyhedra polymerize to form the structural unit. The clusters constituting the FBBs (fundamental building blocks) may polymerize to form the following types of structural unit: (1) isolated polyhedra; (2) clusters; (3) chains and ribbons; (4) sheets; and (5) frameworks. The major advantage of this approach to structure hierarchy is the fact that the hypothetical structure-building process outlined above resembles (our ideas of) crystallization from an aqueous solution, whereby complexes in aqueous and hydrothermal solutions condense to form crystal structures, or fragments of linked polyhedra in a magma condense to form a crystal. Although our knowledge of these processes is rather vague from a mechanistic perspective, the foundations of the structure hypothesis give us a framework within which to think about the processes of crystallization and dissolution.
Major element and oxygen isotope geochemistry of vapour-phase garnet from the Topopah Spring Tuff at Yucca Mountain, Nevada, USA
- R. J. Moscati, C. A. Johnson
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1029-1041
-
- Article
- Export citation
-
Twenty vapour-phase garnets were studied in two samples of the Topopah Spring Tuff of the Paintbrush Group from Yucca Mountain, in southern Nevada. The Miocene-age Topopah Spring Tuff is a 350 m thick, devitrified, moderately to densely welded ash-flow tuff that is zoned compositionally from high-silica rhyolite to latite. During cooling of the tuff, escaping vapour produced lithophysae (former gas cavities) lined with an assemblage of tridymite (commonly inverted to cristobalite or quartz), sanidine and locally, hematite and/or garnet. Vapour-phase topaz and economic deposits associated commonly with topaz-bearing rhyolites (characteristically enriched in F) were not found in the Topopah Spring Tuff at Yucca Mountain. Based on their occurrence only in lithophysae, the garnets are not primary igneous phenocrysts, but rather crystals that grew from a F-poor magmaderived vapour trapped during and after emplacement of the tuff. The garnets are euhedral, vitreous, reddish brown, trapezohedral, as large as 2 mm in diameter and fractured. The garnets also contain inclusions of tridymite. Electron microprobe analyses of the garnets reveal that they are almandinespessartine (48.0 and 47.9 mol.%, respectively), have an average composition of (Fe1.46Mn1.45Mg0.03Ca0.10)(Al1.93Ti0.02)Si3.01O12 and are comparatively homogeneous in Fe and Mn concentrations from core to rim. Composited garnets from each sample site have δ18O values of 7.2 and 7.4%. The associated quartz (after tridymite) has δ18O values of 17.4 and 17.6%, values indicative of reaction with later, low-temperature water. Unaltered tridymite from higher in the stratigraphic section has a δ18O of 11.1% which, when coupled with the garnet δ18O values in a quartz-garnet fractionation equation, indicates isotopic equilibration (vapour-phase crystallization) at temperatures of ∼600°C. This high-temperature mineralization, formed during cooling of the tuffs, is distinct from the later and commonly recognized low-temperature stage (generally 50−70°C) of calcite, quartz and opal secondary mineralization, formed from downward-percolating meteoric water, that locally coats fracture footwalls and lithophysal floors.
Balićžunićite, Bi2O(SO4)2, a new fumarole mineral from La Fossa crater, Vulcano, Aeolian Islands, Italy
- D. Pinto, A. Garavelli, D. Mitolo
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1043-1055
-
- Article
- Export citation
-
Balićžunićite, ideally Bi2O(SO4)2, is a new mineral found as a high-temperature fumarole sublimate (T = 600°C) at La Fossa crater, Vulcano, Aeolian Islands, Italy. It occurs as aggregates of mm-sized prismatic and elongated crystals (∼50 μm across and up to 200 μm long) associated with anglesite, leguernite, one other potentially new Bi-oxysulfate mineral, lillianite, galenobismutite, bismoclite, Cd-rich sphalerite, wurtzite, pyrite and pyrrhotite. Balićžunićite is colourless to white or pale brown, transparent and non-fluorescent. It has a vitreous lustre and a white streak. Electron microprobe analysis gives the following average chemical composition (wt.%): Bi2O3 68.68 and SO3 23.73, total 92.41. The empirical chemical formula, calculated on the basis of 9 anions p.f.u., is Bi1.99S2O9. The calculated density is 5.911 g/cm3.
Balićžunićite is triclinic, space group P
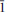 , with a 6.7386(3), b 11.1844(5), c 14.1754(7) Å, α 80.082(2)°, β 88.462(2)°, γ 89.517(2)°, V = 1052.01(8) Å3 and Z = 6. The six strongest reflections in the X-ray powder-diffraction data [d in Å(I) (hkl)] are: 3.146 (100) (033), 3.486 (21) (004), 3.409 (12) (0
, with a 6.7386(3), b 11.1844(5), c 14.1754(7) Å, α 80.082(2)°, β 88.462(2)°, γ 89.517(2)°, V = 1052.01(8) Å3 and Z = 6. The six strongest reflections in the X-ray powder-diffraction data [d in Å(I) (hkl)] are: 3.146 (100) (033), 3.486 (21) (004), 3.409 (12) (0 1), 3.366 (7) (200), 5.562 (4) (
1), 3.366 (7) (200), 5.562 (4) (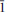 11), 5.433 (4) (111). Balićžunićite is the natural analogue of the stable low-temperature a form of synthetic Bi2O(SO4)2. The name is in honour of Tonci Balić-Žunić(born 1952), Professor of Mineralogy at the Natural History Museum of the University of Cophenagen. Both the mineral and the mineral name have been approved by the IMA-CNMNC Commission (IMA2012-098).
11), 5.433 (4) (111). Balićžunićite is the natural analogue of the stable low-temperature a form of synthetic Bi2O(SO4)2. The name is in honour of Tonci Balić-Žunić(born 1952), Professor of Mineralogy at the Natural History Museum of the University of Cophenagen. Both the mineral and the mineral name have been approved by the IMA-CNMNC Commission (IMA2012-098).
Crystal-chemical study of wavellite from Zbirov, Czech Republic
- F. Capitelli, G. Della Ventura, F. Bellatreccia, A. Sodo, M. Saviano, M. R. Ghiara, M. Rossi
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1057-1070
-
- Article
- Export citation
-
The crystal chemistry of wavellite from Zbirov (Czech Republic), ideally Al3(PO4)2(OH,F)3·5H2O, was addressed by means of a multi-methodological approach based on electron microprobe analysis (EMPA) using wave-dispersive spectroscopy, single-crystal X-ray diffraction, powder and singlecrystal infrared spectroscopy and Raman spectroscopy. The EMPA data showed the presence of significant F replacing OH in the sample studied. The structure was solved in the Pcmn orthorhombic space group, with the following unit-cell constants: a = 9.6422(7), b = 17.4146(15), c = 7.0094(2) Å, V = 1176.98(10) Å3. Phosphorus atoms display tetrahedral (PO4) coordination, while Al cations display octahedral coordination. The mineral framework can be viewed as the repetition of cationic arrays made up of AlO6 polyhedra, bridged by PO4 groups and further joined by O–H⋯O hydrogen bonds. The single-crystal unpolarized Fourier transform infrared (FTIR) spectrum shows combination bands indicating the presence of both OH and H2O in the structure. Both FTIR and Raman spectra show a broad absorption extending from 3600 to 2800 cm−1 resulting from the overlapping of several components due to the water molecules and the OH group. The frequencies observed are comparable to those expected on the basis of the Libowitzky relationship for the range of D–H⋯A bond systems in the structure.



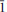 ] and exhibiting the forms {010}, {100}, {10
] and exhibiting the forms {010}, {100}, {10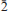 }, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm
}, {201} and {102}, or as tablets flattened on {102} and exhibiting the forms {102} and {110}. Crystals are transparent with a vitreous lustre. The mineral has a pale tan streak, Mohs hardness of 2½, brittle tenacity, splintery fracture and two perfect cleavages, on {010} and {101}. The calculated density is 4.112 g cm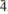 02,112) and 1.5364(31)(multiple). The structure, refined to
02,112) and 1.5364(31)(multiple). The structure, refined to 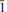 ], was investigated by single-crystal neutron diffraction and electron microprobe analysis in wavelength-dispersive mode. Neutron-intensity data were collected at 293 K and anisotropic structure refinement was performed. At the end of the refinement no peak larger than ±1.3 fm/Å
], was investigated by single-crystal neutron diffraction and electron microprobe analysis in wavelength-dispersive mode. Neutron-intensity data were collected at 293 K and anisotropic structure refinement was performed. At the end of the refinement no peak larger than ±1.3 fm/Å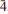 2
2 , with
, with 
 , with
, with  1), 3.366 (7) (200), 5.562 (4) (
1), 3.366 (7) (200), 5.562 (4) (