Introduction
Schrankiana Strand, 1942 are gastrointestinal parasites of anurans, distributed throughout Central and South America (Campião et al., Reference Campião, Morais, Dias, Aguiar, Toledo, Tavares and Silva2014; González and Hamann, Reference González and Hamann2014; González et al., Reference González, Hamann and Duré2021a). To date, Schrankiana comprises 8 species, and most of them were reported parasitising anurans belonging to Leptodactylidae from Brazil (Baker and Vaucher, Reference Baker and Vaucher1988; González and Hamann, Reference González and Hamann2014; Campião et al., Reference Campião, Ribas, Silva, Dalazen and Tavares2017; Carmo et al., Reference Carmo, Carvalho, Ávila and Vrcibradic2024).
The systematic status, phylogenetic position and evolutionary history of Schrankiana remain uncertain. The genus has been allocated in Atractidae (Chabaud, Reference Chabaud, Anderson, Chabaud and Willmott2009; González and Hamann, Reference González and Hamann2014; Campião et al., Reference Campião, Silva, Dalazen, Paiva and Tavares2016; Da Graça et al., Reference Da Graça, Oda, Lima, Guerra, Gambale and Takemoto2017; González et al., Reference González, Duré, Palomas, Schaefer, Etchepare and Acosta2021b; Chero et al., Reference Chero, Cruces, Cacique, Ponce, Iannacone, Alvariño, Sanchez, Sáez, Lopez and Silva2023). However, Adamson and Baccam (Reference Adamson and Baccam1988) and Gibbons (Reference Gibbons and Gibbons2010) transferred Schrankiana to Cosmocercidae.
Currently, the identification of Schrankiana spp. is based on meristic data with a few qualitative characters. These species exhibit morphological similarities, and their measurements often overlap. Freitas (Reference Freitas1959) described Schrankiana formosula in Leptodactylus fuscus (Schneider, 1799) from the state of Rio de Janeiro and S. inconspicata in Leptodactylus labyrinthicus Spix (1824) from the state of Salvador, Brazil. Despite the comprehensive morphological description, the authors did not state clear differences between both species (see Freitas, Reference Freitas1959; Baker and Vaucher, Reference Baker and Vaucher1988). Additionally, those 2 species are among the most commonly reported parasitising anurans from Brazil (Goldberg et al., Reference Goldberg, Bursey, Caldwell, Vitt and Costa2007, Reference Goldberg, Bursey, Caldwell and Shepard2009; Campião et al., Reference Campião, Morais, Dias, Aguiar, Toledo, Tavares and Silva2014, Reference Campião, Ribas, Silva, Dalazen and Tavares2017).
In the present study, we found nematodes parasitising Leptodactylus pentadactylus (Laurenti, 1768) that resembled S. formosula. However, due to morphological similarities among Schrankiana spp. we re-examined the type series S. formosula and S. inconspicata and observed that the species are morphologically identical. Thus, we provide a redescription of S. formosula, including an analyses of the intraspecific morphological variability, scanning electron microscope (SEM) characterization and a proposal of the systematic relationships of the genus using DNA.
Materials and methods
Host collection, morphological studies and map of species distribution
During a helminthological survey carried out in September 2021, 12 specimens of L. pentadactylus were collected by an active search in the ‘Beija-flor Brilho de Fogo’ Extractive Reserve, Pedra Branca do Amapari municipality, state of Amapá, Brazil (0°47′30.6″N, 51°58′42.1″W).
After collection, the anurans were anaesthetised, measured, weighed and necropsied for helminthological examination. Nematodes were removed from the digestive tract, washed in saline solution (NaCl 0.9%), killed in heated 70% ethanol and preserved in the same solution at −20°C. For morphological and morphometric analyses, the nematodes were washed in distilled water, cleared in glycerin, mounted on temporary slides and examined under an Olympus BX41 microscope (Olympus, Tokyo, Japan) coupled with a drawing tube (without zoom adjustment). The illustrations were prepared using the CorelDraw 2021 software and processed using Adobe Photoshop Version 21.0.2 software.
We re-examined the type series of S. formosula from state of Rio de Janeiro (holotype: CHIOC 22595a; allotype: CHIOC 22595b and paratypes: CHIOC 22579a, c, d, f, g, h, i, j; 22596e) and S. inconspicata from state of Bahia (holotype: CHIOC 22578a; allotype: CHIOC 22578b and paratypes: CHIOC 22579 e, g, h, i, j, n; CHIOC 29579m–n) deposited in the helminthological collection of the Oswaldo Cruz Institute (CHIOC) of Rio de Janeiro, Brazil. The prevalence and mean intensity values are reported according to Bush et al. (Reference Bush, Lafferty, Lotz and Shostak1997). The amphibian hosts were identified according to Frost (Reference Frost2024).
A total of 4 nematodes (males and females) were post-fixed in 1% osmium tetroxide (OsO4), dehydrated in an increasing ethanol series and critical-point dried in carbon dioxide (CO2). Subsequently, the helminths were mounted on metallic stubs, coated with gold-palladium, and examined under a Vega3 (TESCAN, Brno, Czech Republic) SEM in the Laboratory of Structural Biology, Biological Sciences Institute, Federal University of Pará (UFPA), Brazil.
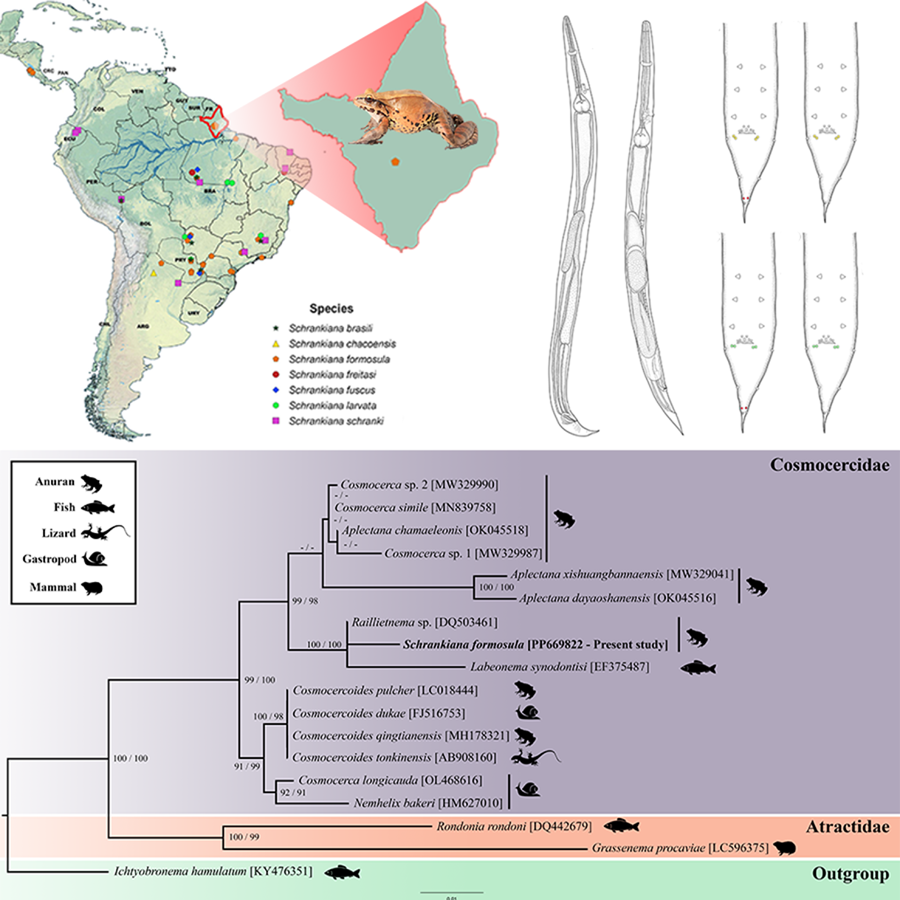
We conducted a bibliographic reference search to compile the records of Schrankiana species, using 7 electronic databases (Google, Google Scholar, PubMed, Scielo, Science Direct, Scopus and Web of Science). The keywords were combined amongst themselves: Atractidae, Cosmocercidae, Schrankiana, Helminths and Leptodactylidae. Species without specific diagnosis (‘gr.’, ‘af.’ and ‘sp.’) were excluded from our checklist. Additionally, a map illustrating the distribution of Schrankiana spp. was generated using a spreadsheet and QGIS 3.28 software (Quantum, 2024)). This compilation included published records, data available and information from the present study.
Comparative Data analyses
We also tested 22 variables in males and 26 variables in females of S. formosula from L. pentadactylus (present study) and re-examined the type series of S. formosula from L. fuscus and S. formosula (= S. inconspicata) from L. pentadactylus using a principal component analysis (PCA) to assess the importance of each variable within the dataset and its applied variance. We followed the methodology proposed by González et al. (Reference González, Valeria and Monika2019).
After this ordination analyses, we reduced the multivariate dataset into a smaller group of composite variables with a limited loss of information (McGarigal et al., Reference Mcgarigal, Cushman and Stavord2000). Thus, we applied multivariate analysis of variance (MANOVA), including the most relevant components according to the PCA to test the hypothesis that there are differences between metric variables of males and females of the 3 groups analysed.
In cases of Pillai was significant, a 2-factor ANOVA was performed for each variable. Before the analyses, variables were logarithmically transformed [ln(x)] to fit a normal distribution. Analyses were performed using the PAST 4.11 (Hammer et al., Reference Hammer, Harper and Ryan2001).
Molecular analyses and phylogenetic study
A male specimen was preserved in 100% ethanol and stored in a freezer at −20°C for molecular analyses. Genomic DNA was extracted using NucleoSpin Tissue (Macherey-Nagel, Düren, Germany) according to the manufacturer's instructions. The SSU rDNA nuclear gene (18S) was amplified using the protocol and primers described in Gomes et al. (Reference Gomes, Olifiers, Santos, Simões and Maldonado-Júnior2015). The resulting amplicons were visualized on 1.5% agarose gel electrophoresis with GelRed Nucleic Acid Stain (Biotium, Hayward, California, USA) under Uv light transilluminator. PCR products were purified Illustra GFX PCR DNA and Gel Band kit (GE Healthcare, Chicago, IL, USA) according to the manufacturer's instructions and sequenced using the BigDye Terminator v3.1 Cycle Sequencing kit (Applied Biosystems, USA). Amplicons were sequenced on Applied Biosystems™ 3730 DNA Analyser at the DNA Sequencing Platform of the Oswaldo Cruz Foundation (RPT01A/PDTIS/FIOCRUZ).
Contiguous sequences were assembled in Geneious 7.1.3 (Kearse et al., Reference Kearse, Moir, Wilson, Stones-Havas, Cheung, Sturrock and Drummond2012) and deposited in the National Center for Biotechnology Information (NCBI) (http://www.ncbi.nml.nih.gov). The 18S rDNA datasets were aligned and trimmed using Muscle (Edgar, Reference Edgar2004) in Geneious 7.1.3 software (Kearse et al., Reference Kearse, Moir, Wilson, Stones-Havas, Cheung, Sturrock and Drummond2012). We obtained the saturation-substitutions index of each aligned matrix using the software DAMBE 5 (Xia, Reference Xia2013). The genetic divergence was conducted in the MEGA11 software package (Kimura, Reference Kimura1980; Tamura et al., Reference Tamura, Peterson, Peterson, Stecher, Nei and Kumar2011). The most appropriate evolutionary nucleotide substitution model was GTR + I + G, determined by the Akaike Information Criterion in the jModelTest program (Posada, Reference Posada2008).
Phylogenetic reconstructions were performed using Maximum Likelihood (ML) in RAxML 8.2.12 and Bayesian Inference (BI) in MrBayes 3.2.7a software, respectively (Guindon and Gascuel, Reference Guindon and Gascuel2003; Ronquist and Huelsenbeck, Reference Ronquist and Huelsenbeck2003). Both analyses were conducted in CIPRES Science Gateway (Miller et al., Reference Miller, Pfeiffer and Schwartz2010). ML inference was performed using bootstrap support values of 1000 repetitions, and only nodes with a bootstrap percentage greater than 70% were considered well-supported.
Bayesian analyses employed the following settings for the dataset: Iset nst = 6, rates = invgamma, ngammacat = 4, nucmodel = 4by4, code = universal, prset statefreqpr = dirichlet (1,1,1,1), shapepr = fixed (0.5390) and pinvar = fixed (0.5290). Markov chain Monte Carlo (MCMC) search chains were run with 10 000 000 generations, saving 1 tree every 1500 generations. The first 25 000 generations were discarded on the burn-in, and the consensus tree (majority rule) was estimated using the remaining topologies, and we added commands sumt relburnin = yes, and sump relburnin = yes. Only nodes with Bayesian posterior probabilities greater than 90% were considered well-supported. The trees were visualized and edited in the software FigTree v1.3.3 (Rambaut, Reference Rambaut2009). We used Ichtyobronema hamulatum (Moulton, 1931) (access number: KY476351) as an outgroup in all phylogenies.
Results
Systematics
Family: Cosmocercidae Travassos, Reference Travassos1925
Genus: Schrankiana Strand, 1942
Species: Schrankiana formosula Freitas, Reference Freitas1959
Taxonomic summary
Type host: Leptodactylus fuscus (Schneider, 1799) (Amphibia: Leptodactylidae)
Additional hosts: Leptodactylus pentadactylus (Laurenti, 1768); Leptodactylus labyrinthicus (Spix, 1824); Leptodactylus elenae Heyer, 1978.
Type locality: state of Rio de Janeiro, Brazil (22°54′10″N, 43°12′28″W)
Additional localities: ‘Beija-Flor Brilho de Fogo’ Extractive Reserve, Pedra Branca do Amapari municipality, state of Amapá, Brazil (0°47′30.6″N, 51°58′42.1″W) (present study); state of Bahia, Brazil (24°3′47.47″S, 54°18′50.14″W); Salto del Guiara municipality, department of Canindeyú, Paraguay (24°1′12″N, 54°20′24″W); Arroyo Itabo Guazu municipality, department of Alto Paraná, Paraguay (25°4′60″S, 54°40′0″W); Coronel Oviedo municipality, department of Caaguazú, Paraguay (25°26′60.00″S, 56°01′0.01″W)
Site of infection: large intestine
Parasitological descriptors: Prevalence 33% (4 of 12 analysed); mean intensity (230.5) and abundance (76.8). The infection parameters will be based only on the material collected in Amapá.
Voucher material: 15 males (MPEG 000289) and 15 females (MPEG 000290) were deposited in the Emílio Goeldi Paraense Museum.
GenBank Accession number: PP669822
Description (morphological description based on re-examination of type series and new material collected). Small slender nematodes (Fig. 1A and B). Cuticle with thin transverse striations (Fig. 2A and B). Sexual dimorphism evident, females larger than males (Fig. 1A and B). Lateral alae, weakly developed, extending from anterior third of oesophagus to just anterior to anus in both sexes (Fig. 2A). Somatic papillae, distributed over body surface. Oral opening triangular with 3 distinct lips; dorsal lip with a pair of papillae; subventral lips with 1 large papilla and 1 amphid each; all of them with cuticular flange overhanging mouth opening (Fig. 2B). Oesophagus divided into anterior short pharynx, cylindrical corpus, slightly narrower isthmus and well-developed posterior bulb with evident valvular apparatus (Fig. 1A and B). Nerve ring, located at middle portion of the oesophagus (Fig. 1A and B). Excretory pore large and slit-like anterior to bulb (Figs 1A, B, 2A and C). Viviparous. Monodelphic and prodelphic; vulva located slightly anterior to anus. Tail conical and sharply pointed in both sexes (Figs 1D, E and 2D). Males with short, equal and slightly sclerotised spicules curved ventrally, with proximal ends expanded, and sharply pointed distally (Fig. 3). Gubernaculum sclerotised, elongated, distal end pointed (Fig. 3). Caudal papillae arranged as follows: 3 pairs precloacal, 3 pairs slightly anterior to fringed cloacal lip plus unpaired papilla situated between them; 5–6 pairs postcloacal: anterior half of tail with 2 pairs of adjacent or in tandem subventral papillae and 1 pair lateral at same level of them; posterior half of tail with 1 lateral pair, 1 subventral pair present or absent and 1 pair subdorsal (Figs 1D, 2D, 4A and D). Posterior cloacal lip with cuticular comb-like fringe (Fig. 4A and D). Vagina well-developed, directed anteriorly, divided into vagina vera, and vagina uterina (Fig. 1B and E). Uteri with morulae embryonated (Fig. 1B). All the measurements obtained from the material collected in this study, re-examined in the type series and in previous studies, are given in Tables 1 and 2.

Figure 1. Line drawings of Schrankiana formosula from Leptodactylus pentadactylus. (A) Male specimen, lateral view; (B) female specimen, lateral view; (C) spicules and gubernaculum, lateral view; (D) caudal region of male, lateral view; (E) caudal region of female, lateral view. Scale bars: A–B = 200 μm; C = 50 μm; D = 100 μm; E = 50 μm.

Figure 2. Scanning electron micrographs of Schrankiana formosula from Leptodactylus pentadactylus. (A) Anterior end, ventral view; (B) cephalic extremity, ventral view; (C) detail of excretory pore, ventral view; (D) posterior end of male, ventral view. Abbreviations: Cl, cloaca; ep, excretory pore; arrows, papillae. Scale bars: A = 100 μm; B–D = 20 μm.

Figure 3. Spicules and gubernaculum of S. formosula. (A) Male from L. pentadactylus, lateral view (present study); (B) male from L. fuscus paratype S. formosula, ventrolateral view (CHIOC 14614c); male from L. pentadactylus paratype S. formosula (= S. inconspicata), ventrolateral view (CHIOC 1954c).

Figure 4. Variability of male caudal papillae of Schrankiana formosula, based on re-examined type series and newly collected material. The caudal papillae numbers are presented as pre cloacal: pairs of papillae slightly anterior to fringed cloacal lip plus unpaired papilla: post cloacal pairs. (A) Male caudal distribution pattern 3:3 + 1:6; (B) male caudal distribution pattern 3:3 + 1:5; (C) male caudal distribution pattern 3:3 + 1:6; (D) male caudal distribution pattern 3:3 + 1:5. Papillae in yellow represent the first post-cloacal pair that varies in position (green). Papillae presented in red are the fifth pair, which may be present or absent in some specimens. Scale bars: A–D = 100 μm.
Table 1. Morphometric analyses of S. formosula from the present study, the type series of Schrankiana formosula, and S. formosula (= S. inconspicata)

All measurements are presented in micrometres, unless otherwise indicated.
a From the anterior end.
Table 2. Morphometric data of Schrankiana formosula from different studies

All measurements are presented in micrometres, unless otherwise indicated.
a From the anterior end.
Metrical characters
The PCA analyses in males showed that the first (PCA1) and the second (PCA2) axis explained 26.58 and 17.16%, respectively. Combined, PCA1 and PCA2 explained 43.74% of the total variance. The first axis reflected the influence of the oesophagus, pharynx, corpus and tail lengths, while the second axis reflected the influence of oesophageal bulb width and the ratio of nerve ring to the body length (Table 3). In females, the first (PCA1) and the second (PCA2) axis explained 36.21 and 14.93%, respectively. Combined, PCA1 and PCA2 explained 51.14% of the total variance (Table 4). The first axis reflected the influence of excretory pore from anterior end, corpus and oesophagus lengths, while the second axis reflected the influence of nerve ring from anterior end, the ratio of tail to body length and morulae embryonated width.
Table 3. Results of principal component analysis of morphometric characters of males Schrankiana formosula (n = 22): coefficients for standardized measurements and percentage of explained variation

a From the anterior end.
Table 4. Results of principal component analysis of morphometric characters of females Schrankiana formosula (20): coefficients for standardized measurements and percentage of explained variation

a From the anterior end.
The comparison between S. formosula (present study), re-examined type series of S. formosula and S. formosula (= S. inconspicata) showed statistical significance in both sexes (males: MANOVA Pillai = 1.434; F = 6.328; P < 0.02; females: MANOVA Pillai = 1.692; F = 11.89; P < 0.00). In the specific morphological comparison, males of S. formosula exhibited significant differences in 4 morphological characters (Table 5). Females of S. formosula exhibited significant differences in all morphological characters, except in the morulae embryonated width (Table 6).
Table 5. Summary of the unidirectional analyses of the morphological characters of male Schrankiana formosula

Bold values denote statistical significance.
Table 6. Summary of the unidirectional analyses of the morphological characters of the female Schrankiana formosula

Bold values denote statistical significance.
Notes on Schrankiana spp. distribution
We found 7 Schrankiana spp. parasitizing 23 anuran hosts from 5 countries: Argentina, Brazil, Costa Rica, Paraguay and Peru. The Leptodactylidae has the highest number of Schrankiana species registered (7), followed by Hylidae (2), Bufonidae (2) and Brachycephalidae (1). All type hosts of Schrankiana spp. are anurans of the genus Leptodactylus Fitzinger, 1826.
Schrankiana formosula is the most common species, reported in 15 host species from 5 countries. Based on the present morphological analyses, we also considered S. inconspicata a synonym of S. formosula species in the distribution map (Table 7) (Fig. 5).
Table 7. Schrankiana species list with host records, host family, and the geographic locality (country).

*Indicate the type-host.

Figure 5. Schrankiana species distribution map and host records.
Molecular analyses and phylogenetic study
We sequenced the 18S rDNA gene from S. formosula and obtained a sequence with 786 pb. The alignment of nuclear gene 18S rDNA upon trimming to the shortest sequence length resulted in 748 pb. Xia's test provided no evidence for substitution saturation in the data matrix. Detailed information on nematode species included in the molecular analyses is provided in Table 8.
Table 8. Nematode species, hosts, localities, GenBank accession numbers and references used in phylogenetic analyses

Pairwise genetic comparison of S. formosula showed low genetic divergence from Raillietnema sp. (0.81%) and Labeonema synodontisi (Vassiliadès, 1973) Koubková, Baruš, Hodová and Šimková, 2008 (2.61%) (see Supplementary 1). The topology of the phylogenetic trees performed on ML and BI revealed similar phylogenies among representatives of the Atractidae and Cosmocercidae (Fig. 6). The sequences of the atractids Grassenema procaviae Petter, 1959 and Rondonia rondoni Travassos, 1920 formed a well-supported monophyletic clade (100 bootstrap and 99 posterior probability).

Figure 6. Maximum likelihood topology based on 18S rDNA using Ichtyobronema hamulatum as outgroup. GenBank accession numbers are indicated next to species names. Numbers beside the nodes represent support value by bootstrap for maximum likelihood analyses and posterior probabilities for Bayesian analyses, respectively (bootstrap scores >70 and posterior probabilities >90).
We observed that sequences of species of Cosmocercidae (99 bootstrap and 100 posterior probability) formed 3 large clades. The first was composed of Cosmocercoides spp.+ Cosmocerca longicauda (Linstow, 1885) and Nemhelix bakeri Morand and Petter, 1986 (91 bootstrap and 99 posterior probability); the second grouped with 2 species of Aplectana Railliet and Henry, 1916 from China + a group consisting of 3 species of Cosmocerca spp. and Aplectana chamaleonis (Baylis, 1929) (62 bootstrap and 81 posterior probability); the latter clade is composed by S. formosula, Raillietnema sp. and L. synodontisi (100 bootstrap and 100 posterior probability) (Fig. 6).
Discussion
Schrankiana inconspicata as a synonym of S. formosula and species differentiation
The specimens studied herein were allocated in Schrankiana based on buccal and pharyngeal structures not elaborate, without specialized cuticularised formations; the morphology of the oesophagus divided into the pharynx, muscular corpus, isthmus well-marked and valved bulb; and spicules short, not much longer than gubernaculum, according to Chabaud (Reference Chabaud, Anderson, Chabaud and Willmott2009) those are the main characters used to identify this genus.
The main morphological characteristics used to distinguish species of Schrankiana include the morphology of the cephalic end; length of the oesophagus; lateral alae extension; vulva location; length of vagina, and male caudal characteristics (papillae, gubernaculum and spicules) (Baker and Vaucher, Reference Baker and Vaucher1988; González and Hamann, Reference González and Hamann2014; Draghi et al., Reference Draghi, Drago and Lunaschi2020). Thus, S. formosula can be easily distinguished from S. chacoensis, S. fuscus, S. freitasi and S. larvata by the number of precloacal papillae (3 pairs in S. formosula vs 4–5 pairs in the other species), and resemble S. inconspicata, S. schranki and S. brasili.
Schrankiana formosula differs from S. schranki by having smaller spicules (42–86 in S. formosula vs 84–101 in S. schranki) and different number of postcloacal papillae (5–6 in S. formosula vs 3 pairs in S. schranki).
Schrankiana formosula differs from S. brasili by the smaller body size in males (1.44–2.30 in S. formosula vs 3.92–4.69 in S. brasili) and females (1.60–2.81 in S. formosula vs 4.62–6.53 in S. brasili); shorter oesophagus (325–560 in S. formosula vs 1000–1200 in S. brasili); absence of the corpus divided into procorpus and metacorpus (present in S. brasili); shorter distance from the vulva to the posterior end (230–390 in S. formosula vs 550–620 in S. brasili); shorter distance of the nerve ring (130–210 in S. formosula vs 340–370 in S. brasili) and excretory pore (280–340 in S. formosula vs 800–900 in S. brasili) from the anterior end.
The specimens resemble S. inconspicata in all measurements, except for the length of the oesophagus in females (325–560 in S. formosula vs 540–710 in S. inconspicata) (Table 1). The original description provided by Freitas (Reference Freitas1959) does not detail the differences between S. formosula and S. inconspicata. However, Baker and Vaucher (Reference Baker and Vaucher1988) distinguished S. inconspicata by an unusual rod-shaped modification of the anterior end of the oesophageal corpus and the presence of an amuscular zone. Additionally, in females, S. inconspicata has lateral alae that end well anterior to the anus (while it is near to the anus in S. formosula) and an elongated uterine vagina (shorter in S. formosula).
We re-examined the type series of S. inconspicata and did not observe the rod-shaped modification, and this structure was not highlighted by Freitas (Reference Freitas1959). We noticed that the short and weakly developed amuscular zone of the oesophagus reported by Baker and Vaucher (Reference Baker and Vaucher1988) is an artefact that might appear during the processing of the specimens. In females, vagina vera length showed variation and overlapped values to those observed for S. formosula and S. inconspicata (see Table 1; Freitas, Reference Freitas1959; Baker and Vaucher, Reference Baker and Vaucher1988). Males exhibit similar spicules (42–86 in S. formosula vs 49–72 in S. inconspicata) and gubernaculum lengths (27–52 in S. formosula vs 29–53 in S. inconspicata). The number and arrangement of caudal papillae of S. inconspicata are the same as described for S. formosula in the present study (Fig. 4) (Table 1).
Therefore, based on all morphological and morphometric similarities observed during re-examination of the type material, we consider S. inconspicata as a synonym of S. formosula, and the specimens that Baker and Vaucher (Reference Baker and Vaucher1988) identified as S. inconspicata might represent a different species.
In the Neotropical region, Schrankiana spp. are widely distributed in Brazil (Fig. 5) and also found in Argentina, Costa Rica, Paraguay and Peru (Table 7). The distribution and host record data suggest that these species have a low host specificity. Previous taxonomic studies suggested that specialist species appeared to be predominant in parasite communities (Combes, Reference Combes and Combes2005; Agosta et al., Reference Agosta, Janz and Brooks2010; Engelstädter and Fortuna, Reference Engelstädter and Fortuna2019). However, parasite host specificity is not inflexible and can vary according to the composition of the host assemblage and the environment, with the parasite communities of anurans often composed of generalist species (Aho, Reference Aho, Esch, Bush and Aho1990; Campião et al., Reference Campião, Morais, Dias, Aguiar, Toledo, Tavares and Silva2014; González et al., Reference González, Valeria and Monika2019; Cardoso et al., Reference Cardoso, Jesus, Silva-Filho, Willkens, Santana, Santos, Santos and Melo2021; Euclydes et al., Reference Euclydes, Dudczak and Campião2021, Reference Euclydes, Torre, Dudczak, Melo and Campião2022). Thus, we suggest that the paradox that parasites do not change in hosts may have led Freitas (Reference Freitas1959) to describe S. formosula and S. inconspicata as different species due to their occurrence in distinct hosts.
Morphological and morphometric variation in Schrankiana formosula
We did not find intraspecific variation in most morphometric data based on the re-examination of the type series, the original description by Freitas (Reference Freitas1959), and the study by Baker and Vaucher (Reference Baker and Vaucher1988), based on material from L. fuscus and L. elenae from Paraguay (see Tables 1 and 2). Of all the measured characters, 6 contributed to this variability in each sex (Tables 3 and 4). Of these, only 4 male characters and 5 female characters were statistically significant between the groups (Tables 5 and 6).
Among the variables that best-discriminated males were characteristics of the oesophagus (pharynx, corpus and oesophagus lengths), and tail length; while in females were characteristics of the oesophagus (corpus and oesophagus lengths), the ratio of the tail to body length, excretory pore and nerve ring from the anterior end (Tables 7 and 8). In fact, the body length strongly influences these characteristics and variations in female growth.
Most of the characteristics proposed by Baker and Vaucher (Reference Baker and Vaucher1988) to differentiate Schrankiana species, such as cephalic end, extent of the lateral alae, location of the vulva, size of the vagina, spicules and gubernaculum presented uniformity in terms of morphology and did not significantly influence on the variability. The bulb width was not statistically significant, and the longest oesophagus was observed in the largest specimens. Thus, the metric difference is considered to reflect the variability of S. formosula.
Intraspecific variations have been reported in other species of the Cosmocercidae such as: Aplectana hylambatis Baylis 1927, Aplectana hamatospicula (Walton, 1940), Cosmocercoides amapari Rebêlo, Santos and Melo 2022, Cosmocercoides variabilis Harwood, 1930 and Cosmocercoides pulcher (Wilkie, 1930) (Vanderburgh and Anderson, Reference Vanderburgh and Anderson1987; Vhora and Bolek, Reference Vhora and Bolek2013; González et al., Reference González, Valeria and Monika2019; Rebêlo et al., Reference Rebêlo, Santos, Tavares-Costa, Dias-Souza, Müller, Jesus, Costa-Campos, Santos and Melo2023). The authors report that the morphological and morphometric variations observed are usually related to generalist species and can be influenced by hosts or localities (González et al., Reference González, Valeria and Monika2019). Thus, as S. formosula is a generalist species, the morphological and morphometric differences are considered intraspecific variations that can be associated with differences in host and environmental attributes. As proposed by González et al. (Reference González, Valeria and Monika2019), our results reinforce the importance of examining the maximum number of specimens possible from different hosts and localities to identify intraspecific variations.
The number and pattern of the caudal papillae in S. formosula are poorly established in the literature. In the original description, Freitas (Reference Freitas1959) reports 3 pairs of sublateral precloacal papillae, 1 papillae on the anterior cloacal lip and 3 postcloacal papillae. Later, Baker and Vaucher (Reference Baker and Vaucher1988) describe 4–5 pairs of subventral precloacal papillae, 3 pairs of papillae on the anterior cloacal lip, with an unpaired papilla situated between them, and 5 pairs of postcloacal papillae.
The newly studied specimens exhibit 3 pairs of precloacal papillae, according to Freitas (Reference Freitas1959). The other papillae were according to the description by Baker and Vaucher (Reference Baker and Vaucher1988): 3 pairs anterior to the cloacal lip, with an unpaired papilla between them, and 5–6 pairs of postcloacal papillae. However, Baker and Vaucher (Reference Baker and Vaucher1988) did not mention the intraspecific morphological variation in the number of postcloacal papillae, and the number of precloacal papillae resembles that of the original description. Additionally, we observed the same number, variation and arrangement of caudal papillae in the type series as described in the present study (Fig. 4).
We also observed that Baker and Vaucher (Reference Baker and Vaucher1988) represented, but did not comment in the manuscript, that the first 2 post-cloacal papillae of S. formosula are slightly diagonal (in tandem) (see Fig. 1 in Baker and Vaucher, Reference Baker and Vaucher1988), while in S. inconspicata those papillae are more in lateral position (see Figure 3 in Baker and Vaucher, Reference Baker and Vaucher1988). However, we observed this variation in both type series (S. inconspicata and S. formosula deposited in CHIOC) and also in newly collected material. After analysing the material that we collected, we concluded that these differences might be artefacts of specimen positioning on the microscope slide.
Phylogeny and systematic position of Schrankiana
Our study presents the first molecular and phylogenetic analyses of the genus Schrankiana. The family Cosmocercidae included the genera Aplectana, Cosmocerca Diesing, 1861, Cosmocercoides Wilkie, 1930, Nemhelix Morand and Petter, 1986, Schrankiana and Labeonema Puylaert, 1970 (99 bootstrap and 100 posterior probability), while the family Atractidae was represented by Grassenema Petter, 1959 and Rondonia Travassos, 1920 (100 bootstrap and 99 posterior probability). These results are according to the classification proposed by Adamson and Baccam (Reference Adamson and Baccam1988) and Gibbons (Reference Gibbons and Gibbons2010), who transferred the genera Labeonema and Schrankiana from the Atractidae to the Cosmocercidae.
Schrankiana formosula formed a well-supported clade with Raillietnema sp. and L. synodontisi (100 bootstrap and 100 posterior probability), and previous molecular studies show a similar pattern (Fig. 6) (Pereira et al., Reference Pereira, Tavares, Paiva, Scholz and Luque2015; Cavalcante et al., Reference Cavalcante, Silva, Santos, Chagas-Moutinho and Santos2016; McElwain et al., Reference McElwain, Warren, Pereira, Ksepka and Bullard2019; Saito et al., Reference Saito, Hayashi, Hayashi, Akita, Une, Kuroki, Shibahara and Takashima2021). Labeonema synodontisi and Raillietnema sp. share morphological similarities with S. formosula, such as cephalic, oesophageal and male caudal structures (size and shape of the spicules, and distribution of caudal papillae), reinforcing the close relationship between them (Fig. 6). The low divergence between Raillietnema sp. and S. formosula can be attributed to those species belonging to anuran hosts in the Neotropical region. In contrast, the higher divergence value observed in L. synodontisi may be associated with the geographical distance between taxa and the different type of host, as this sequence is from a fish from Africa. However, the relationships between them remained unresolved.
Historically, Schrankiana was allocated in the family Atractidae by Chabaud (Reference Chabaud1957), who considered this classification only based on the monodelphic reproductive system of females and aspects of the life cycle such as viviparity and auto-infection. However, only these characteristics could not be decisive in allocating morphologically related genera, such as Raillietnema Travassos, 1927 and Labeonema (Koubková et al., Reference Koubková, Baruš, Hodová and Šimková2008). Later, Adamson and Baccam (Reference Adamson and Baccam1988) and Gibbons (Reference Gibbons and Gibbons2010) moved Schrankiana to Cosmocercidae based on morphological aspects of cephalic structure, arrangement of caudal papillae, oesophagus and excretory pore, hypothesizing that these characters are plesiomorphic, and autoinfection has arisen at least twice in the superfamily Cosmocercoidea. We observed that S. formosula grouped within representatives of Cosmocercidae, forming a sister group of Labeonema and Railletnema, giving an additional piece of evidence that these genera should be placed in Cosmocercidae.
In our phylogeny, the family Atractidae formed a monophyletic group, composed by the species G. procaviae and R. rondoni with high support. Previous studies also showed the monophyly of this group (Cavalcante et al., Reference Cavalcante, Silva, Santos, Chagas-Moutinho and Santos2016; Saito et al., Reference Saito, Hayashi, Hayashi, Akita, Une, Kuroki, Shibahara and Takashima2021; Chen et al., Reference Chen, Ni, Gu, Sinsch and Li2021a, Reference Chen, Gu, Ni and Li2021b). However, Adamson and Baccam (Reference Adamson and Baccam1988) stated that the family Atractidae is characterized by a single synapomorphy: an oesophagus that is distinctly divided at the junction of corpus and isthmus, supported in phylogenetic analyses. The authors asserted that the group requires revision to identify more shared characteristics by the genera.
In our analyses, we did not recover the monophyly of the genus Aplectana, due to the clade A. chamaleonis and Cosmocerca spp., previous studies also received the same grouping (Sinsch et al., Reference Sinsch, Dehling, Sheid and Balczum2020; Chen et al., Reference Chen, Ni, Gu, Sinsch and Li2021a, Reference Chen, Gu, Ni and Li2021b; Harnoster et al., Reference Harnoster, Du Preez and Svitin2022; Ni et al., Reference Ni, Chen, Zhen, Gu and Li2022; Svitin et al., Reference Svitin, Kuzmin, Harnoster, Nel and Du Preez2023). It is important to highlight the limited morphological data available in the literature to confirm A. chamaeleonis identification (see Sinsch et al., Reference Sinsch, Dehling, Sheid and Balczum2020; Chen et al., Reference Chen, Gu, Ni and Li2021b), which raises the hypothesis that the sequence could belong to the genus Cosmocerca.
The sequence of C. longicauda grouped in a clade with N. bakeri as a sister group of Cosmocercoides. Recent molecular phylogenetic works showed that Cosmocercoides might be a monophyletic group (see Saito et al., Reference Saito, Hayashi, Hayashi, Akita, Une, Kuroki, Shibahara and Takashima2021; Harnoster et al., Reference Harnoster, Du Preez and Svitin2022; Ni et al., Reference Ni, Chen, Zhen, Gu and Li2022; Svitin et al., Reference Svitin, Kuzmin, Harnoster, Nel and Du Preez2023; Tuschida et al., Reference Tuschida, Urabe and Nishikawa2023). However, these studies did not consider the sequence of C. longicauda and N. bakeri. Additionally, the recent work by Svitin et al. (Reference Svitin, Kuzmin, Harnoster, Nel and Du Preez2023) suggested that the authors misidentified C. longicauda (OL468616 and OL468682) and that the species should be placed in the genus Cosmocercoides.
We also checked the publication and information provided on GenBank about the sequence of C. longicauda. We could not find the published paper with possible morphological data to corroborate the species identification; based on the research project title, the host might be a snail or a slug. Thus, these data reinforce the potential misidentification of this species. Additionally, Cosmocerca spp. are rarely found in snails, unlike Cosmocercoides spp. (Svitin et al., Reference Svitin, Kuzmin, Harnoster, Nel and Du Preez2023).
Based on this, we reinforce the hypothesis of Svitin et al. (Reference Svitin, Kuzmin, Harnoster, Nel and Du Preez2023) that the C. longicauda sequences (OL468616, OL468682) belong to Cosmocercoides. To support this conclusion, we observed that C. longicauda shows lower genetic divergence in Cosmocercoides (0.81% divergence between the species) than in Cosmocerca (1.91–4.45% divergence between the species). Thus, the phylogenetic position of Cosmocercoides remains uncertain. Furthermore, future molecular studies from Cosmocercidae species, especially from different biogeographical regions, may clarify the phylogenetic position of the genus.
Final remarks
In this study, we redescribed the species S. formosula based on morphology, using light and scanning electron microscopy and molecular analyses. We also re-examined the type material, synonymizing S. inconspicata with S. formosula, one of the most prevalent species of parasites of Leptodactylus in Brazil. Therefore, the geographic distribution of Schrankiana is important for future investigations into the diversity and evolutionary history of the group.
Finally, we performed the first molecular analyses of Schrankiana, elucidating the phylogenetic position of the group, and demonstrated that Schrankiana is a member of the family Cosmocercidae and not Atractidae as previously classified.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S003118202400091X.
Data availability statement
All data from this study are available in the article and supplementary material. Additionaly, the specimens were deposited in the “Museu Paraense Emílio Goeldi” museum.
Acknowledgements
We are grateful to students from the Laboratory of Cellular Biology and Helminthology ‘Profa. Dra. Reinalda Marisa Lanfredi’ (Federal University of Pará, Belém, Brazil) and students from the Laboratory of Herpetology of the Federal University of Amapá. We appreciate the help of MSc. Evelyn Lebrego Cardoso with the support in statistical analyses. We are grateful to the help of Ph.D. Edilene Oliveira da Silva from the Laboratory of Structural Biology (LBE – ICB), at the Federal University of Pará (UFPA), Brazil and MSc. Ronald Ferreira Jesus for their technical support in SEM analysis. We value the help of MSc. Karina Varella Arpon, Ph.D. Beatriz Elise de Andrade Silva and B.Sc. Daniela da Costa Vinhas from the Oswaldo Cruz Institute, Rio de Janeiro, Brazil with molecular analyses. We are grateful to Ph.D. Marcelo Knoff from the helminthological collection of the Oswaldo Cruz Institute (CHIOC, Brazil). We thank the professionals from the Chico Mendes Institute for Biodiversity Conservation (ICMBIO) for granting the necessary collection permits.
Author contributions
A. J. S. Félix wrote the main draft and prepared images. A. N. Santos and C. E. Costa-Campos helped with specimen observations and SEM analyses, reviewing and writing the manuscript. A. Maldonado-Júnior, J. K. S. Neves and G. L. Rebêlo carried out PCR, sequencing and phylogeny. J. N. Santos, L. F. F. Trindade and F. T. V. Melo wrote the manuscript, revised and prepared the line drawings. All authors reviewed the manuscript.
Financial support
This work was supported by Coordination for the Improvement of High Higher Education Personnel, Brazil (CAPES); PROPESP/UFPA; Amazon Foundation for Research and Studies Support (FAPESPA)/CNPq–PRONEM (01/2021 process number 794027/2013); the National Council for Scientific and Technological Development (CNPq) (grant number 431809/2018-6 Universal); and Productivity Scholarship Grant (CNPq) to J. N. Santos (process: 05552/2019-8); F. T. V. Melo. CNPq (process: 314116/2021-4); Carlos Eduardo Costa-Campos (process: 307697/2022-3).
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical standards
All applicable institutional, national and international guidelines for the care and use of animals were followed. Host specimens were collected under permits Institute for the Environment and Renewable Resources – IBAMA/ICMBio (SISBIO: No 53527–4) and Ethics Committee on the Use of Animals of the Federal University of Pará (CEUA/UFPA: No 8341260821).