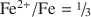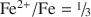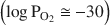Volume 49 - Issue 6 - December 2001
Research Article
Mechanisms and Crystal Chemistry of Oxidation in Annite: Resolving the Hydrogen-Loss and Vacancy Reactions
- D. G. Rancourt, P. H. J. Mercier, D. J. Cherniak, S. Desgreniers, H. Kodama, J.-L. Robert, E. Murad
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 455-491
-
- Article
- Export citation
-
A synthetic octahedral-site-vacancy-free annite sample and its progressive oxidation, induced by heating in air, were studied by powder X-ray diffraction (pXRD), Mössbauer spectroscopy, nuclear reaction analysis (NRA), Raman spectroscopy, X-ray fluorescence (XRF) spectroscopy, gas chromatography (GC), thermogravimetric analysis (TGA), differential thermal analysis (DTA), scanning electron microscopy (SEM), and size-fraction separation methods. For a set heating time and as temperature is increased, the sample first evolves along an annite-oxyannite join, until all H is lost via the oxybiotite reaction (Fe2+ + OH− ⇌ Fe3+ + O2− + H↑). It then evolves along an oxyannite-ferrioxyannite join, where ideal ferrioxyannite, KFe3+8/3□1/3AlSi3O12, is defined as the product resulting from complete oxidation of ideal oxyannite, KFe3+2Fe2+AlSi3O12, via the vacancy mechanism (3 Fe2+ ⇌ 2 Fe3+ + [6]□ + Fe↑). A pillaring collapse transition is observed as a collapse of c near the point where
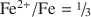 and all OH groups are predicted and observed to be lost. Quantitative analyses of H, using NRA, GC, and Raman spectroscopy, corroborate this interpretation and, in combination with accurate ferric/ferrous ratios from Mössbauer spectroscopy and lattice parameter determinations, allow a clear distinction to be made between vacancy-free and vacancy-bearing annite. The amount of Fe in ancillary Fe oxide phases produced by the vacancy mechanism is measured by Mössbauer spectroscopy to be 11.3(5)% of total Fe, in agreement with both the theoretical prediction of 1/9 = 11.1% and the observed TGA weight gain. The initiation of Fe oxide formation near the point of completion of the oxybiotite reaction (
and all OH groups are predicted and observed to be lost. Quantitative analyses of H, using NRA, GC, and Raman spectroscopy, corroborate this interpretation and, in combination with accurate ferric/ferrous ratios from Mössbauer spectroscopy and lattice parameter determinations, allow a clear distinction to be made between vacancy-free and vacancy-bearing annite. The amount of Fe in ancillary Fe oxide phases produced by the vacancy mechanism is measured by Mössbauer spectroscopy to be 11.3(5)% of total Fe, in agreement with both the theoretical prediction of 1/9 = 11.1% and the observed TGA weight gain. The initiation of Fe oxide formation near the point of completion of the oxybiotite reaction (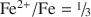 ) is corroborated by pXRD, TGA, Raman spectroscopy, and appearance of an Fe oxide hyperfine field sextet in the Mössbauer spectra. The region of Fe oxide formation is shown to coincide with a region of octahedral site vacancy formation, using a new Mössbauer spectral signature of vacancies that consists of a component at 2.2 mm/s in the [6]Fe3+ quadrupole splitting distribution (QSD). The crystal chemical behaviors of annite-oxyannite and of oxyannite-ferrioxyannite are best contrasted and compared to the behaviors of other layer-silicate series in terms of b vs. [D] (average octahedral cation to O bond length). This also leads to a diagnostic test for the presence of octahedral site vacancies in hydrothermally synthesized annite, based on a graph of b vs. Fe2+/Fe. The implications of the observed sequence of thermal oxidation reactions for the thermodynamic relevance of the oxybiotite and vacancy reactions in hydrothermal syntheses are examined and it is concluded that the oxybiotite reaction is the relevant reaction in the single-phase stability field of annite, at high hydrogen fugacity and using ideal starting cation stoichiometry. The vacancy reaction is only relevant in a multi-phase field, at lower hydrogen fugacity, that includes an Fe oxide equilibrium phase (magnetite) that can effectively compete for Fe, or when using non-ideal starting cation stoichiometries.
) is corroborated by pXRD, TGA, Raman spectroscopy, and appearance of an Fe oxide hyperfine field sextet in the Mössbauer spectra. The region of Fe oxide formation is shown to coincide with a region of octahedral site vacancy formation, using a new Mössbauer spectral signature of vacancies that consists of a component at 2.2 mm/s in the [6]Fe3+ quadrupole splitting distribution (QSD). The crystal chemical behaviors of annite-oxyannite and of oxyannite-ferrioxyannite are best contrasted and compared to the behaviors of other layer-silicate series in terms of b vs. [D] (average octahedral cation to O bond length). This also leads to a diagnostic test for the presence of octahedral site vacancies in hydrothermally synthesized annite, based on a graph of b vs. Fe2+/Fe. The implications of the observed sequence of thermal oxidation reactions for the thermodynamic relevance of the oxybiotite and vacancy reactions in hydrothermal syntheses are examined and it is concluded that the oxybiotite reaction is the relevant reaction in the single-phase stability field of annite, at high hydrogen fugacity and using ideal starting cation stoichiometry. The vacancy reaction is only relevant in a multi-phase field, at lower hydrogen fugacity, that includes an Fe oxide equilibrium phase (magnetite) that can effectively compete for Fe, or when using non-ideal starting cation stoichiometries.
Current Problems with the Nomenclature of Phyllosilicates
- B. B. Zvyagin
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 492-499
-
- Article
- Export citation
-
The nomenclature of phyllosilicates is discussed in relation to structural variations of layers and interlayers. Some discrepancies in the nomenclature which arose due to historical reasons and different viewpoints may be reconciled if the choice of the construction units (layers or their parts) is related to definite sets of structures for which these units are common. Features of idealized models approximating real structures should be considered for classification and derivation of nomenclature sets, whereas priority is given to structural variations before lattice and symmetry characteristics. Layer polymorphs, pseudo-polytypes, polytypes (both simple and complex), and OD structures are distinguished. Particular results obtained for micas as a model example of phyllosilicates in relation to modular structures in general are considered in an Appendix.
The CS/K Exchange in Muscovite Interlayers: An Ab Initio Treatment
- Kevin M. Rosso, James R. Rustad, Eric J. Bylaska
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 500-513
-
- Article
- Export citation
-
Plane-wave pseudopotential total energy calculations have been applied to investigate the structure and energetics of the Cs/K exchange into interlayer sites in muscovite mica. Novel muscovite structures were designed to isolate the effects of 2:1 layer charge, cation size/interlayer site shape, and tetrahedral Al/Si substitutions on the exchange. All atom and cell-parameter optimizations were performed with the intention to mimic the constant pressure, non-isovolumetric exchange conditions thought to be found at frayed-edge sites. Under conditions where the cell parameters are allowed to relax, the overall Cs/K exchange reaction is surprisingly close to isoenergetic. The forward reaction is more strongly favored with increasing layer charge. For the condition of zero layer charge and no interlayer site distortion, the difference in the optimal interlayer spacing for Cs relative to K is very small, indicating a baseline indifference of the muscovite structure to cation size. The presence of 2:1 layer charge or tetrahedral rotations arising from Al/Si substitutions clearly change this outcome. Analysis of the dependence of the interlayer spacing on layer charge shows that while the spacing collapses with increasing layer charge for K as the interlayer cation, the reverse is true for Cs. We attribute the contrasting behavior to inherent differences in the ability of these cations to screen 2:1 layer-layer repulsions. Such effects might be involved during exchange at frayed-edge sites where interlayer spacings are increased. This is known, from experiment, to be very selective for Cs. Overall, the exchange energetics are so low that the Cs/K exchange rate and degree of irreversibility are likely to be dominated by diffusion kinetics.
Quantitative X-Ray Diffraction Analysis of Clay-Bearing Rocks From Random Preparations
- Jan Środoń, Victor A. Drits, Douglas K. McCarty, Jean C. C. Hsieh, Dennis D. Eberl
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 514-528
-
- Article
- Export citation
-
An internal standard X-ray diffraction (XRD) analysis technique permits reproducible and accurate calculation of the mineral contents of rocks, including the major clay mineral families: Fe-rich chlorites + berthierine, Mg-rich chlorites, Fe-rich dioctahedral 2:1 clays and micas, Al-rich dioctahedral 2:1 clays and micas, and kaolinites. A single XRD pattern from an air-dried random specimen is used. Clays are quantified from their 060 reflections which are well resolved and insensitive to structural defects. Zincite is used as the internal standard instead of corundum, because its reflections are more conveniently located and stronger, allowing for a smaller amount of spike (10%). The grinding technique used produces powders free of grains coarser than 20 µm and suitable for obtaining random and rigid specimens.
Errors in accuracy are low, <2 wt. % deviation from actual values for individual minerals, as tested on artificial shale mixtures. No normalization is applied and thus, for natural rocks, the analysis is tested by the departure of the sum of the measured components from 100%. Our approach compares favorably with other quantitative analysis techniques, including a Rietveld-based technique.
Microtextures and the Origin of Muscovite-Kaolinite Intergrowths in Sandstones of the Utrillas Formation, Basque Cantabrian Basin, Spain
- Javier Arostegui, María Jesús Irabien, Fernando Nieto, Javier Sangüesa, María Cruz Zuluaga
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 529-539
-
- Article
- Export citation
-
Muscovite-kaolinite intergrowths found in Albian sandstones of the Basque Cantabrian basin (northern Spain) were studied by optical, scanning and electron microscopy and electron microprobe analysis. Kaolinitization begins at grain edges, forming the characteristic fanned-out textures, and propagates toward the interior along the cleavages of muscovite. Kaolinite and muscovite occur as thick packets, being free of interlayering. Phase boundaries between both minerals show bidimensional crystallographic continuity, and no intermediate phases have been identified. The data obtained suggest that muscovite only supplied a template suitable for the epitactic crystallization of kaolinite, while Al was available in sufficient amounts due to the dissolution of detrital K-feldspar. Very small packets of magnetite or maghemite showing a coherent orientation with the kaolinite crystals have been recognized, and could be responsible for the small Fe contents usually detected in electron microprobe analyses of kaolinite.
Textural relationships between authigenic kaolinite and deformation microstructures in the intergrowths, combined with previous information about burial conditions, show that alteration proceeded during a late stage of the diagenetic history, related to the uplift of the studied materials as a result of the Alpine orogeny.
A Deep-Water Glauconitization Process on the Ivory Coast—Ghana Marginal Ridge (ODP Site 959): Determination of Fe3+-Rich Montmorillonite in Green Grains
- A. Wiewióra, P. Giresse, S. Petit, A. Wilamowski
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 540-558
-
- Article
- Export citation
-
The mineral and chemical composition of green glauconitic grains from ODP Site 959 (2100 m water depth) located on the northern flank of the Ivory Coast—Ghana Marginal Ridge was studied. Recurrent winnowing of a 20 m thick Pleistocene succession resulted in a low accumulation rate and stratigraphic hiatuses. The green clay material typically occurs as fillings in the chambers of pelagic foraminifers. The amount of green clay present in sediments older than 1 Ma is small, and greater in younger material. Mud composed of smectite, kaolinite, traces of mica, calcite and quartz was the precursor material that filled the chambers of the foraminifers. Processes at the water-sediment interface slowly modified this composition. Kaolinite was dissolved; smectite lost Al but gained Fe, K and layer charge. In that matrix, the nanocrystals of neoformed smectite are observed. The infrared (IR) spectra showed OH-stretching and bending vibrations due to groups incorporating Fe3+. The spectra are in agreement with the crystallochemical formulae of Fe3+-rich montmorillonite as determined by point-by-point analyses on the neoformed crystallites and on the surrounding matrix. The layer charge in this Fe3+-rich montmorillonite is almost wholly octahedral as shown in crystallochemical formulae and documented independently by a new IR method. The tetrahedral charge appeared when the Fe content increased by > 1.2 Fe per formula unit. With the maturation process, the increased role of the closed layers is observed, with the color of grains becoming greener. We have documented for the first time glauconitization proceeding at a depth of 2100 m at a temperature near 3°C. The most important factors of the process are: accumulation of terrigenous clayey material in the foraminiferal chambers, Fe supply from a nearby continent, and a lengthy residence at the water-sediment interface in the zone of the winnowing and low sediment accumulation rate.
Authigenic Phyllosilicates in the Middle Triassic Kremikovtsi Sedimentary Exhalative Siderite Iron Formation, Western Balkan, Bulgaria
- Zhelyazko Damyanov, Margarita Vassileva
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 559-585
-
- Article
- Export citation
-
Two distinct assemblages of authigenic phyllosilicates were distinguished in the Kremikovtsi sedimentary exhalative (SEDEX) siderite iron formation (SIF) and noted as important tracers of two styles of mineralization characteristic of this type of ore deposit. Hydrothermal-sedimentary layer silicates are represented by rare occurrences of relict microcrystalline Mg-rich berthierine with a relatively low degree of structural ordering, associated closely with framboidal pyrite as an intergranular matrix cementing sparry siderite grains; the larger silicates are also represented by the diagenetic transformation product of berthierine, chamosite. Berthierine precipitated under anoxic conditions during advanced early diagenesis after chert deposition. Hydrothermal-epigenetic phyllosilicates (berthierine, chamosite, illite-smectite (I-S), and kaolinite) from the barite-sulfide assemblage are characterized by: crystalline and undeformed habits; relatively larger particle size, low-temperature polytypes, low to no mixed layering, and a high degree of crystallinity; absence of impurities and dominant monomineralic aggregates; affiliation to initial open spaces and deposition mainly as vug fillings and linings. They formed under pronounced control by the vuggy porosity of the siderite host caused by the invasion of acid (pH = 3–5), hot (200–230°C) hydrothermal fluids probably at the stage of burial diagenesis of the SIF under relatively stable reducing conditions fluctuating near the sulfide/sulfate stability boundary
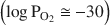 . The greater Al concentration in hydrothermal solution than in seawater determines the affiliation of phyllosilicates in the Phanerozoic SEDEX SIFs to aluminous species (berthierine, chamosite) unlike low to non aluminous ones (greenalite, stilpnomelane) in the Precambrian IFs. The berthierine compositions, expressed by the Mg/Fe vs. Al/Si ratios, are a sensitive indicator of the geological conditions under which they formed (marine, non-marine, hydrothermal ore and pre-ore), thus allowing the genetic discrimination of this mineral from various localities.
. The greater Al concentration in hydrothermal solution than in seawater determines the affiliation of phyllosilicates in the Phanerozoic SEDEX SIFs to aluminous species (berthierine, chamosite) unlike low to non aluminous ones (greenalite, stilpnomelane) in the Precambrian IFs. The berthierine compositions, expressed by the Mg/Fe vs. Al/Si ratios, are a sensitive indicator of the geological conditions under which they formed (marine, non-marine, hydrothermal ore and pre-ore), thus allowing the genetic discrimination of this mineral from various localities.
Mineralogy of Dickite and Nacrite From Northern Taiwan
- Pei Yuan Chen, Ming Kuang Wang, Deng Shiu Yang
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 586-595
-
- Article
- Export citation
-
Nacrite and dickite are found in two localities in northern Taiwan. One, containing nacrite and dickite, is associated with a gold-enargite deposit as a vug-filling clay in the Chinkushih (CKS) mine district near the northern coast. The other is the occurrence of dickite in the interstices of a Miocene coarse-grained quartzose sandstone in the Nanshihchiao (NSC) area, near Taipei city. X-ray diffraction (XRD), differential thermal analysis (DTA), scanning electron microscope (SEM), and petrographic examinations were used to characterize the mineralogical features. Nacrite most often takes the unusual form of rhombic platelets, and dickite is commonly elongated in habit with the shapes possibly related to their origin. Based on geological evidence, we believe that both nacrite and dickite are of hydrothermal origin. In the CKS area, the formation of nacrite and dickite is related to the hypogene gold-enargite mineralization. In contrast, the transformation of dickite in the NSC area is due to the influence of the raised temperatures of sandstone formation, resulting from volcanic activity during the Kungkuan stage after the deposition of the sandstone.
Kaolinite to Halloysite-7 Å Transformation in the Kaolin Deposit of São Vicente De Pereira, Portugal
- Iuliu Bobos, Joelle Duplay, João Rocha, Celso Gomes
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 596-607
-
- Article
- Export citation
-
The transformation of kaolinite to halloysite-7 Å was identified in the kaolin deposit of São Vicente de Pereira (SVP), using X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR) and transmission electron microscopy (TEM). Both the 02, 1̄ and 13̄, 13 reflections show changes in the XRD patterns along the kaolinite to halloysite-7 Å transition, and the FTIR spectra show changes corresponding to both OH− and Si-O-stretching bands and Al-O-Si-bending vibrations. The interlayer water content in the kaolinite structure increases during transition. The two-layer periodicity of well-ordered kaolinite and rolling up of kaolinite plates are observed using high-resolution transmission electron microscopy (HRTEM). Long and short tubes exhibit halloysite-7 Å. No structural Fe was found in the kaolinite samples. Analytical electron microscopy (AEM) indicates no substitution of Al3+ for Si4+. The Si/Al ratio shows values of ∼1 for the kaolinite and rolled kaolinite plates. The 27Al magic angle spinning neutron magnetic resonance (MAS-NMR) spectra display a resonance centered at ∼1 ppm, assigned to six-coordinated aluminum. The transformation of kaolinite to halloysite-7 Å is controlled by surface reaction.
Announcement
Forthcoming Papers
-
- Published online by Cambridge University Press:
- 28 February 2024, p. 620
-
- Article
-
- You have access Access
- Export citation