Key points
• Drought and heat stress significantly reduced the number of seeds that germinated and delayed germination in sorghum genotypes as independent factors not in combination.
• Genotypes were classified into six distinct groups according to their overall performance with check varieties that were assessed i.e. ‘SV4 and Macia’ classified with moderate performers.
• Genotypes NPGRC1593, NPGRC1782, IS30015, NPGRC1476 and IS13837 showed superior tolerance to heat stress.
• Genotypes NPGRC1593, NPGRC1782, NPGRC1156, IS24426, IS13837 and NPGRC1476 showed superior tolerance to drought stress.
Introduction
Three salient stages of early crop growth that contribute to the attainment of optimum yield are germination, emergence and seedling establishment. Thus, farmers who target high yields invest a lot of time and resources in land preparation, securing and planting quality seeds to ensure these critical stages are uninterrupted. Irrefutably, seed quality is one of the most essential inputs that determines crop productivity. Seed viability, optimum edaphic and environmental factors are the conspicuous prerequisites for germination, a weighty and incipient process that spans from seed imbibition to radicle emergence in early crop growth and establishment (Khaeim et al., Reference Khaeim, Kende, Jolánkai, Kovács, Gyuricza and Tarnawa2022). Insufficient soil moisture and excessive temperatures in seeding zones at planting are notable hindrance factors in crop production in semi-arid tropics (SATs) (Bayu et al., Reference Bayu, Rethman, Hammes, Pieterse, Grimbeek and Van der Linde2005; Kapoor et al., Reference Kapoor, Savita, Marco, Sharma, Mathusany and Sharma2020). Accordingly, these areas are characterized by soils with poor water holding capacity, high evaporative demands due to excessive atmospheric temperatures and erratic rainfall. However, changes and variability in climate characterized by excessive atmospheric temperatures, reduced, uneven spatial and temporal distributions of rainfall coupled with porous soils renders concern on germination in rainfed crop production systems (Geilbyo et al., Reference Geilbyo, Toth, Farkas, Horel, Kasi and Bakacsi2018). It is more pressing for a small grain crop like sorghum, which is climate smart and most suitable to aforementioned conditions of proven drought and heat stress tolerance (Phiri et al., Reference Phiri, Nhliziyo, Madzivire, Sithole and Nyathi2020; Nciizah et al., Reference Nciizah, Nciizah, Mubekaphi, Nciizah, Oguge, Ayal, Adeleke and da Silva2021).
Critical and incipient stages like germination have been targeted by plant breeders, physiologists and botanists especially in screening germplasm for tolerance to abiotic stressors like heat, drought and salinity (Krasenky and Jonak, Reference Krasenky and Jonak2012; Tsago et al., Reference Tsago, Andargie and Takele2014). However, most studies have evaluated the effects of drought and heat stress on sorghum seed germination separately, leading to inconclusive results (Abreha et al., Reference Abreha, Enyew, Carlsson, Vetukuri, Feyissa, Motlhaodi, Ng'uni and Geleta2022). Time and resources are saved in such studies when large populations of germplasm are evaluated for tolerance under combined conditions like drought and heat stress, which often coincide in any case (Fahad et al., Reference Fahad, Bajwa, Nazir, Anjum and Farooq2017). Soil moisture deficit and salinity have always been singled out as the most severe inhibitors of seed germination (Ndlovu et al., Reference Ndlovu, Staden and Maphosa2021). Imbibition marks the beginning of the germination process, though it is a physical process driven by the water potential gradient between dry seed and soil. Ideal soil moisture content is estimated at 35 to 45% in germinating seeds (Queiroz et al., Reference Queiroz, Oliveira, Steiner, Zuffo, Zoz, Vendruscolo, Silva, Mello, Cabral and Menis2019), and optimum germination temperature range for sorghum is 20–28°C (Adamcik et al., Reference Adamcik, Tomaseck, Pulkrabek, Pazderu and Dvorak2016). Low soil osmotic potential of 0 to 0.8 MPa has been proven to reduce final germination percentage (FGP) and germination index (GI) by inhibiting seed imbibition (Shahriari et al., Reference Shahriari, Ali Adam, Ghizan, Addul and Ali Adam2014; Queiroz et al., Reference Queiroz, Oliveira, Steiner, Zuffo, Zoz, Vendruscolo, Silva, Mello, Cabral and Menis2019), while mean germination time (MGT) was observed to be prolonged by lower osmotic potential (Ahmad et al., Reference Ahmad, Ahmad, Ashraf, Ashraf and Waraich2009; Abreha et al., Reference Abreha, Enyew, Carlsson, Vetukuri, Feyissa, Motlhaodi, Ng'uni and Geleta2022). Delayed germination increases the risk of losing seed to desiccation, predators like birds, rats and other pests as well as pathogens.
When combined, heat stress and drought stress are very detrimental to germination with far reaching implications as they often coincide during early and late plantings (Fahad et al., Reference Fahad, Bajwa, Nazir, Anjum and Farooq2017). In SATs, soil temperatures are likely to be higher which results in high evaporation of moisture especially in the uppermost soil layer (in the range of 0–5 cm) which is the planting depth for small seeded cereals like sorghum (Bayu et al., Reference Bayu, Rethman, Hammes, Pieterse, Grimbeek and Van der Linde2005). Despite the increasing incidences of combined drought and heat stress which has been proven to be very detrimental to all stages of most crops, the two factors are often studied separately (Bheemanahalli et al., Reference Bheemanahalli, Ramamoorthy, Poudel, Samiappan, Wijewardane and Reddy2022). Furthermore, most seminal work has focused on flowering and grain filling stages. This study is one of the few that seeks to investigate the two factors combined at germination of sorghum landraces, breeding lines and check varieties selected from a large pool of world and national gene banks and have never been tested. The study will provide new insights on the response of the selected high potential tropical sorghum genotypes to drought and heat stress either combined or occurring independently at germination. This offers a quick and cheap screening technique of the germplasm that will inform breeders and plant physiologists in crop improvement programs.
Field evaluation of emergence tends to be complicated by complex interactions of soil properties and other field conditions, thus the widespread adoption of an in vitro technique using polyethylene glycol (PEG) induced osmotic stress (Bobade et al., Reference Bobade, Amarshettivar, Rathod, Ghorade and Kayande2019). The technique has proven to be effective, low cost, easy to handle and less laborious for screening large pools of genetic material for abiotic stress tolerance (Tsago et al., Reference Tsago, Andargie and Takele2014), to aid crop improvement programs. This study was aimed at assessing the response of 50 sorghum accessions to drought, heat and combined heat and drought stresses at germination.
Materials and methods
Experimental site and material
A laboratory experiment was conducted at Lupane State University, Department of Crop and Soil Sciences laboratory. Fifty sorghum genotypes acquired from the Genetic Resources and Biotechnology Institute of Zimbabwe and International Crop Research Institute of the Semi-Arid Tropics (ICRISAT) Matopo were selected from a pool of 300 genotypes based on plant morpho-physiological characteristics that included stem height, days to maturity, susceptibility to lodging and 100 grain weight from a baseline study conducted from the previous season (2019/20). Two commercial sorghum varieties namely ‘Macia’ and ‘SV4’ were included as check varieties.
Treatments and management of the experiment
Seeds of fifty selected sorghum accessions (online Supplementary Table S1) were germinated under controlled osmotic and temperature conditions in a laboratory as follows: osmotic stress only (20% (m/v) PEG 6000 solution induced osmotic potential of −0.85 MPa according to a procedure by Foti et al. (Reference Foti, Cosentino, Patane and Agosta2002), heat stress only (45°C day and 25°C night incubation temperatures), combined heat and osmotic stresses (a combination of the two aforementioned treatments) and no stress (optimum temperatures of 25°C day and 22°C night incubation and deionized water) as a control.
Fifty seeds of homogenous size and age for each genotype were surface sterilized by immersion in 1.5% sodium hypochlorite solution for 5 min and then thoroughly rinsed five times in distilled water. Thereafter, they were placed in sterilized petri dishes lined with double Whatman No. 2 filter papers moistened with 7 ml of either distilled water for no-stress treatments or 20% (w/v) PEG 6000 solution of −0.85 MPa osmotic potential for osmotic stressed treatments. The petri dishes were sealed with parafilm to reduce water loss.
The experiment was laid in a split plot following a two factorial completely randomized design with three replicates. The main factor for splitting was the temperature at two levels, i.e. optimum (25°C) and heat stress (45°C). The two sub-factors were the 50 genotypes and two levels of osmotic conditions, i.e. no osmotic stress (PEG solution) and low osmotic potential (distilled water). The resultant was 200 treatment combinations i.e. 50 genotypes × 2 osmotic levels × 2 temperature levels were each replicated three times to give 600 experimental units. The experiment was kept in an incubator for 7 d. The experiment was repeated in order to increase the sample size and the two sets were done in tandem.
Data collection
Count data on the germinated seeds was collected every 24 h for 5 d. Seeds that had between 1 and 2 mm protruding coleorhizae were considered germinated. The following four germination parameters were derived from the daily seed count:
i) Final germination percentage (FGP)
(1)$${\rm FGP} = \displaystyle{{Number\;of\;normally\;germinated\;seeds} \over {total\;number\;of\;sown\;seeds}} \times 100\% $$
ii) Mean germination time (day)
(2)$${\rm MGT} = \displaystyle{{T_i \times N_i} \over N}, \;$$
where Ni is the number of newly germinated seeds at time Ti
-
Germination index
(3)$${\rm GI} = \displaystyle{{Gt} \over {Tt}}$$
where Gt is the number of seeds germinated on day t, and Tt is the number of days
a) Coefficient of variation of germination time (% seed/day)
(4)$${\rm CVt} = \left({\displaystyle{{S_{\rm t}} \over {\;\;t}}} \right)100, \;$$
where S t is the standard deviation of germination time and t is mean germination time
Data analysis
Response of sorghum genotypes to the four most important germination parameters i.e. FGP, MGT, GI and CVt to heat stress, drought stress and their interactions was modelled using generalized linear models with either Poisson or negative binomial log-link functions in R studio packages. Principal component analysis (PCA) of pooled means of the assessed germination parameters for 50 sorghum genotypes was conducted using the multivariate analysis PCA function in Minitab version 14. Furthermore, the hierarchical agglomerative cluster analysis was performed using dissimilarity coefficients and unweighted pair group method arithmetic average (UPGMA) of clustering for tree construction using DARwin 6.0.21 software. Principal coordinate analysis (PCoA) was also performed using DARwin to illustrate a two-dimension map of the distribution of genotypes based on their performance in the treatments.
Results
Multivariate analysis of the influence of genotypes on germination parameters under heat and drought stress
The generalized linear model results revealed significant differential responses within the sorghum genotypes under in vitro induced environments, i.e. temperature and drought stress for the four assessed germination parameters. GI is the only parameter that was significantly affected by the interaction between the genotypes and temperature stress (G × T). GI together with FGP were significantly affected by the interaction of the genotype and drought stress (G × D). No significant influence was observed from interaction of the three factors that were under investigation, i.e. genotype, drought and temperature stress across all six parameters that were measured. However, significant main effects of the three factors were noted on FGP and GI. Significant influence of temperature stress was noted across all six parameters. Hierarchical cluster analysis based on unweighted pair group method analysis (UPGMA) (Fig. 3) and Principal Coordinate analysis (Fig. 4) plots confirmed that the number of germinating seeds and their speed of germination under temperature and drought stress was dependent on genotype. The two complementary plots classified the genotypes into six distinct groups, according to their overall performance in all six germination parameters that were assessed in this study. The PCA indicated that the first and the second components accounted for 62.5 and 17.3% respectively, of the variation that existed among the germination parameters (Table 1).
Table 1. Principal component analysis in 50 diverse sorghum genotypes for five germination parameters
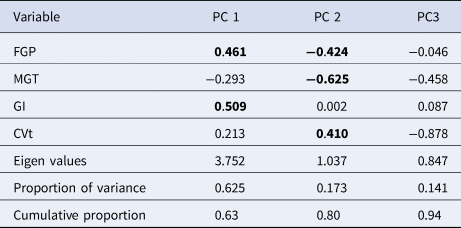
Principal component analysis of the germination parameters
Diversity in tolerance to drought and heat stress during germination in 50 sorghum genotypes that were assessed in this study was expressed in some of the four germination parameters that were measured. The first two significant components expressed a cumulative variation of 80% in the assessed germination variables (Table 1) as shown by their Eigen values > 1. Most of the variation attributed to the first component was contributed by GI which had the highest positive factor loading value of 0.51. FGP also contributed positively in the first component. The significant relationship to diversity in germination of the genotypes in the second component was due to the highest negative loading value of −0.63 observed in MGT and FGP with a loading value of −0.424. Some positive contribution by the coefficient variation of germination time loading values of 0.41 was observed in the second component.
Evaluation of heat and drought stress using germination index
Following the significant influence of the main effects of drought stress (D), heat stress (T) and their interactions with genotypes (G × D) and (G × T) in the GLM analysis, the GI means were selected and used to screen the 50 genotypes for their tolerance to drought and heat stress separately. Seeds germinated under non-drought stressed conditions (distilled water) had a significantly higher GI of 34.99 compared to 18.62 for their counterparts germinated in drought stressed conditions (lower osmotic potential) simulated using 20% PEG solution. Supra-optimal temperatures of 45°C also suppressed germination more than optimum temperatures of 25°C. A statistically lower mean GI of 24.51 was observed in seeds germinated at 45°C compared to 29.51 for seeds germinated at 25°C. Both stressors independently reduced the number of seeds that germinated and prolonged time to germination. The differential response of the genotypes to these stresses is presented in Figs. 1 and 2 respectively.
More than 40% of the accessions under the heat stressed treatment had a GI mean higher than the overall GI mean of 20 and showed no significant differences to about 60% of the accessions germinated under optimal temperature conditions (Fig. 1). Landraces IS13837 and NPGRC1156 performed exceptionally well under both supra-optimal and optimum temperature conditions. Five genotypes (Fig. 1) had GI means under optimum temperatures which were not statistically different from their counterparts germinated under supra-optimal conditions. ‘Macia’ and ‘SV4’ check varieties were amongst the poorest performing varieties under supra-optimal conditions with mean germination indices of 17.22 and 20.11 respectively. However, the check varieties were amongst the best performers under optimum temperatures.

Figure 1. Mean germination indices of sorghum genotypes germinated under in vitro induced temperature stress (supra-optimal 45°C) and optimum (25°C) environments. The vertical bars at the apex of each bar denote the standard error of means and bars marked with different letters show significant differences in GI means. Genotypes have been arranged in descending order of their observed GI means under heat stressed conditions.
Five genotypes exhibited a superior performance under drought stress with GI means above 25 under drought stress which was not statistically different from the top performing genotypes germinated under optimum osmotic conditions. Three of the five poorest performing genotypes had GI means less than 10 under low osmotic potential (drought stress) (Fig. 2). The two check varieties ‘Macia’ and ‘SV4’, usually grown in drought prone regions, performed moderately well, yielding GI means of 21.15 and 20.24 respectively. Though the combined effect of the two stressors was not statistically significant, genotypes NPGRC1782, IS24426, NPGRC1593, NPGRC1156 and NPGRC1476 showed superiority were the effects of heat and drought stress when assessed independently.

Figure 2. Mean germination indices (GI) of sorghum genotypes germinated under in vitro induced drought stress and no stress conditions. The vertical bars at the apex of each bar denote the standard error of means and bars marked with different letters show significant differences in GI means. Genotypes have been arranged in descending order of their GI means under drought stressed conditions.
Cluster analysis of 50 sorghum genotypes using four germination traits
Agglomerative clustering of the 50 sorghum genotypes based on overall performance in all the germination traits in the four drought and temperature treatment combinations grouped the genotypes into six distinct clusters. Cluster I and III were the largest groups with 15 and 13 genotypes respectively. While Clusters V and VI had the least number of genotypes of 2 and 1 respectively (Fig. 3). The performance of genotypes in each group is detailed in Table 2.

Figure 3. Dendrogram by hierarchical agglomerate cluster analysis of the 50 assessed sorghum genotypes.
Table 2. Overall mean performance of clusters for different germination parameters in 50 sorghum genotypes
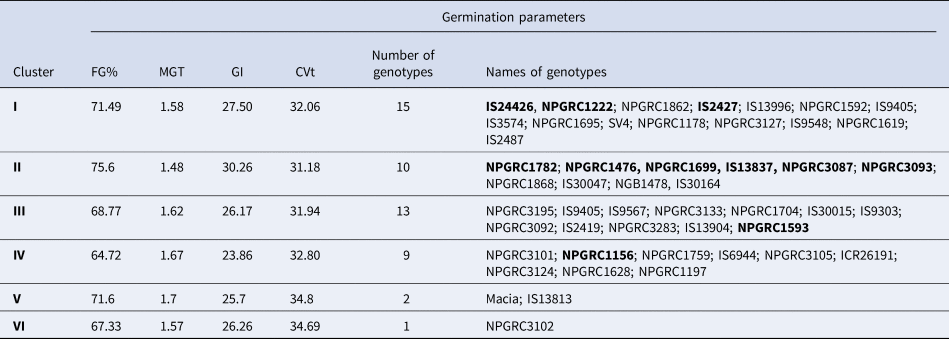
Genotypes in bold were the most tolerant to both osmotic and temperature stress during germination.
The clustering was also confirmed by the principal coordinate analysis (PcoA) plot (Fig. 4) which produces a map for visualization of the genotypes based on their dis/similarity. The scattered genotypes in all the four quarters of the PcoA plot show a high degree of genetic variation among the assessed genotypes, Furthermore the two-dimensional map gives a clear indication on the proximity of the genotypes in each cluster based on the distance computed using the variations in performance in PCA. The closer the genotypes the more similar they are for example IS9405 and NPGRC1868 in Cluster 2, IS2419 and IS9303 in cluster III.

Figure 4. Principal coordinate analysis (PCoA) for 50 genotypes based on their overall performance in the 5 assessed germination parameters.
Comparison of the clusters under selected key germination parameters
Comparative analysis of performance of the six clusters under the four selected germination parameters indicated that cluster II had the highest number of genotypes that exhibited tolerance to independent effects of drought and heat stress followed by cluster I (Table 2). Genotypes in cluster II had the highest mean FGP of 75.6%, GI of 30.26 and the lowest MGT of 1.48 d. The least tolerant genotypes to heat and drought stress, i.e. IS26191, IS9405 and IS9303 as presented previously (Figs. 1 and 2) were found in clusters III and IV. Cluster IV had the lowest GI of 23.86 and as expected the highest MGT of 1.67 d. The check varieties fell into different groups with ‘SV4’ in cluster I and ‘Macia’ in cluster V.
Discussion
Differential response of genotypes in seed physiological tests like germination under limiting factors like drought and heat stress has been established in an array of crops including sorghum (Shahriari et al., Reference Shahriari, Ali Adam, Ghizan, Addul and Ali Adam2014; Moyo et al., Reference Moyo, Ndlovu, Moyo, Kudita and Maphosa2015; Nyoni et al., Reference Nyoni, Ndlovu and Maphosa2020). Diversity in germination related parameters under stress conditions was confirmed by the cluster analysis results which grouped the genotypes into six distinct groups (Table 2). Most of the tolerant genotypes were landraces from semi-arid areas of Zimbabwe like Chiredzi and KwaZulu-Natal Province of South Africa. This is suggestive of coevolution of these landraces in these hot, dry environments which allows them to adapt to such unfavourable conditions. The two check varieties ‘Macia’ and ‘SV4’ were grouped with genotypes that performed moderately under drought stress but showed lack of tolerance to heat stress, indicating that the major target of their improvement was probably drought stress more than heat stress. Recent studies have also confirmed the existence of genotypic differences in tolerance of drought and heat stress in sorghum (Masetto et al., Reference Masetto, Oba and Jesus2017; Govindaraj et al., Reference Govindaraj, Djanaguiraman, Djalovic, Shailani, Rawat, Singla-Pareek and Prasad2021). The results of this study followed suit in exhibiting some variations in response of genotypes to heat and drought stress, though the significant dual effect of two stressors on genotypes was not observed in this study as hypothesized. This means the two stressors influenced in vitro germination of the assessed genotypes independently, though in the field they often occur together and are likely to interact significantly to delay and reduce germination. Occurrence of different genotypes with tolerance to both and each of the factors separately could be indicative of independent genetic mechanisms controlling both.
The complex interactions of genotypes, temperature and drought stress during germination is not conclusively understood (Abreha et al., Reference Abreha, Enyew, Carlsson, Vetukuri, Feyissa, Motlhaodi, Ng'uni and Geleta2022). Evidence points to elevated osmotic pressure in hot soils due to depletion of moisture, more so in saline conditions (Dehnavi et al., Reference Dehnavi, Zahedi, Agnieszka, Cardenas and Piernik2020). Maize is reported to be more sensitive to both heat and drought stress than sorghum, a study by Khaeim et al. (Reference Khaeim, Kende, Jolánkai, Kovács, Gyuricza and Tarnawa2022), observed that maize seeds failed to germinate at 40°C. In a separate study by Mingli et al. (Reference Mingli, Meng, Kaichang and Na2015) of two maize varieties on the effects of drought stress of almost the same intensity with the one used in the present study revealed that germination percentage was reduced by a margin of between 28 to 50%.
GI was selected to represent the germination parameters in screening for tolerance in genotypes due to its highest contribution to the variation in the first component observed in the PCA. GI, a function of FGP and MGT, is a very accurate and comprehensive measure which takes into account the extent and speed of germination (Kader, Reference Kader2005). A higher GI indicates a higher FGP and a lower MGT, i.e. more seeds germinating within the shortest period of time. The energy and time taken for germinating seeds to osmotically adjust under high osmotic pressure renders the differential response to speed and extent of germination signified by the variations in the GI (Shahriari et al., Reference Shahriari, Ali Adam, Ghizan, Addul and Ali Adam2014). Generally, in this study, drought stress suppressed germination more than heat stress as shown by a difference of 10 units between the highest values recorded for mean GI for the two factors. Accordingly, genotypes such as NPGRC1782, IS24426, NPGRC1593, NPGRC1156 and NPGRC1476 that exhibited high GI under the main effects of the two factors have potential and further studies on the genetic basis of their tolerance mechanisms could inform plant breeding efforts that intend to exploit the material as parents.
Supra-optimal temperatures above 25°C have been proven to inhibit germination even when soil moisture conditions are favourable (Patane et al., Reference Patane, Saita and Tubeileh2012). The antagonistic effects of heat stress on germinating seeds are reportedly amplified by imbibition at high temperatures (Wen, Reference Wen2015). This is attributed to the inhibition of enzyme dependent reactions and even total halt due to changes at supra-optimal temperatures. Though not significant to over 90% of the assessed genotypes, three genotypes appeared in the top of the list in both main heat and drought stress treatments namely; NPGRC 1782, NPGRC1593 and IS13837.
Conclusions and recommendations
Drought and heat stress independently affected germination in sorghum genotypes. Their dual effect was not statistically significant in the sorghum genotypes that were assessed in this study. Supra-optimal temperatures of 45°C differentially delayed germination and suppressed the extent of germination in sorghum genotypes. A similar and even more detrimental effect was observed with in vitro induced drought stress. Ten genotypes showed the highest tolerance to heat stress and two genotypes showed the highest tolerance to drought stress benchmarked using the overall mean GI. A huge diversity in drought and heat stress tolerance was exhibited in the assessed germplasm. The genotypes were grouped into six classes based on their performance in terms of extent and time taken to germinate. No exceptions where observed in the performance of the assessed genotypes but genotypes NPGRC1782, NPGRC1593 and IS13837 were among the top three performing genotypes, and IS26191, NPGRC9405 and IS12391 were among the least performing genotypes under the main effects of the heat and drought stress. The results can be used in selecting the tolerant genotypes in either heat or drought stress during germination for crop improvement purposes depending on the trait of interest. Further screening of the genotypes that showed potential in this study is recommended under field conditions.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S1479262124000212.
Acknowledgements
Germplasm used in this research was obtained from the Benefit-sharing Fund project PR-316-Uganda that has been funded by the Food and Agriculture Organization's (FAO) International Treaty on Plant Genetic Resources for Food and Agriculture (ITPGRFA), https://www.fao.org/plant-treaty/areas-of-work/benefit-sharing-fund/projects-funded/bsf-details/en/c/1198836/?iso3=UGA.
Author contributions
Elton Ndlovu: Conceptualization; acquisition of research materials, conceptualization; data collection, data analysis, writing – original draft. Mcebisi Maphosa: Acquisition of research materials, data analysis, writing – original draft. Johannes Van Staden: Writing – original draft.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing interests
None.








