Volume 77 - Issue 7 - October 2013
Research Article
Crystal chemical and structural investigation of levyne-Na
- P. Ballirano, G. Cametti
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2887-2899
-
- Article
- Export citation
-
A sample of levyne-Na from Dunseverick, Antrim, Northern Ireland, UK, has been characterized from the crystal chemical and structural point of view. Its mean crystal chemical formula, calculated on the basis of 54(Si+Al), is Na7.72Ca4.62K1.31Sr0.08Mg0.02(Al19.55Si34.45O107.50)·50.5H2O. Levyne-Na is associated with erionite-Na whose chemical formula is Na6.08Ca0.8 5K2.55Mg0.16(Al10.41Si25.59O72.13)·30.6H2O, calculated on the basis of 36(Si+Al). Their compositions are consistent with those of levyne/erionite inter/overgrowth reported in the literature. The structural analysis, carried out by the Rietveld method on laboratory parallel-beam X-ray powder diffraction data, is in good agreement with the chemical composition representing a significant improvement with respect to the available data so far. Extra-Framework (EF) cations are allocated at four sites, Ca1, Na3, K4 and Na5 with partial occupancy. Water molecules are distributed among five sites. Possible coexistence schemes of cations, forced by the avoidance of short contacts are proposed and validated by bond valence analysis.
The crystal structure of chalcoalumite: mechanisms of Jahn-Teller-driven distortion in [6]Cu2+-containing oxysalts
- F. C. Hawthorne, M. A. Cooper
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2901-2912
-
- Article
- Export citation
-
The crystal structure of chalcoalumite, ideally Cu2+Al4(SO4)(OH)12(H2O)3, monoclinic, P21/n, Z = 4:a 10.228(3), b 8.929(3), c 17.098(6) Å, β 95.800(11)°, V 1553.6(1.5) Å3, has been refined to R1 = 3.08% for 4,022 unique observed (4σ) reflections collected on a Bruker D8 three-circle diffractometer equipped with a rotating-anode generator, multilayer optics and an APEX-II CCD detector. In the structure of chalcoalumite, there is one S site, tetrahedrally coordinated by four O anions, with <S–O> = 1.472 Å. There are four Al sites with site-scattering values in accord with occupancy by Al and <Al–O> distances of 1.898–1.919 Å. There is one Cu site occupied by Cu2+ and coordinated by six anions in the [4 + 2] arrangement typical for octahedrally coordinated Cu2+. The short <Cu–O> distance of 2.086 Å is in accord with the low degree of bond-length distortion of the Cu octahedron. There are 19 anion sites: 4 sites are occupied by O atoms that are bonded to the S cation, 12 sites are occupied by (OH) groups that bond to all octahedrally coordinated cations, and 3 sites are occupied by (H2O) groups that are held in the structure solely by hydrogen bonding. The structure of chalcoalumite consists of interrupted sheets of edge-sharing Al and Cu octahedra of the form [Cu2+Al4(OH)12]2+ that intercalate layers of (SO4) tetrahedra and (H2O) groups. Chalcoalumite is a member of the nickelalumite group.
Cu2+ϕ6(ϕ = O2–, (OH)–, (H2O)0) octahedra show a wide range of bond-length distortion away from the holosymmetric arrangement, driven by spontaneous symmetry-breaking of the degenerate electronic ground-state in holosymmetric octahedral coordination. Here, we examine the structural mechanisms that allow large octahedron distortions of this type. There are two mechanisms: (1) coupling of (usually parallel) octahedron distortions to a vibrational phonon, inducing a (often ferroelastic) phase transition in M2+-Cu2+ solid-solutions; (2) cooperative orientational disorder, where bond topology (polyhedron linkage) allows large differences in bond lengths within polyhedra to accord with the valence-sum rule of bond-valence theory.
Margarite, corundum, gahnite and zincohögbomite in a blackwall, Raleigh Terrane, Eastern Piedmont Province, USA
- B. E. Owens, H. E. Belkin, J. M. Zerolis
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2913-2929
-
- Article
- Export citation
-
We report an unusual occurrence of margarite, corundum, gahnite and zincohögbomite from the Raleigh terrane in the Piedmont Province of Virginia. The assemblage occurs in a chlorite-rich blackwall associated with a small metamorphosed ultramafic rock body. The blackwall is dominated by chlorite, but also contains distinctive clusters (1–4 mm across) that typically consist of ragged Zn-rich spinel grains surrounded by masses of randomly oriented margarite. Also spatially associated with spinel are smaller grains of corundum and högbomite. In most cases, spinel and högbomite are sufficiently enriched in Zn to be called gahnite and zincohögbomite, respectively. Some högbomite grains are distinctly banded in back-scattered electron images, primarily reflecting variations in Zn-content. Textures suggest that högbomite formed at the expense of spinel (although locally the reverse relationship holds), and högbomite compositions mimic those of spinel. Margarite appears to be a later phase, and textures imply formation via reactions with spinel, corundum and, possibly, chlorite. This occurrence of Zn-rich spinel and högbomite is clearly related to the bulk composition of the blackwall rock, which contains >2500 ppm Zn. The origin of this Zn-rich composition is unclear, but could be due to a small amount of sphalerite or zincian-staurolite in the protolith.
Oxyplumboroméite, Pb2Sb2O7, a new mineral species of the pyrochlore supergroup from Harstigen mine, Värmland, Sweden
- U. Hålenius, F. Bosi
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2931-2939
-
- Article
- Export citation
-
Oxyplumboroméite, Pb2Sb2O7, is a new mineral of the roméite group of the pyrochlore supergroup (IMA 2013-042). It is found together with calcite and leucophoenicite in fissure fillings in tephroite skarn at the Harstigen mine, Värmland, Sweden. The mineral occurs as yellow to brownish yellow rounded grains or imperfect octahedra. Oxyplumboroméite has a Mohs hardness of ∼5, a calculated density of 6.732 g/cm3 and is isotropic with a calculated refractive index of 2.061. Oxyplumboroméite is cubic, space group Fd
 m, with the unit-cell parameters a = 10.3783(6) Å, V = 1117.84(11) Å3 and Z = 8. The strongest five X-ray powder-diffraction lines [d in Å(I)(hkl)] are: 2.9915(100)(222), 2.5928(32)(400), 1.8332(48)(440), 1.5638(38)(622) and 1.1900(12)(662). The crystal structure of oxyplumboroméite was refined to an R1 index of 3.02% using 160 unique reflections collected with MoKα radiation. Electron microprobe analyses in combination with crystal-structure refinement, infrared, Mössbauer and electronic absorption spectroscopy resulted in the empirical formula A(Pb0.92Ca0.87Mn0.09Sr0.01Na0.05)Σ1.93B(Sb1.73Fe3+0.27)Σ2.00X+Y[O6.64(OH)0.03]Σ6.67. Oxyplumboroméite is the Pb analogue of oxycalcioroméite, ideally Ca2Sb2O7.
m, with the unit-cell parameters a = 10.3783(6) Å, V = 1117.84(11) Å3 and Z = 8. The strongest five X-ray powder-diffraction lines [d in Å(I)(hkl)] are: 2.9915(100)(222), 2.5928(32)(400), 1.8332(48)(440), 1.5638(38)(622) and 1.1900(12)(662). The crystal structure of oxyplumboroméite was refined to an R1 index of 3.02% using 160 unique reflections collected with MoKα radiation. Electron microprobe analyses in combination with crystal-structure refinement, infrared, Mössbauer and electronic absorption spectroscopy resulted in the empirical formula A(Pb0.92Ca0.87Mn0.09Sr0.01Na0.05)Σ1.93B(Sb1.73Fe3+0.27)Σ2.00X+Y[O6.64(OH)0.03]Σ6.67. Oxyplumboroméite is the Pb analogue of oxycalcioroméite, ideally Ca2Sb2O7.
Crystallographic and spectroscopic characterization of a natural Zn-rich spinel approaching the endmember gahnite (ZnAl2O4) composition
- V. D’Ippolito, G. B. Andreozzi, F. Bosi, U. Hålenius, L. Mantovani, D. Bersani, R. A. Fregola
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2941-2953
-
- Article
- Export citation
-
The crystal chemistry of a natural, gem-quality, blue-grey Zn-rich spinel crystal from Jemaa, Kaduna State, Nigeria, was studied using electron microprobe, single-crystal X-ray diffraction, optical absorption and Raman spectroscopies. The composition of the crystal approaches the gahnite endmember (ZnAl2O4), ∼94 mol.%, with the remainder being dominated by a hercynite component (FeAl2O4). The unit-cell dimension is 8.0850(3) Å and the tetrahedral and octahedral bond distances are determined as T–O 1.9485(6) Å and M–O 1.9137(3) Å. Crystal chemical analysis resulted in the empirical structural formula T(Zn0.94Fe2+0.03Al0.03)M (Al1.96Fe2+0.03Fe3+0.01)O4, which shows Zn and Al almost fully ordered in the tetrahedrally and octahedrally coordinated T and M sites, respectively. Raman spectra obtained using the excitation of the blue 473.1 nm line of a Nd:YAG laser display three of the five Raman-active modes predicted for the general oxide spinel group of minerals. These are the Eg mode at 420.6 cm–1 and the T2g modes at 510 cm–1 and 661 cm–1, due to vibrations in the AlO6 octahedra. Optical absorption spectra recorded in the UV/VIS-NIR-MIR range 2000 29000 cm–1 show a dominant absorption band at ∼5000 cm–1 which is caused by spin-allowed electronic d–d transitions in Fe2+ located at the T sites. The blue-grey hue exhibited by the sample is mainly due to spin-forbidden electronic transitions in TFe2+ and to MFe2+ ↔MFe3+ intervalence charge transfer, and the poor saturation of the colour is due to the small concentration of Fe2+ and Fe3+.
Veblenite, K2☐2Na(Fe2+5Fe3+4Mn2+7☐)Nb3Ti(Si2O7)2(Si8O22)2O6(OH)10(H2O)3, a new mineral from Seal Lake, Newfoundland and Labrador: mineral description, crystal structure, and a new veblenite Si8O22 ribbon
- F. Cámara, E. Sokolova, F. C. Hawthorne, R. Rowe, J. D. Grice, K. T. Tait
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2955-2974
-
- Article
- Export citation
-
Veblenite, ideally K2☐2Na(Fe2+5Fe3+4Mn2+7☐)Nb3Ti(Si2O7)2(Si8O22)2O6(OH)10(H2O)3, is a new mineral with no natural or synthetic analogues. The mineral occurs at Ten Mile Lake, Seal Lake area, Newfoundland and Labrador (Canada), in a band of paragneiss consisting chiefly of albite and arfvedsonite. Veblenite occurs as red brown single laths and fibres included in feldspar. Associated minerals are niobophyllite, albite, arfvedsonite, aegirine-augite, barylite, eudidymite, neptunite, Mn-rich pectolite, pyrochlore, sphalerite and galena. Veblenite has perfect cleavage on {001} and splintery fracture. Its calculated density is 3.046 g cm–3. Veblenite is biaxial negative with α 1.676(2), β 1.688(2), γ 1.692(2) (λ 590 nm), 2Vmeas = 65(1)°, 2Vcalc = 59.6°, with no discernible dispersion. It is pleochroic in the following pattern: X = black, Y = black, Z = orange-brown. The mineral is red-brown with a vitreous, translucent lustre and very pale brown streak. It does not fluoresce under short and long-wave UV-light. Veblenite is triclicnic, space group P
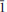 , a 5.3761(3), b 27.5062(11), c 18.6972(9) Å, α 140.301(3), β 93.033(3), γ 95.664(3)°, V = 1720.96(14) Å3. The strongest lines in the X-ray powder diffraction pattern [d(Å)(I)(hkl)] are: 16.894(100)(010), 18.204(23)(0
, a 5.3761(3), b 27.5062(11), c 18.6972(9) Å, α 140.301(3), β 93.033(3), γ 95.664(3)°, V = 1720.96(14) Å3. The strongest lines in the X-ray powder diffraction pattern [d(Å)(I)(hkl)] are: 16.894(100)(010), 18.204(23)(0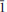 1), 4.271(9)(
1), 4.271(9)(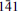 , 040, 120), 11.661(8)(001), 2.721(3)(
, 040, 120), 11.661(8)(001), 2.721(3)(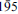 ), 4.404(3)(
), 4.404(3)( ,
, 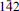 ), 4.056(3)(031, 1
), 4.056(3)(031, 1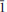 2;
2;  ,
, 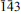 ), 3.891(2)(003).
), 3.891(2)(003).The chemical composition of veblenite from a combination of electron microprobe analysis and structural determination for H2O and the Fe2+/Fe3+ ratio is Nb2O5 11.69, TiO2 2.26, SiO2 35.71, Al2O3 0.60, Fe2O3 10.40, FeO 11.58, MnO 12.84, ZnO 0.36, MgO 0.08, BaO 1.31, SrO 0.09, CaO 1.49, Cs2O 0.30, K2O 1.78, Na2O 0.68, H2O 4.39, F 0.22, O = F –0.09, sum 95.69 wt.%. The empirical formula [based on 20 (Al+Si) p. f. u. is (K0.53Ba0.28Sr0.03☐0.16)Σ1(K0.72Cs0.07☐1.21)Σ2(Na0.72Ca0.17☐1.11)Σ2(Fe2+5.32Fe3+4.13Mn2+5.97Ca0.70Zn0.15Mg0.07☐0.66)Σ17(Nb2.90Ti0.93Fe3+0.17)Σ4(Si19.61Al0.39)Σ20O77.01H16.08F0.38. The simplified formula is (K, Ba, ☐)3(☐, Na)2(Fe2+, Fe3+, Mn2+)17(Nb,Ti)4(Si2O7)2(Si8O22)2O6(OH)10(H2O)3. The infrared spectrum of the mineral contains the following bands (cm–1): 453, 531, 550, 654 and 958, with shoulders at 1070, 1031 and 908. A broad absorption was observed between ~3610 and 3300 with a maximum at ~3525. The crystal structure was solved by direct methods and refined to an R1 index of 9.1%. In veblenite, the main structural unit is an HOH layer, which consists of the octahedral (O) and two heteropolyhedral (H) sheets. The H sheet is composed of Si2O7 groups, veblenite Si8O22 ribbons and Nb-dominant D octahedra. This is the first occurrence of an eight-membered Si8O22 ribbon in a mineral crystal structure. In the O sheet, (Fe2+, Fe3+, Mn2+) octahedra share common edges to form a modulated O sheet parallel to (001). HOH layers connect via common vertices of D octahedra and cations at the interstitial A(1,2) and B sites. In the intermediate space between two adjacent HOH layers, the A(1) site is occupied mainly by K; the A(2) site is partly occupied by K and H2O groups, the B site is partly occupied by Na. The crystal structure of veblenite is related to several HOH structures: jinshanjiangite, niobophyllite (astrophyllite group) and nafertisite. The mineral is named in honour of David R. Veblen in recognition of his outstanding contributions to the fields of mineralogy and crystallography.
Meisserite, Na5(UO2)(SO4)3(SO3OH)(H2O), a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah, USA
- J. Plášil, A. R. Kampf, A. V. Kasatkin, J. Marty, R. Škoda, S. Silva, J. Čejka
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2975-2988
-
- Article
- Export citation
-
Meisserite (IMA2013-039), Na5(UO2)(SO4)3(SO3OH)(H2O), is a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah (USA). It is named in honour of the prominent Swiss mineralogist Nicolas Meisser. The new mineral was found in a sandstone matrix and is associated with chalcanthite, copiapite, ferrinatrite, gypsum, johannite and another new Na-bearing uranyl sulfate, belakovskiite (IMA2013-075). Meisserite is a secondary mineral formed by the post-mining weathering of uraninite. The mineral is triclinic, P
 , a = 5.32317(10), b = 11.5105(2), c = 13.5562(10) Å, α = 102.864(7)°, β = 97.414(7)°, γ = 91.461(6)°, V = 801.74(6) Å3, and Z = 2. Crystals are prisms elongated on [100], up to 0.3 mm long, exhibiting the forms {010} and {001}. Meisserite is pale green to yellowish green, translucent to transparent and has a very pale yellow streak. It is brittle, with fair cleavage on {100} and {001}, and uneven fracture. The Mohs hardness is estimated at 2. Meisserite is somewhat hygroscopic and easily soluble in water. The calculated density based on the empirical formula is 3.208 g/cm3. Meisserite exhibits bright yellow green fluorescence under both long- and shortwave UV radiation. The mineral is optically biaxial (–), with α = 1.514(1), β = 1.546(1), γ = 1.557(1) (measured in white light). The measured 2V is 60(2)° and the calculated 2V is 60°. Dispersion is r > v, perceptible, and the optical orientation is X ≈ a, Z ≈ c*. The mineral is pleochroic, with X (colourless) < Y (pale yellow) ≈ Z (pale greenish yellow). The empirical formula of meisserite (based on 19 O a.p.f.u.) is Na5.05(U0.94O2)(SO4)3[SO2.69(OH)1.31](H2O). The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and also weaker O–H stretching vibrations. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl)Irel]: 13.15 (001) 81, 6.33 (0
, a = 5.32317(10), b = 11.5105(2), c = 13.5562(10) Å, α = 102.864(7)°, β = 97.414(7)°, γ = 91.461(6)°, V = 801.74(6) Å3, and Z = 2. Crystals are prisms elongated on [100], up to 0.3 mm long, exhibiting the forms {010} and {001}. Meisserite is pale green to yellowish green, translucent to transparent and has a very pale yellow streak. It is brittle, with fair cleavage on {100} and {001}, and uneven fracture. The Mohs hardness is estimated at 2. Meisserite is somewhat hygroscopic and easily soluble in water. The calculated density based on the empirical formula is 3.208 g/cm3. Meisserite exhibits bright yellow green fluorescence under both long- and shortwave UV radiation. The mineral is optically biaxial (–), with α = 1.514(1), β = 1.546(1), γ = 1.557(1) (measured in white light). The measured 2V is 60(2)° and the calculated 2V is 60°. Dispersion is r > v, perceptible, and the optical orientation is X ≈ a, Z ≈ c*. The mineral is pleochroic, with X (colourless) < Y (pale yellow) ≈ Z (pale greenish yellow). The empirical formula of meisserite (based on 19 O a.p.f.u.) is Na5.05(U0.94O2)(SO4)3[SO2.69(OH)1.31](H2O). The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and also weaker O–H stretching vibrations. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl)Irel]: 13.15 (001) 81, 6.33 (0 2) 62, 5.64 (0
2) 62, 5.64 (0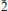 1,020) 52, 5.24 (100,012,
1,020) 52, 5.24 (100,012, 01) 100, 4.67 (101) 68, 3.849 (
01) 100, 4.67 (101) 68, 3.849 (
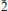 1,102,022) 48, 3.614 (0
1,102,022) 48, 3.614 (0 2,
2,
 3) 41, and 3.293 (
3) 41, and 3.293 ( 13,004) 43. The crystal structure of meisserite (R1 = 0.018 for 3306 reflections with Iobs > 3σI) is topologically unique among known structures of uranyl minerals and inorganic compounds. It contains uranyl pentagonal bipyramids linked by SO4 groups to form chains. Na+ cations bond to O atoms in the chains and to an SO3OH group and an H2>O group between the chains, thereby forming a heteropolyhedral framework.
13,004) 43. The crystal structure of meisserite (R1 = 0.018 for 3306 reflections with Iobs > 3σI) is topologically unique among known structures of uranyl minerals and inorganic compounds. It contains uranyl pentagonal bipyramids linked by SO4 groups to form chains. Na+ cations bond to O atoms in the chains and to an SO3OH group and an H2>O group between the chains, thereby forming a heteropolyhedral framework.
Fluorcalciomicrolite, (Ca,Na,☐)2Ta2O6F, a new microlite-group mineral from Volta Grande pegmatite, Nazareno, Minas Gerais, Brazil
- M. B. Andrade, D. Atencio, A. I. C. Persiano, J. Ellena
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2989-2996
-
- Article
- Export citation
-
Fluorcalciomicrolite, (Ca, Na, ☐)2Ta2O6F, is a new microlite-group, pyrochlore supergroup mineral approved by the CNMNC (IMA 2012-036). It occurs as an accessory mineral in the Volta Grande pegmatite, Nazareno, Minas Gerais, Brazil. Associated minerals include: microcline, albite, quartz, muscovite, spodumene, “lepidolite”, cassiterite, tantalite-(Mn), monazite-(Ce), fluorite, “apatite”, beryl, “garnet” , epidote, magnetite, gahnite, zircon, “tourmaline” , bityite, hydrokenomicrolite, and other microlite-group minerals under study. Fluorcalciomicrolite occurs as euhedral, untwinned, octahedral crystals 0.1–1.5 mm in size, occasionally modified by rhombododecahedral faces. The crystals are colourless and translucent; the streak is white, and the lustre is adamantine to resinous. It does not fluoresce under ultraviolet light. Mohs' hardness is 4½–5, tenacity is brittle. Cleavage is not observed; fracture is conchoidal. The calculated density is 6.160 g/cm3. The mineral is isotropic, ncalc. = 1.992. The Raman spectrum is dominated by bands of B–X octahedral bond stretching and X–B–X bending modes. The chemical composition (n = 6) is (by wavelength dispersive spectroscopy, H2O calculated to obtain charge balance, wt.%): Na2O 4.68, CaO 11.24, MnO 0.01, SrO 0.04, BaO 0.02, SnO20.63, UO20.02, Nb2O53.47, Ta2O576.02, F 2.80, H2O 0.48, O=F–1.18, total 98.23. The empirical formula, based on 2 cations at the B site, is (Ca1.07Na0.81☐0.12)Σ2.00(Ta1.84Nb0.14Sn0.02)Σ2.00[O5.93(OH)0.07]6.00[F0.79(OH)0.21]. The strongest eight X-ray powder-diffraction lines [d in Å (I)(hkl)] are: 5.997(59)(111), 3.138(83)(311), 3.005(100)(222), 2.602(29)(400), 2.004(23)(511), 1.841(23)(440), 1.589(25)(533), and 1.504(24)(444). The crystal structure refinement (R1 = 0.0132) gave the following data: cubic, Fd
 m, a = 10.4191(6) Å, V = 1131.07(11) Å3, Z = 8.
m, a = 10.4191(6) Å, V = 1131.07(11) Å3, Z = 8.
CNMNC Newsletter 17
New minerals and nomenclature modifications approved in 2013
- P. A. Williams, F. Hatert, M. Pasero, S. J. Mills
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2997-3005
-
- Article
- Export citation
Research Article
The alunite supergroup under high pressure: the case of natrojarosite, NaFe3(SO4)2(OH)6
- F. Nestola, S. J. Mills, B. Periotto, L. Scandolo
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3007-3017
-
- Article
- Export citation
-
A single crystal of natrojarosite, NaFe3(SO4)2(OH)6, was investigated by single-crystal X-ray diffraction at high-pressure conditions (up to 8.8 GPa) using a diamond-anvil cell. The unit-cell parameters were determined at 11 different pressures and no indications of a phase transition were found up to the maximum pressure reached. The volume and axial moduli were fitted to a third-order Birch–Murnaghan equation-of-state which gave the following values: V0 = 769.6(2) Å3, KT0 = 50.6(9) GPa, K' = 9.9(4); a = 7.3172(6) Å, KT0 = 104(2), K' = 7.6(9); c = 16.5965(20) Å, KT0 = 24.6(4) and K' = 7.1(2). The crystal structure of natrojarosite was refined at seven different pressures up to 8.779(11) GPa [a = 7.3170(4), c = 16.5955(5) Å and V = 769.46(9) Å3 in R
 m at 0.00010(1) GPa and a = 7.1594(8), c = 15.6003(17) Å and V = 692.49(8) Å3 at 8.779(11) GPa]. The structural analysis shows that the 12-fold Na polyhedron accommodates most of the deformation by a large volume decrease (14%) and strong polyhedral distortion (63%). Our results indicate that natrojarosite has the most compressible structure of the supergroup studied so far, and has a very strong axial anisotropy.
m at 0.00010(1) GPa and a = 7.1594(8), c = 15.6003(17) Å and V = 692.49(8) Å3 at 8.779(11) GPa]. The structural analysis shows that the 12-fold Na polyhedron accommodates most of the deformation by a large volume decrease (14%) and strong polyhedral distortion (63%). Our results indicate that natrojarosite has the most compressible structure of the supergroup studied so far, and has a very strong axial anisotropy.
Deveroite-(Ce): a new REE-oxalate from Mount Cervandone, Devero Valley, Western-Central Alps, Italy
- A. Guastoni, F. Nestola, P. Gentile, F. Zorzi, M. Alvaro, A. Lanza, L. Peruzzo, M. Schiazza, N. M. Casati
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3019-3026
-
- Article
- Export citation
-
Deveroite-(Ce), ideally Ce2(C2O4)3·10H2O, is a new mineral (IMA 2013-003) found in the alpine fissures of Mount Cervandone, overlooking the Devero Valley, Piedmont, Italy. It occurs as sprays of colourless elongated tabular, acicular prisms only on cervandonite-(Ce). It has a white streak, a vitreous lustre, is not fluorescent and has a hardness of 2–2.5 (Mohs' scale). The tenacity is brittle and the crystals have a perfect cleavage along {010}. The calculated density is 2.352 g/cm3. Deveroite-(Ce) is biaxial (–) with 2V of ∼77°, is not pleochroic and the extinction angle (β ∧ c) is ∼27°. No twinning was observed. Electron microprobe analyses gave the following chemical formula: (Ce1.01Nd0.33La0.32Pr0.11Y0.11Sm0.01Pb0.04U0.03Th0.01Ca0.04)2.01(C2O4)2.99·9.99H2O. Although synchrotron radiation was not used to solve the structure of deveroite-(Ce) the extremely small size of the sample (13 μm × 3 μm × 1 μm) did not allow us to obtain reliable structural data. However, it was possible to determine the space group (monoclinic, P21/c) and the unit-cell parameters, which are: a = 11.240(8) Å, b = 9.635(11) Å, c = 10.339(12) Å, β = 114.41(10)°, V = 1019.6 Å3. The strongest lines in the powder diffraction pattern [d in Å (I)(hkl)] are: 10.266(100)(100); 4.816(35.26)(21
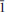 ); 3.415(27.83)(300); 5.125(24.70)(200); and 4.988(22.98)(111). Deveroite-(Ce) is named in recognition of Devero valley and Devero Natural Park.
); 3.415(27.83)(300); 5.125(24.70)(200); and 4.988(22.98)(111). Deveroite-(Ce) is named in recognition of Devero valley and Devero Natural Park.
Oxycalcioroméite, Ca2Sb2O6O, from Buca della Vena mine, Apuan Alps, Tuscany, Italy: a new member of the pyrochlore supergroup
- C. Biagioni, P. Orlandi, F. Nestola, S. Bianchin
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3027-3037
-
- Article
- Export citation
-
The new mineral species oxycalcioroméite, Ca2Sb5+2O6O, has been discovered at the Buca della Vena mine, Stazzema, Apuan Alps, Tuscany, Italy. It occurs as euhedral octahedra, up to 0.1 mm in size, embedded in dolostone lenses in the baryte + pyrite + iron oxides ore. Associated minerals are calcite, cinnabar, derbylite, dolomite, hematite, 'mica', pyrite, sphalerite and 'tourmaline'. Oxycalcioroméite is reddish-brown in colour and transparent. It is isotropic, with ncalc = 1.950.
Electron microprobe analysis gave (wt.%; n = 6) Sb2O5 63.73, TiO2 3.53, SnO2 0.28, Sb2O3 10.93, V2O3 0.68, Al2O3 0.28, PbO 0.68, FeO 5.52, MnO 0.13, CaO 13.68, Na2O 0.83, F 1.20, O = F – 0.51, total 100.96. No H2O, above the detection limit, was indicated by either infrared or micro-Raman spectroscopies. The empirical formula, based on 2 cations at the B site, is (Ca1.073Fe2+0.338Sb3+0.330Na0.118Pb0.013Mn0.008)Σ=1.880(Sb5+1.734Ti0.194V0.040Al0.024Sn0.008)Σ=2.000(O6.682F0.278)Σ6.960. The crystal structure study gives a cubic unit cell, space group Fd
 m, with a 10.3042(7) Å, V 1094.06(13) Å3, Z = 8. The five strongest X-ray powder diffraction lines are [d(Å)I(visually estimated)(hkl)]: 3.105(m)(311); 2.977(s)(222); 2.576(m)(400); 1.824(ms)(440); and 1.556(ms)(622). The crystal structure of oxycalcioroméite has been solved by X-ray single-crystal study on the basis of 114 observed reflections, with a final R1 = 0.0114. It agrees with the general features of the members of the pyrochlore supergroup.
m, with a 10.3042(7) Å, V 1094.06(13) Å3, Z = 8. The five strongest X-ray powder diffraction lines are [d(Å)I(visually estimated)(hkl)]: 3.105(m)(311); 2.977(s)(222); 2.576(m)(400); 1.824(ms)(440); and 1.556(ms)(622). The crystal structure of oxycalcioroméite has been solved by X-ray single-crystal study on the basis of 114 observed reflections, with a final R1 = 0.0114. It agrees with the general features of the members of the pyrochlore supergroup.
Barikaite, Pb10Ag3(Sb8As11)Σ19S40, a new member of the sartorite homologous series
- D. Topa, E. Makovicky, H. Tajedin, H. Putz, G. Zagler
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3039-3046
-
- Article
- Export citation
-
Barikaite, ideally Pb10Ag3(Sb8As11)Σ19S40, is a new mineral species from the Barika Au-Ag deposit, Azarbaijan Province, western Iran. It was formed in fractures developed in silica bands situated in massive banded pyrite and baryte ores. These fractures house veinlets that contain a number of Ag-As-Sb-Pb-rich sulfosalts, tetrahedrite-tennantite, realgar, pyrite and electrum. Barikaite appears as inclusions in guettardite. The mineral is opaque, greyish black with a metallic lustre; it is brittle without any discernible cleavage. In reflected light barikaite is greyish white, pleochroism is distinct, white to dark grey. Internal reflections are absent. In crossed polars, anisotropism is distinct with rotation tints in shades of grey. The reflectance data (%, in air) are: 37.0, 39.3 at 470 nm, 34.1, 36.9 at 546 nm, 33.1, 36.2 at 589 nm and 31.3, 34.1 at 650 nm. The Mohs hardness is 3–3½, microhardness VHN50 exhibits the range 192 – 212, with a mean value of 200 kg mm–2. The average results of five electron-microprobe analyses in a grain are (in wt.%): Pb 35.77(33), Ag 5.8(1), Tl 0.15(08), Sb 18.33(09), As 15.64(16), S 24.00(15), total 99.69(10) wt.%, corresponding to Pb9.31Ag2.90Tl0.04(Sb8.12As11.26)Σ19.36S40.37 (on the basis of 32Me + 40S = 72 a.p.f.u.). The simplified formula, Pb10Ag3(Sb8As11)Σ19S40, is in accordance with the results of a crystal-structure analysis, and requires Pb 37.89, Ag 5.91, Sb 17.79, As 15.05 and S 23.42 (wt.%). The variation of chemical composition is minor, the empirical formula ranging from Pb10.39Ag2.32Tl0.02Sb7.52As11.27S40.49 to Pb9.24Ag2.93Tl0.04Sb8.13As11.35S40.31. Barikaite has monoclinic symmetry, space group P21/n and unit-cell parameters a 8.5325(7) Å, b 8.0749(7) Å, c 24.828(2) Å, and b 99.077(6)o, Z = 1. Calculated density for the empirical formula is 5.34 (g cm–3). The strongest eight lines in the (calculated) powder-diffraction pattern [d in Å(I)(hkl)] are: 3.835(63)(022), 3.646(100)(016), 3.441(60)(212), 3.408(62)(
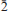 14), 2.972(66)(
14), 2.972(66)(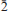 16), 2.769(91)(222), 2.752(78)(
16), 2.769(91)(222), 2.752(78)(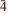 24) and 2.133(54)(402). Barikaite is the N = 4 member of the sartorite homologous series with a near-equal role of As and Sb, which have an ordered distribution pattern in the structure. It is a close homeotype of rathite and more distantly related to dufrénoysite (both distinct, pure arsenian N = 4 members) and it completes the spectrum of Sb-rich members of the sartorite homologous series. The new mineral and its name have been approved by the IMA-CNMNC (IMA 2012-055).
24) and 2.133(54)(402). Barikaite is the N = 4 member of the sartorite homologous series with a near-equal role of As and Sb, which have an ordered distribution pattern in the structure. It is a close homeotype of rathite and more distantly related to dufrénoysite (both distinct, pure arsenian N = 4 members) and it completes the spectrum of Sb-rich members of the sartorite homologous series. The new mineral and its name have been approved by the IMA-CNMNC (IMA 2012-055).
Leverettite from the Torrecillas mine, Iquique Provence, Chile: the Co-analogue of herbertsmithite
- A. R. Kampf, M. J. Sciberras, P. A. Williams, M. Dini, A. A. Molina Donoso
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3047-3054
-
- Article
- Export citation
-
The new mineral leverettite (IMA 2013-011), ideally Cu3CoCl2(OH)6, was found at the Torrecillas mine, Salar Grande, Iquique Province, Chile, where it occurs as a supergene alteration phase in association with akaganéite, anhydrite, chalcophanite, goethite, halite, manganite, pyrite, quartz and todorokite. Crystals of leverettite are steep rhombohedra to 1 mm with {101} prominent and modified by {001}, sometimes forming V-shaped twins by reflection on {10
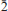 }. The crystals can also form finger-like, parallel stacked growths along the c axis. The new mineral is medium to deep green in colour and has a light green streak. Crystals are transparent with a vitreous lustre. Mohs hardness is ∼3 and the crystals have a brittle tenacity, a perfect cleavage on {101} and a conchoidal fracture. The measured density is 3.64(2) g cm–3 and calculated density based on the empirical formula is 3.709 g cm–3. Optically, leverettite is uniaxial (–) with ω and ε > 1.8 and exhibits pleochroism with O (bluish green) > E (slightly yellowish green). The empirical formula, determined from electron-microprobe analyses is Cu3(Co0.43Cu0.40Mn0.17Ni0.07Mg0.01)Σ1.08Cl1.87O6.13H6. Leverettite is trigonal (hexagonal), space group R
}. The crystals can also form finger-like, parallel stacked growths along the c axis. The new mineral is medium to deep green in colour and has a light green streak. Crystals are transparent with a vitreous lustre. Mohs hardness is ∼3 and the crystals have a brittle tenacity, a perfect cleavage on {101} and a conchoidal fracture. The measured density is 3.64(2) g cm–3 and calculated density based on the empirical formula is 3.709 g cm–3. Optically, leverettite is uniaxial (–) with ω and ε > 1.8 and exhibits pleochroism with O (bluish green) > E (slightly yellowish green). The empirical formula, determined from electron-microprobe analyses is Cu3(Co0.43Cu0.40Mn0.17Ni0.07Mg0.01)Σ1.08Cl1.87O6.13H6. Leverettite is trigonal (hexagonal), space group R m, unit-cell parameters a = 6.8436(6) and c = 14.064(1) Å, V = 570.42(8) Å3, Z = 3. The eight strongest X-ray powder diffraction lines are [dobs Å (I)(hkl)]: 5.469(90)(101), 4.701(18)(003), 2.905(22)(021), 2.766(100)(113), 2.269(66)(024), 1.822(26)(033), 1.711(33)(220), 1.383(23)(128). The structure, refined to R1 = 0.023 for 183 Fo > 4σF reflections, shows leverettite to be isostructural with herbertsmithite and gillardite.
m, unit-cell parameters a = 6.8436(6) and c = 14.064(1) Å, V = 570.42(8) Å3, Z = 3. The eight strongest X-ray powder diffraction lines are [dobs Å (I)(hkl)]: 5.469(90)(101), 4.701(18)(003), 2.905(22)(021), 2.766(100)(113), 2.269(66)(024), 1.822(26)(033), 1.711(33)(220), 1.383(23)(128). The structure, refined to R1 = 0.023 for 183 Fo > 4σF reflections, shows leverettite to be isostructural with herbertsmithite and gillardite.
Front matter
MGM volume 77 issue 7 Cover and Front matter
-
- Published online by Cambridge University Press:
- 05 July 2018, pp. f1-f6
-
- Article
-
- You have access Access
- Export citation


 ,
,  , 040, 120), 11.661(8)(001), 2.721(3)(
, 040, 120), 11.661(8)(001), 2.721(3)(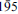 ), 4.404(3)(
), 4.404(3)( ,
, 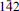 ), 4.056(3)(031, 1
), 4.056(3)(031, 1 ,
, 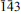 ), 3.891(2)(003).
), 3.891(2)(003). ,
, 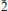 1,020) 52, 5.24 (100,012,
1,020) 52, 5.24 (100,012, 2,
2, m,
m, 
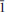 ); 3.415(27.83)(300); 5.125(24.70)(200); and 4.988(22.98)(111). Deveroite-(Ce) is named in recognition of Devero valley and Devero Natural Park.
); 3.415(27.83)(300); 5.125(24.70)(200); and 4.988(22.98)(111). Deveroite-(Ce) is named in recognition of Devero valley and Devero Natural Park.
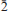 14), 2.972(66)(
14), 2.972(66)(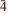 24) and 2.133(54)(402). Barikaite is the N = 4 member of the sartorite homologous series with a near-equal role of As and Sb, which have an ordered distribution pattern in the structure. It is a close homeotype of rathite and more distantly related to dufrénoysite (both distinct, pure arsenian N = 4 members) and it completes the spectrum of Sb-rich members of the sartorite homologous series. The new mineral and its name have been approved by the IMA-CNMNC (IMA 2012-055).
24) and 2.133(54)(402). Barikaite is the N = 4 member of the sartorite homologous series with a near-equal role of As and Sb, which have an ordered distribution pattern in the structure. It is a close homeotype of rathite and more distantly related to dufrénoysite (both distinct, pure arsenian N = 4 members) and it completes the spectrum of Sb-rich members of the sartorite homologous series. The new mineral and its name have been approved by the IMA-CNMNC (IMA 2012-055).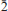 }. The crystals can also form finger-like, parallel stacked growths along the
}. The crystals can also form finger-like, parallel stacked growths along the 