Volume 57 - Issue 3 - June 2009
Article
Selectivity of Co and Ni by K-depleted micas
- Yunchul Cho, Sridhar Komarneni, Sang-il Choi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 279-289
-
- Article
-
- You have access Access
- Export citation
-
Contamination of the environment with heavy metals, including cationic radionuclides, is a serious problem which has yet to be fully overcome. A class of potentially effective cation exchangers for sequestering heavy metals which has received little attention is K-depleted mica. The purpose of this study was to investigate the heavy-metal cation exchange properties of K-depleted phlogopite and biotite, which were prepared from a natural phlogopite and biotite, respectively, using sodium tetraphenylborate (NaTPB). The X-ray diffraction (XRD) patterns showed that interlayer K+ ions were completely replaced with exchangeable Na+ ions, resulting in the expansion of the d001 spacing of both K-depleted phlogopite and K-depleted biotite. In order to investigate the cation exchange selectivity of K-depleted phlogopite and biotite for Co2+ and Ni2+, cation exchange isotherms and Kielland plots were constructed. The isotherms and Kielland plots indicated that both K-depleted phlogopite and biotite are highly selective for Co2+ as well as Ni2+. The XRD patterns after both 2Na+ → Co2+ and Ni2+ exchange reactions suggest that double sheets of interlayer water are present in the interlayer. These K-depleted micas are potential cation exchange materials for removal of some heavy metals such as Ni and radioactive species such as 60Co from solution.
Simple synthesis and characterization of nanoporous materials from talc
- Chunfang Du, Huaming Yang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 290-301
-
- Article
-
- You have access Access
- Export citation
-
Synthetic siliceous mesoporous materials are of great value in many different applications, including nanotechnology, biotechnology, information technology, and medical fields, but historically the resource materials used in their synthesis have been expensive. Recent efforts have focused on indirect synthesis methods which utilize less expensive silicate minerals as a resource material. The purpose of the present study was to investigate talc, a natural silicate mineral, as one such resource. It was used as raw material to prepare two advanced materials: porous silica (PS) and ordered mesoporous silica (MCM-41). The PS, with a specific surface area of 260 m2/g and bimodal pore-size distribution of 1.2 nm and 3.7 nm, was prepared by grinding and subsequent acid leaching. The MCM-41, with a large surface area of 974 m2/g and a narrow pore-size distribution of 2.8 nm, was obtained using a surfactant, cetyltrimethylammonium bromide (CTAB), by hydrothermal treatment using the as-prepared PS as a source of Si. The two resultant materials were characterized by small angle X-ray diffraction (SAXRD) and wide-angle X-ray diffraction (WAXRD), high-resolution transmission electron microscopy (HRTEM), solid-state magic-angle-spinning nuclear magnetic resonance (MAS NMR), Fourier transform infrared spectroscopy (FTIR), and N2 adsorption-desorption measurements. Based on these measurements, possible processes of transformation of PS from talc, upon acid treatment, and the formation of MCM-41 were investigated systemically. Acid leaching induced the transformation of a rigid layered structure to a nearly amorphous one, with micropores formed by a residual layered structure and mesopores formed from a condensed framework. The MCM-41 was a mixture of silanol groups (Si(SiO)3(OH)) and a condensed Q4 framework structure (Si(SiO)4), with a small amount of remaining Q3 layered structure (Si(SiO)3OMg). The increased Q4/Q3 value confirmed greater polymerization of MCM-41 than of PS. At the low CTAB concentration used (2 wt.%), the highly charged silicate species controlled the surfactant geometry. Charge-density matching, together with the degree of polymerization of the silicates, determined the resultant mesophase.
Application of 29Si and 27Al MAS NMR spectroscopy to the study of the reaction mechanism of kaolinite to illite/muscovite
- Marco Mantovani, Alberto Escudero, Ana Isabel Becerro
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 302-310
-
- Article
-
- You have access Access
- Export citation
-
Understanding the mechanisms for illitization of clay minerals has important applications in reconstructing geologic histories and determining the origins of physical and chemical characteristics of buried sediments. While many studies have been carried out on this topic, few have focused on the mechanism of illite formation from kaolinite. The purpose of this study was to investigate more deeply the illitization of kaolinite in KOH solution at a high solid/liquid ratio (1000 mg/mL). X-ray diffraction (XRD) and infrared spectroscopy were used to follow the formation of new crystalline phases and the composition of the octahedral sheet, while the transformation of the Si and Al local environments was analyzed by 29Si and 27Al magic angle spinning nuclear magnetic resonance spectroscopy (MAS NMR). The results revealed that the first reaction stage consists of the diffusion of Al from the octahedral to the tetrahedral sheet of the kaolinite TO layers, giving rise to the precursors of the illite/muscovite nuclei. Combination of XRD with 27Al MAS NMR measurements indicated that a minimum amount of tetrahedral Al is required in the original TO layer before condensation of a second tetrahedral sheet occurs to complete the formation of the illite/muscovite TOT layers.
The occurrence and origin of the söğüt kaolinite deposits in the Paleozoic Saricakaya granite-granodiorite complexes and overlying Neogene sediments (Bilecik, northwestern Turkey)
- Selahattin Kadir, Firdevs Kart
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 311-329
-
- Article
-
- You have access Access
- Export citation
-
The Söğüt kaolinite deposits at Çaltı, İnhisar, and Küre, Turkey, are an important source of raw materials for the ceramics industry of that country, but no detailed mineralogical or geochemical characterizations of these economically important materials have been carried out to date. The purpose of this study was to fill this gap by performing mineralogical, geochemical, and isotopic characterizations of these kaolinite deposits which occur within Paleozoic granite-granodiorite complexes that are crosscut by aplite and pegmatite dikes, and overlain by Neogene sedimentary units. These units are dominated by quartz veins and networks of subvertical fractures and weak zones that were invaded by hydrothermal fluids, resulting in their kaolinization and silicification. Altered units and related host rocks were examined using polarized-light microscopy, X-ray diffractometry, scanning electron microscopy, infrared spectroscopy, and chemical and isotopic methods. Feldspar crystals are either sericitized or kaolinized, and mica exhibits partial chloritization; Fe-Ti-Mn oxides occur within fractures. Kaolinite crystals occur in authigenic vermiform or plate-like stacked forms, having contacts with resorbed feldspar crystals which locally exhibit thick, platy, and subparallel orientations relative to microfractures, the pathways for hydrothermal-fluid injection. Altered feldspar relicts are associated mainly with kaolinite, smectite, quartz crystals, and illite/mica. Increase in (Al+Fe)/Si in the kaolinized units (relative to host-rock granite and granodiorite complexes and silicification), depletion of Ba+Rb, and a negative Eu anomaly reveal that the alteration of feldspar by hydrothermal fluid, the character of which was determined from O- and H-isotopic values, resulted in the precipitation of kaolinite. Thus, the Söğüt kaolinite deposit possibly formed by hydrothermal alteration and a feldspar dissolution-precipitation mechanism in both the granite-granodiorites complexes and related overlying sedimentary units under acidic environmental conditions, which developed via depletion of the soluble elements Na and Ca.
Synthesis of a Mg-Cd-Al layered double hydroxide and sorption of selenium
- Birte Hansen, Hilde Curtius, Reinhard Odoj
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 330-337
-
- Article
-
- You have access Access
- Export citation
-
Leaching experiments with metallic uranium-aluminum research-reactor fuel elements in repository-relevant MgCl2-rich salt brine (brine 2) were performed. A Mg-Al layered double hydroxide (LDH) with chloride as the interlayer anion was identified as a crystalline secondary-phase component. In the present study, the incorporation of Cd into the structure of the Mg-Al-Cl LDH was investigated. Synthesis by a coprecipitation method was performed and the Mg-Cd-Al-Cl LDH obtained was characterized. The sorption behavior of selenium on the LDH was investigated in water, clay pore-water (Mont-Terri-type), and brine 2. Using a LDH concentration of 10 g/L, the sorption kinetics were rapid and equilibrium was reached within 12 h. The sorption of selenium decreased with increasing amount of chloride anions in the solutions. The chloride anions acted as competing anions for the sorption of selenium. The effect of pH on selenium sorption was investigated and a large buffer capacity of the LDH was observed. For the range of selenium concentration used, linear sorption isotherms were obtained which obeyed the Freundlich and Dubinin-Radushkevich models. From these, the energies of selenium sorption were calculated to be in the range of ion-exchange processes.
Three new, quick CEC methods for determining the amounts of exchangeable calcium cations in calcareous clays
- R. Dohrmann, S. Kaufhold
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 338-352
-
- Article
-
- You have access Access
- Export citation
-
The cation exchange capacity (CEC) is one of the most important properties of clays in terms of their performance in both natural and technical processes. For decades, common methods for determining exchangeable cations have failed when calcareous clays or soils were examined, because calcite is at least partly dissolved throughout the exchange experiment which in turn increases measureable Ca2+ concentrations. As a result, exchangeable Ca2+ and the sum of exchangeable cations by far exceed the amount of negative charges. In the past, the silver-thiourea method (AgTU) has been modified to overcome this problem (AgTUcalcite), but is unsatisfactory as the method is laborious. In the present study three new methods based on two alternative metal complexes, cobalt(III) hexamine (CoHex) and copper(II) triethylenetetramine (Cu-trien), are proposed. The optimum solid/liquid ratios of these methods and the optimum complex concentration of Cu-trien are reported, depending on the mineralogical composition of the samples. The key development is that the exchange solutions are saturated with respect to calcite prior to the experiment. Approximately 70–90% of the dissolution of calcite present as an admixture in a clay sample is suppressed in the subsequent cation exchange experiment, but not all. The Ca2+ exchange is not suppressed and there is no evidence for any precipitation of this Ca2+. Three possibilities for how to handle this problem are discussed, one of which is to perform no further correction. The resulting error arises from the remaining calcite solubility of the different solutions after pre-treatment with calcite. This corresponds to errors of 0.2–1.3 (CoHexcalcite)and 0.7–8.4 (Cu-triencalcite) meq/100 gCa2+ for samples with small and large CEC values, respectively. As a consequence of the poor performance of Cu-triencalcite for samples with large CEC, a more concentrated Cu-trien5 × calcite solution was developed which performed much better: 0.1–0.8 meq/100 g(Cu-trien5 × calcite). For Cu-trien5 × calcite and CoHexcalcite at least, the errors are in the range of the non-systematic scattering for exchangeable Ca2+ determination. Therefore, the methods suggested provide ‘operationally correct’ Ca2+ values without additional effort. Moreover, owing to the high selectivity of the index cations applied in the present study, only one exchange step is required, providing a significant advantage over the AgTUcalcite method.
Factors governing the formation of lithiophorite at atmospheric pressure
- Haojie Cui, Lei You, Xionghan Feng, Wenfeng Tan, Guohong Qiu, Fan Liu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 353-360
-
- Article
-
- You have access Access
- Export citation
-
Lithiophorite is a naturally occurring Mn oxide mineral commonly found in soils and sediments. The usual method of synthesizing lithiophorite is via a hydrothermal process in an autoclave at relatively high temperature and pressure. In the present study, an alternative, reflux method, at atmospheric pressure, for synthesis of lithiophorite was developed successfully. The influence of reaction duration, temperature, type of precursor birnessite (H-birnessite, Na-birnessite, aged Na-birnessite), and pH on the formation of lithiophorite were investigated by reflux treatment of lithium-aluminum hydroxide complex ion (
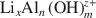 )-exchanged birnessite. The results show that the degree of conversion of lithiophorite decreases with decreasing reaction temperature. Lithiophorite can be obtained at pH values from 5.0 to 9.0, but a circumneutral pH is more favorable for formation at atmospheric pressure. Conversion of Na-birnessite (Bir-OH) to lithiophorite is more favored than aged Na-birnessite (Bir-OH-A). Lithiophorite was not obtained by refluxing the
)-exchanged birnessite. The results show that the degree of conversion of lithiophorite decreases with decreasing reaction temperature. Lithiophorite can be obtained at pH values from 5.0 to 9.0, but a circumneutral pH is more favorable for formation at atmospheric pressure. Conversion of Na-birnessite (Bir-OH) to lithiophorite is more favored than aged Na-birnessite (Bir-OH-A). Lithiophorite was not obtained by refluxing the 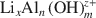 ion-exchanged H-birnessite (Bir-H) sample. The rate of conversion of lithiophorite increases with increasing reflux time. Lithiophorite synthesized by a reflux process has pseudo-hexagonal crystals of 0.1–0.5 µm with a chemical composition of Li0.24Al0.46MnO2.67(H2O)1.25. The results have important implications for the origin and underlying mechanism of lithiophorite formation in the environment.
ion-exchanged H-birnessite (Bir-H) sample. The rate of conversion of lithiophorite increases with increasing reflux time. Lithiophorite synthesized by a reflux process has pseudo-hexagonal crystals of 0.1–0.5 µm with a chemical composition of Li0.24Al0.46MnO2.67(H2O)1.25. The results have important implications for the origin and underlying mechanism of lithiophorite formation in the environment.
Surface properties of illite-smectite minerals as detected by interactions with rhodamine 6G dye
- V. Šucha, A. Czímerová, J. Bujdák
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 361-370
-
- Article
-
- You have access Access
- Export citation
-
Interactions between smectite clay minerals and various organic dyes have been studied extensively, but little information has accumulated from dye interactions with mixed-layer illite-smectite (I-S) minerals, especially regarding relationships with clay layer expandability, layer charge, particle size/shape, and molecular aggregation of organic dye molecules. The purpose of this study was to investigate the surface interactions of a set of mixed-layer illite-smectites from different geological environments with Rhodamine 6G dye. The samples used have different amounts of expandable smectite interlayers, different particle size and/or shape, and different layer-charge density at the surface. Five smectites with differences in layer charge and some non-expandable layer silicates were also tested. The interactions detected by UV-vis spectroscopy show no reaction between R6G and non-expandable minerals (kaolinite, mica), and intense reactions forming H-aggregates and monomers with smectites and illite-smectites. The intensity of H-aggregate formation increases with increase in the layer charge of smectites. Mixed-layer illite-smectites interact with R6G more intensely than do smectites. H-aggregate and monomer formation increases with the illitization process for randomly ordered illite-smectites (R = 0) and decreases in the course of illitization for the ordered illite-smectites (R > 0).
Application of chemical geothermometry to low-temperature trioctahedral chlorites
- Atsuyuki Inoue, Alain Meunier, Patricia Patrier-Mas, Cecile Rigault, Daniel Beaufort, Philippe Vieillard
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 371-382
-
- Article
-
- You have access Access
- Export citation
-
Low-temperature chlorites formed in diagenetic to low-grade metamorphic environments generally have greater Si contents and larger numbers of octahedral vacancies, and smaller Fe+Mg contents than higher-grade metamorphic chlorites. The compositional variations are characterized approximately by four end-member components: Al-free trioctahedral chlorite, chamosite, corundophilite, and sudoite. The solid solution is considered to be a random mix of cations and vacancies in the octahedral sites. Using the compositions of chlorites from Niger, Rouez, and Saint Martin diagenetic-hydrothermal series, a new, more convenient geothermometer, applicable to low-T chlorites is proposed and comparison made with geothermometers proposed previously. The chlorites studied contain appreciable amounts of Fe(III) (>14% of the total Fe), determined by Mössbauer spectroscopy. The calculations under which all Fe was regarded as ferrous gave considerable overestimates for the formation temperature, irrespective of the geothermometer used. This problem was reduced by taking into account the presence of Fe(III) in the octahedral sites. The geothermometer from this study gave more reasonable estimates than the geothermometers proposed by Walshe (1986) and Vidal et al. (2001), particularly in the case of the Niger chlorites which crystallized in the lowest-temperature conditions. The ordered-site substitution model of solid solution developed by Vidal et al. (2001) predicted satisfactorily the formation temperature of the Rouez chlorites and of some of the Saint Martin chlorites, suggesting that the chlorite compositions are controlled by the
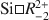 exchange at low-T conditions while they are controlled by Tschermak exchange at higher temperatures. The decreasing number of vacancies with temperature are poorer in Fe-rich than in Fe-poor chlorites. Furthermore, the ordered-site occupation of cations and vacancies in trioctahedral chlorite occurs concomitantly with the compositional changes ruled by increasing temperature conditions.
exchange at low-T conditions while they are controlled by Tschermak exchange at higher temperatures. The decreasing number of vacancies with temperature are poorer in Fe-rich than in Fe-poor chlorites. Furthermore, the ordered-site occupation of cations and vacancies in trioctahedral chlorite occurs concomitantly with the compositional changes ruled by increasing temperature conditions.
Isomerization of 1-butene catalyzed by surfactant-modified, Al2O3-pillared clays
- Eduardo González, Douglas Rodríguez, Lenin Huerta, Alexander Moronta
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 383-391
-
- Article
-
- You have access Access
- Export citation
-
Recent studies indicate that a template method for creating Al-pillared clays, in which surfactant micelles foster the creation of a homogeneous mesoporous network within the pillar, effectively enhance catalyst performance and adsorbent properties. No studies, however, have described the relative effects of the surfactant concentration and Al content on the textural and acidic properties and on the catalytic activity of the Al-pillared clays. The purpose of the present study was to fill this gap, using the isomerization of 1-butene as the test process for catalytic activity. Modified pillared clays (MPC) were prepared from a synthetic clay, TS-1, using different amounts of a non-ionic surfactant (Igepal CO-720) and a fixed concentration of a solution containing the Al polycation [Al13O4-(OH)24]7+. MPC with a fixed amount of surfactant and different amounts of Al were also prepared. The catalysts were characterized by X-ray diffraction (XRD), X-ray fluorescence (XRF), temperature-programmed desorption of ammonia, 27Al magic-angle spinning nuclear magnetic resonance (27Al MAS NMR), and N2 adsorption/desorption isotherms. Isomerization of 1-butene at 250°C was used to test the catalytic activity. Analyses by XRD and XRF showed that the synthesized solids were amorphous and that the amount of pillaring by Al increased with the amount of Al complex used. Interestingly, the surface area and pore volume were directly proportional to the amount of surfactant employed and decreased with increasing amounts of Al pillaring. All solids showed activity for 1-butene isomerization, with a maximum conversion of ∼75%. Only cis- and trans-2-butene were observed. The absence of isobutene suggested that acid sites of moderate strength were formed, in agreement with the results obtained from the desorption of ammonia.
Near-infrared spectroscopic analysis of acid-treated organo-clays
- Jana Madejová, Helena Pálková, Martin Pentrák, Peter Komadel
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 392-403
-
- Article
-
- You have access Access
- Export citation
-
The potential use of near-infrared (NIR) spectroscopy as a characterization tool for organo-clays would be a great asset but little work has been done in this regard because the application of NIR to clay mineral studies is a relatively new phenomenon. The purpose of this study was to use NIR spectroscopy to investigate the effect of alkylammonium cations on the acid dissolution of a high-charge montmorillonite (SAz-1). Detailed analysis of the spectra of Li+-, TMA+- (tetramethylammonium), and HDTMA+- (hexadecyltrimethylammonium) saturated SAz-1 montmorillonite in the NIR region was achieved by comparing the first overtone (2ν) and combination (ν+δ) bands of XH groups (X = O, C) with the fundamental stretching (ν) and bending (δ) vibrations observed in the mid-infrared (MIR) region. Comprehensive analysis of the vibrational modes of CH3-N, CH3-C, and -CH2-C groups of TMA+ and HDTMA+ cations detected in the MIR and NIR regions was also performed. Both MIR and NIR spectra demonstrated that exchange of Li+ by TMA+ only slightly improved the resistance of SAz-1 layers to dissolution in 6 M HCl at 80°C, while exchange by the larger HDTMA+ cations almost completely protected the montmorillonite layers from acid attack. Use of NIR spectra in reaching these conclusions was crucial. Only in the NIR region could the creation of SiOH groups be monitored, which is an important indicator of the acidification of the montmorillonite surface. The OH-overtone region in the spectra of Li-SAz-1 and TMA-SAz-1 revealed that the SiOH band near 7315 cm−1 increases in intensity with enhanced acid treatment. In contrast, no SiOH groups were identified in the NIR spectra of HDTMA-SAz-1 treated in HCl, indicating that HDTMA+ completely covers the inner and outer surfaces of the montmorillonite and hinders access ofprotons to the Si-O− bonds created upon acid treatment.

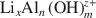 )-exchanged birnessite. The results show that the degree of conversion of lithiophorite decreases with decreasing reaction temperature. Lithiophorite can be obtained at pH values from 5.0 to 9.0, but a circumneutral pH is more favorable for formation at atmospheric pressure. Conversion of Na-birnessite (Bir-OH) to lithiophorite is more favored than aged Na-birnessite (Bir-OH-A). Lithiophorite was not obtained by refluxing the
)-exchanged birnessite. The results show that the degree of conversion of lithiophorite decreases with decreasing reaction temperature. Lithiophorite can be obtained at pH values from 5.0 to 9.0, but a circumneutral pH is more favorable for formation at atmospheric pressure. Conversion of Na-birnessite (Bir-OH) to lithiophorite is more favored than aged Na-birnessite (Bir-OH-A). Lithiophorite was not obtained by refluxing the 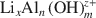 ion-exchanged H-birnessite (Bir-H) sample. The rate of conversion of lithiophorite increases with increasing reflux time. Lithiophorite synthesized by a reflux process has pseudo-hexagonal crystals of 0.1–0.5 µm with a chemical composition of Li
ion-exchanged H-birnessite (Bir-H) sample. The rate of conversion of lithiophorite increases with increasing reflux time. Lithiophorite synthesized by a reflux process has pseudo-hexagonal crystals of 0.1–0.5 µm with a chemical composition of Li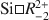 exchange at low-
exchange at low-