Volume 69 - December 2021
Article
Review: Progress with Functional Materials Based on Loess Particles
- Ya Shen, Haiyan Yan, Rongmin Wang, Pengfei Song, Yufeng He
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 301-314
-
- Article
- Export citation
-
Loess is a large-scale deposit which is easy to mine and widely distributed on the epipedon. The clay fraction of loess, also known as ‘loessial clay’, is a very important component of loess which affects its properties and performance. From a ‘materials’ perspective, the clay fraction of loess has been ignored. Recently, loess particles have attracted interest because of their potential applications. The focus in the current review is on the methods of modifying loess particles and their application as functional materials. The major components of loess particles are clays, calcite, and quartz, with the clays including kaolinite, illite, montmorillonite, and chlorite. Loess has a range of particle sizes, types, and dispersibilities. The particles agglomerate readily, mainly because cementation occurs readily in the clay fraction. Loess particles can be modified and their properties can be improved by compaction, separation, purification, acidification, calcination, surfactant modification, geopolymerization, and polymer modification. Loess-based functional materials have been used as sorbents, eco-friendly superabsorbents, soil and water conservation materials, humidity-regulating materials, and building materials. Separated and purified loess particles can adsorb metal ions and harmful elements directly. Surfactant-modified loess particles can remove organic compounds effectively. After modification with polymers, loess particles exhibit greater capacity for the removal of environmental pollutants such as harmful metal ions and dyes. As a superabsorbent, modified loess shows excellent thermal stability and swelling behavior. Calcined loess could be utilized as an energy-saving building material with good humidity-regulating performance, and geological polymerization has further expanded the scope of applications of loess in architecture. In summary, loess-based functional materials, which are inexpensive and ecologically friendly, deserve more attention and further development.
Formation of Humboldtine During the Dissolution of Hematite in Oxalic Acid – Density Functional Theory (DFT) Calculations and Experimental Verification
- Paula Vehmaanperä, Bo Gong, Patrick H.-L. Sit, Riina Salmimies, Bernardo Barbiellini, Antti Häkkinen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 655-662
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Understanding the reactions taking place in the hematite-oxalic acid system is important in order to clean iron oxides from filters and to remove iron from mineral concentrates. Previous studies reported the formation of an unwanted solid phase during this process. The objective of the current work, therefore, was to visualize and rationalize the iron dissolution steps taking place in the hematite–oxalic acid reaction by combining density functional theory (DFT) calculations and experimental data. The results of DFT calculations indicated that a precipitate was formed in this reaction; XRD analysis of the solid phase after the dissolution experiment revealed the formation of humboldtine as the precipitate. The attachment of oxalate on the hematite surface and the reduction of Fe(III) to Fe(II) were key steps for humboldtine formation. Both simulations and the experimental results showed that greater oxalic acid concentrations yielded more precipitate, suggesting a simple and novel route to synthesize humboldtine, a material which is relevant to the demand for clean energy.
A Molecular Dynamics Simulation Study of Fe-Containing Palygorskite
- Zhijun Lu, Jinhong Zhou, Xiancai Lu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 399-405
-
- Article
- Export citation
-
Fe is a common substituent in palygorskites (Plg), but its effect on the microscopic properties is unclear. In the current study, molecular dynamics (MD) simulations were carried out to investigate the effect of Fe on the properties of the nano-pores in Plg. The structures and dynamics of water and Na+ ions in the pores were computed by analyzing the MD trajectories. The results revealed that for both Fe-containing and ordinary Plg, zeolitic water molecules can diffuse into the pores with very low mobility whereas Mg-coordinated water fails to escape. Na+ ions show no obvious diffusivity because they are fixed above the Si–Osix-membered rings. Detailed comparison indicates that Fe-substitution has no significant influence on the pore properties of Plg.
Laterite as a Potential Seepage Barrier From a Karst-Depression Tailings Impoundment
- Hai-Yan Gao, Ze-Min Xu, Zhe Ren, Kun Wang, Kui Yang, Yong-Jun Tang, Jun-Yao Luo
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-22
-
- Article
- Export citation
-
In the absence of the necessary valley topography, karst depressions are sometimes used to construct conventional impoundments in order to contain tailings. Leakage is a primary concern for such impoundments. The purpose of the current study was to determine the characteristics and barrier performance of laterite mantling karst depressions, using, as an example, the Wujiwatang (WJWT) tailings impoundment, located in the Gejiu mining area, southwestern China. The geotechnical-hydrogeological properties, geochemistry, mineral compositions, and particle shapes of the laterite were investigated by geotechnical techniques, chemical analysis, X-ray diffraction (XRD), and scanning electron microscopy (SEM). The results showed that the laterite contained poorly sorted particles that covered a wide spectrum of grain sizes (<5 mm to <50 nm), and was unexpectedly categorized as silty clay or silt with a high liquid limit. The continuous gradation and small D90 value helped the laterite achieve saturated hydraulic conductivities in the range of <10–6 cm/s required for impoundment liners. The laterite beneath the tailings impoundment was finer-grained and had a lower permeability than that of the laterite on the depression walls within the same depression. Geochemically and mineralogically, the laterite was classified as true laterite and its major mineralogical constituents were gibbsite and goethite with chlorite occurring in trace amounts. The laterite was dominated by subspherolitic–spherolitic cohesionless grains (concretions) made up of Al, Fe, Ti, and Mn oxides and hydroxides. The laterite did not have plasticity indices in the clay range. Fortunately, slopewash prior to tailings containment selectively transported the finer oxide concretions to the depression floor, creating a natural low-permeability barrier for the WJWT tailings impoundment. This is undoubtedly important for the planning and design of future karst depression-type tailings impoundments around the world.
Review
Halloysite-Based Nanosystems for Biomedical Applications
- Francesca Persano, Giuseppe Gigli, Stefano Leporatti
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 501-521
-
- Article
- Export citation
-
Halloysite nanotubes (HNTs) are hollow clay nanotubes in the nanometer size range, made up of double-layered aluminum silicate mineral layers. HNTs represent an extremely versatile, safe, and biocompatible nanomaterial, used in a wide range of applications in biomedicine and nanomedicine. For example, they are used as transporters for the controlled release of drugs or genes, in tissue engineering, in the isolation of stem cells and cancer cells, and in bioimaging. Consequently, the assessment of the biocompatibility of HNTs has acquired considerable importance. In recent years, HNT composites have attracted attention due to their improved biocompatibility, compared to HNTs, suggesting potential for applications in tissue engineering or as vehicles for drugs or genes. In this review, recent advances in the application of HNTs and HNT composites in biomedicine are discussed to provide a valuable guide to scientists in the design and development of viable, functional bio-devices for biomedical applications.
Article
Pharmaco-Chemical Analysis and in Vivo Toxicity Assays of Edible Clays Mined in Nigeria and Consumed Globally
- Joy I. Odimegwu, Florence E. Nkemehule, Esther Jane-Sharon Eke, Jumoke Okunnuga, Aminat Omolara Buhari, Sarah Ngozichukwu Okwuegbuna, Chidinma Romanda Duru
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 155-166
-
- Article
- Export citation
-
Edible clays are consumed by diverse groups of people, especially of African descent, living in Africa and abroad, in a behavior known as geophagy. The clays are used topically as an emollient and drying agent and internally to control diarrhea. Scientific information concerning the chemical constituents and toxicity of edible clays is scarce. The aims of the present study, therefore, were to ascertain the chemical composition of white edible clays (WEC) and gray edible clays (GEC); to determine their toxicity profiles using analytical chemical methods; to test the acute and sub-acute toxicity of edible clays in their natural form as consumed; and to compare the raw and processed clays, and also to compare the latter to a proprietary drug known as ‘Mist kaolin’ (Moko®) which contains some clay along with other chemicals. Atomic absorption spectroscopy (AAS) and gas chromatography/mass spectrometry (GC/MS) were used to determine the elements present. White female Wistar mice and rats were used for the acute and sub-acute toxicity analyses, respectively. The results from AAS showed the presence of heavy metals and metalloids in both GEC and WEC, and the GC/MS revealed the presence of contaminants such as indomethacin and ethyl benzene, but quantities were below human toxicity levels. Doses of 100–500 mg/kg of either clay type could be harmful to the digestive system, but all of the tests revealed that edible clay is not toxic to humans unless very large amounts (500–1000 mg/kg of body weight) are consumed.
Geochemistry and Mineralogy of Ice-Dammed Lake Sediments of the Lębork Deposit
- Radosław Rogoziński, Alina Maciejewska
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 315-327
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Varved clay deposits from ice-dammed lakes are a particularly important and broadly applied raw material used for the production of high-quality ceramics (red bricks, roof tiles, etc.), but the mineralogy and geochemistry of these sediments are not fully understood. The aim of the present study was to determine the chemical and mineralogical composition of ice-dammed lake sediments of the Lębork deposit. Major-element analysis of the compositions of selected samples from the ice-dammed lake clays was performed by X-ray fluorescence (XRF) and trace elements were determined by inductively coupled plasma-mass spectrometry. The mineralogical composition of clay samples was determined by X-ray diffraction (XRD). Analyses of the chemical composition of the ice-dammed lake clays of the Lębork deposit showed that the dominant component was SiO2 with a mean content of 56.13 wt.%; the second most abundant component was Al2O3, with a mean content for the entire deposit of 11.61 wt.%. Analysis by ICP-MS indicated the presence of rare earth elements (REE), e.g. cerium, neodymium, lanthanum, and praseodymium; their mean contents are: 56.9, 27.0, 26.3, and 7.3 ppm, respectively. Mineralogical analysis of the varved clays identified quartz, muscovite, calcite, and clay minerals – illite, kaolinite, and montmorillonite. The material filling the Lębork basin is characterized by small lateral and vertical variability in chemical composition. The results of the present study may be of considerable importance in determining the parent igneous, metamorphic, and sedimentary rocks, the weathering products of which supplied material to the ice-dammed lake, as well as in determining the mechanisms and character of the sedimentation process itself.
A Comparison of the Adsorption of Cesium on Zeolite Minerals vs Vermiculite
- D. R. Ferreira, G. D. Phillips, B. Baruah
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 663-671
-
- Article
- Export citation
-
Radiocesium was deposited on the soils of Fukushima Prefecture in Japan after the meltdown of the Fukushima Daiichi Nuclear Power Plant in 2011. The radiocesium bound to 2:1 clay minerals, such as vermiculite, common in the soil of that region and became non-exchangeable due to the strong affinity of these clay minerals for the Cs+ adsorbed. The current study generated adsorption envelopes for Cs+ on three zeolite minerals: zeolite Y, ZSM-5, and ferrierite. Two of these (ZSM-5 and ferrierite) caused monovalent cations to adsorb via a strong inner-sphere mechanism. A comparison of Cs+ adsorption on these zeolites to Na+ adsorption on the same zeolites showed that Cs+ adsorbs much more strongly than Na+, which is explained by its atomic properties. Despite the inner-sphere adsorption of Cs+ on ZSM-5 and ferrierite, the affinity of vermiculite for Cs+ is even stronger. An adsorption envelope for Cs+ on vermiculite failed to show a low-pH adsorption edge even at a pH of 1.01, with adsorption remaining at ~65% of the maximum even at this low pH. The adsorption envelopes for Cs+ on ZSM-5 and ferrierite minerals did show low-pH adsorption edges centered at pH 3.5 and 3.0, respectively, where Cs+ adsorption dropped to zero. The greater affinity of vermiculite for Cs+, even when compared with that for two zeolite minerals known to have significant affinities for monovalent ions, highlights the difficulty in removing Cs+ from contaminated Fukushima soils.
Ion-Exchange Modeling of Divalent Cation Adsorption on SWy-3 Montmorillonite
- Yayu W. Li, Cristian P. Schulthess
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 167-187
-
- Article
- Export citation
-
Ion-exchange modeling is used widely to describe and predict ion-adsorption data on clay minerals. Although the model parameters are usually optimized by curve fitting experimental data, this approach does not confirm the identity of the adsorption sites. The purpose of the present study was to extend to divalent cations a previous study on the retention of monovalent cations on Na-saturated montmorillonite (NaMnt) which optimized some of the model parameters using density functional theory (DFT) simulations. The adsorption strength of divalent cations increased in the order Mg2+ < Cd2+ < Ca2+ < Sr2+ < Ba2+. After adding adsorption of metal hydroxide species (MOH+), the three-site ion-exchange model was able to describe adsorption data over a wide pH range (pH 1–10) on NaMnt. X-ray diffraction (XRD) analyses were conducted to investigate the interlayer dimension of clay samples under various conditions. The cation retention strengths of divalent cations did not correlate with interlayer dimensions. The XRD analyses of the Mnt showed a d001 value of 19.6 Å when saturated with alkaline earth cations, 22.1 Å with Cd2+, 15.6 Å with Na+, and 15.2 Å with H+. In the case of Na+, the 15.6 Å peak decreased gradually and disappeared, and new peaks at 22.1 and 19.6 Å appeared when the percentages of Mg2+ and Ba2+ adsorbed increased on NaMnt. The peak shifted from 22.1 to 20.3 and 19.6 Å when the pH increased for all cations except Cd2+, which stayed constant at 22.1 Å. The coexistence of multiple d001 peaks in the XRD patterns suggested that the interlayer cations were segregated, and that the interlayer ion–ion interactions among different types of ions were minimized.
Montmorillonite-Hydrochar Nanocomposites as Examples of Clay–Organic Interactions Delivering Ecosystem Services
- Guodong Yuan, Jing Wei, Benny K. G. Theng
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 406-415
-
- Article
- Export citation
-
Clay–organic interaction is an important natural process that underpins soil ecosystem services. This process can also be tailored to produce clay–organic nanocomposites for industrial and environmental applications. The organic moiety of the nanocomposites, typically represented by a toxic surfactant, could be replaced by hydrochar formed from biomolecules (e.g. glucose) via hydrothermal carbonization. The effect of montmorillonite (Mnt) and glucose dosage on hydrochar formation, however, has not been clarified. In addition, the mechanisms by which Mnt-hydrochar nanocomposites (CMnt) can detoxify and remove carcinogenic Cr(VI) from aqueous solution are not well understood. In the current study, research milestones in terms of clay–organic interactions are summarized, following which the synthesis and characterization of CMnt for Cr(VI) adsorption are outlined. Briefly, 1 g of Mnt was reacted with 75 mL of glucose solution (0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 mol L−1) by hydrothermal carbonization at 200°C for 16 h. The resultant CMnt samples were analyzed for chemical composition, functional groups, morphological features, and Cr(VI) adsorptive properties. Mnt promoted the conversion of glucose to hydrochars, the particle size of which (~80 nm) was appreciably smaller than that formed in the absence of Mnt (control). Furthermore, the hydrochars in CMnt had an aromatic structure with low hydrogen substitution and high stability (C/H atomic ratio 0.34–0.99). The weakened OH (from hydrochar) and Si–O–Si stretching peaks in the Fourier-transform infrared (FTIR) spectra of CMnt are indicative of chemical bonding between Mnt and hydrochar. The CMnt samples were effective at removing toxic Cr(VI) from acidic aqueous solutions. Several processes were involved, including direct reduction of Cr(VI) to Cr(III), complexation of Cr(III) with carboxyl and phenolic groups of hydrochar, electrostatic attraction between Cr(VI) and positively charged CMnt at pH 2 followed by indirect reduction of Cr(VI) to Cr(III), and Cr(III) precipitation.
Composite Biodegradable Polymeric Matrix Doped With Halloysite Nanotubes for the Repair of Bone Defects in Dogs
- Ekaterina Naumenko, Elena Zakirova, Ivan Guryanov, Farida Akhatova, Mikhail Sergeev, Anastasia Valeeva, Rawil Fakhrullin
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 522-532
-
- Article
- Export citation
-
The use, in veterinary practice, of a three-dimensional biopolymer matrix (based on chitosan, agarose, and gelatin and doped with halloysite nanotubes) as a vehicle for mesenchymal stem cells (MSCs) to repair bone defects is reported here. The nanocomposite, combined with allogenic adipose-derived stem cells, was suitable for the repair of bone defects in dogs when paired with standard surgery involving metal Kirshner wires. The absence of inflammatory reactions to biopolymer matrices with allogenic stem cells was revealed in the case of an animal prone to inflammatory and allergic reactions. In addition, positive dynamics in the fusion of chronic bone defects without rejection reactions was observed after using a biopolymer matrix with MSCs.
Genesis of Palygorskite in the Neogene Baiyanghe Formation in Yangtaiwatan Basin, Northwest China, Based on the Mineralogical Characteristics and Occurrence of Enriched Trace Elements and Ree
- Shuai Zhang, Lihui Liu, Qinfu Liu, Bingjie Zhang, Zhichuan Qiao, Brian J. Teppen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 23-37
-
- Article
- Export citation
-
Palygorskite-rich mudstone interbedded with gypsum occurs in the Neogene Baiyanghe Formation in the Yangtaiwatan basin, northwest China, but the genesis of palygorskite in the mudstone is unclear. The objective of the present study was to clarify the manner by which the palygorskite evolved by analyzing the mineralogical and geochemical characteristics of the mudstone. The mineralogical composition of bulk-rock mudstone consisted of clay minerals, quartz, feldspar, dolomite, calcite, and gypsum. Palygorskite is dominant in the clay fraction together with illite and accessory chlorite and kaolinite. The interwoven rod-like palygorskite aggregates and delicate palygorskite particles indicated an authigenic origin. The bulk rock of palygorskite-rich mudstone was rich in the trace elements Cs, U, B, Li, Sb, Bi, and As, which, together with REE, all showed very positive correlation with major element oxides Al2O3, Fe2O3, MgO, K2O, and TiO2 of the mudstone, indicating that the REE and trace elements occurred mainly in the clay minerals. The detrital illite, chlorite, and kaolinite were the main original host of the REE and enriched trace elements. Statistical analyses showed that the authigenic palygorskite had strong affinity to such elements compared to the detrital clay minerals. In addition, the chondrite-normalized REE of the bulk mudstone showed essentially the same pattern irrespective of the proportions of detrital illite, kaolinite, chlorite, and authigenic palygorskite present in the samples. Thus, the conclusion reached was that palygorskite was generated from the dissolution of detrital clay minerals. The REE and enriched trace elements in authigenic palygorskite were inherited from the detrital clay minerals.
Meifuite, A New Ferrous Phyllosilicate Mineral With Modulated Tetrahedral Sheets Similar to Minnesotaite
- Shiyun Jin, Xiaochun Li, Franklin Hobbs, Stephen Guggenheim, And Huifang Xu
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 672-687
-
- Article
- Export citation
-
A new ferrous phyllosilicate, meifuite, has been discovered in the Yinachang Fe-Cu-REE (rare-earth element) deposit in China. The structural formula, calculated using averaged electron probe microanalysis (EPMA) results, is K0.72Na0.20(Fe5.56Mg0.31Mn0.13)Σ6.00(Si6.95Al1.04)Σ7.99O18.84(OH)4.84 Cl1.33, with an ideal formula of KFe6(AlSi7)O19(OH)4Cl2. The structure of meifuite has a
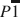 space group symmetry, with unit-cell parameters of a = 22.7773(13) Å, b = 9.5553(5) Å, c =14.3282(8) Å, α = 99.258(4)°, β = 136.750(3)°, γ = 89.899(4)°, Z = 2, and V = 2077.9(2) Å3. Meifuite has a strip-modulated 2:1 layer (T–O–T) structure similar to that of minnesotaite. About 1/8 of the tetrahedra in the T sheet are occupied by Al instead of Si, and the interlayer cavities are partially occupied by K and Na. Some of the OH sites in the octahedral sheet in the layer structure are fully or partially substituted by Cl, which is apparently the primary reason for the meifuite structure being more stable than stilpnomelane, the most common ferrous layer silicate mineral found at similar temperature and pressure conditions. An updated, more accurate structure model of minnesotaite is also provided for comparison with the meifuite structure. The mineral is named after Meifu Zhou in honor of his outstanding contributions to the field of economic geology.
space group symmetry, with unit-cell parameters of a = 22.7773(13) Å, b = 9.5553(5) Å, c =14.3282(8) Å, α = 99.258(4)°, β = 136.750(3)°, γ = 89.899(4)°, Z = 2, and V = 2077.9(2) Å3. Meifuite has a strip-modulated 2:1 layer (T–O–T) structure similar to that of minnesotaite. About 1/8 of the tetrahedra in the T sheet are occupied by Al instead of Si, and the interlayer cavities are partially occupied by K and Na. Some of the OH sites in the octahedral sheet in the layer structure are fully or partially substituted by Cl, which is apparently the primary reason for the meifuite structure being more stable than stilpnomelane, the most common ferrous layer silicate mineral found at similar temperature and pressure conditions. An updated, more accurate structure model of minnesotaite is also provided for comparison with the meifuite structure. The mineral is named after Meifu Zhou in honor of his outstanding contributions to the field of economic geology.
Original Paper
Niclosamide-Exfoliated Anionic Clay Nanohybrid Repurposed as an Antiviral Drug for Tackling Covid-19; Oral Formulation with Tween 60/Eudragit S100
- N. Sanoj Rejinold, Huiyan Piao, Goeun Choi, Geun-Woo Jin, Jin-Ho Choy
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 533-546
-
- Article
- Export citation
-
The ongoing pandemic, COVID-19 (SARS-CoV-2), has afflicted millions of people around the world, necessitating that the scientific community work, diligently and promptly, on suitable medicaments. Although vaccination programs have been run globally, the new variants of COVID-19 make it difficult to restrict the spread of the virus by vaccination alone. The combination of vaccination with anti-viral drug formulation is an ideal strategy for tackling the current pandemic situation. Drugs approved by the United States Food and Drug Administration (FDA), such as Remdesivir, have been found to be of little or no benefit. On the other hand, re-purposing of FDA-approved drugs, such as niclosamide (NIC), has offered promise but its applicability is limited due to its poor aqueous solubility and, therefore, low bioavailability. With advanced nano-pharmaceutical approaches, re-purposing this drug in a suitable drug-carrier for a better outcome may be possible. In the current study, an attempt was made to explore the loading of NIC into exfoliated layered double hydroxide nanoparticles (X-LDH NPs); prepared NIC-X-LDH NPs were further modified with eudragit S100 (ES100), an enteric coating polymer, to make the final product, ES100-NIC-X-LDH NPs, to improve absorption by the gastro/intestinal tract (GIT). Furthermore, Tween 60 was added as a coating on ES100-NIC-X-LDH NPs, not just to enhance its in vitro and in vivo stability, but also to enhance its mucoadhesive property, and to obtain, ultimately, better in vivo pharmacokinetic (PK) parameters upon oral administration. Release of NIC from Tween 60-ES100-NIC-X-LDH NPs was found to be greater under gastro/intestinal solution within a shorter period of time than the uncoated samples. The in vivo analysis revealed that Tween 60-ES100-NIC-X-LDH NPs were able to maintain a therapeutically relevant NIC plasma concentration in terms of PK parameters compared to the commercially available Yomesan®, proving that the new formulation might prove to be an effective oral drug-delivery system to deal with the SARS-CoV-2 viral infections. Further studies are required to ensure their safety and anti-viral efficacy.
Article
Effect of Montmorillonite Layer Charge on the Thermal Stability of Bentonite
- Yating Qin, Tongjiang Peng, Hongjuan Sun, Li Zeng, Yao Li, Can Zhou
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 328-338
-
- Article
- Export citation
-
The thermal stability of bentonite is vitally important for its application in the casting field and the layer charge of montmorillonite (Qm) is one of its central crystal-chemical parameters. As the main component of bentonite, the influence of Qm on montmorillonite properties and behavior needs to be considered if bentonite is to be used in high-temperature environments. The objective of the current study was to investigate the relationship between Qm and the thermal stability of Chinese bentonite samples collected from Wuhu, Anhui Province (marked as WH); Xinyang, Henan Province (marked as XY); and Santai, Sichuan Province (marked as ST) below. The values of Qm were obtained using the O (11) method, and the structural properties of the bentonite samples were characterized by X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR), thermogravimetry-differential scanning calorimetry (TG-DSC), and field emission scanning electron microscopy (FESEM). The results showed that, in the samples investigated, Qm was inversely related to the thermal stability of bentonite. The Qm value (electrons per half unit cell, e/huc) was greatest for sample ST (0.725 e/huc), followed by sample XY (0.470 e/huc), and by sample WH (0.354 e/huc). The dehydroxylation temperature changed related to Qm; the sample with the largest Qm value was WH (701°C), followed by sample XY (684°C), and sample ST (630°C). After the samples were calcined at 600°C, sample WH had the best montmorillonite structural integrity with the greatest degree of reusability (78.21%); while the montmorillonite structures of samples XY and ST were destroyed, and their reusabilities were only 9.48 and 6.01%, respectively.
Efficient Concentration of PB From Water by Reactions With Layered Alkali Silicates, Magadiite and Octosilicate
- Donhatai Sruamsiri, Thipwipa Sirinakorn, Makoto Ogawa
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 416-424
-
- Article
- Export citation
-
Human health problems are often related to contamination of the aqueous environment by toxic metal ions. In the present study, two layered alkali silicates (magadiite and octosilicate) were examined to assess removal of Pb2+ from aqueous solutions in terms of quantity and kinetics. The ion-exchange reaction between the silicates and aqueous solutions of lead(II) acetate at various concentrations was examined at room temperature for 24 h. The adsorption isotherms were H-type, showing the strong interactions between Pb2+ and the silicates. The amounts of Pb2+ adsorbed were as much as 1.23 mmol Pb/g magadiite and 2.32 mmol Pb/g octosilicate, which are larger than the reported values for various ion exchangers. They were larger than the theoretical cation exchange capacities (2.2 and 3.7 meq/g for magadiite and octosilicate, respectively), suggesting that the collection of Pb2+ included the precipitation as basic lead salts in addition to the ion exchange. The adsorption isotherms for magadiite and octosilicate fitted the Langmuir equation with correlation coefficients, R2, of 0.9991 and 0.9972, respectively. The adsorption of Pb2+ onto the layered alkali silicates from acidic aqueous solution was examined to obtain smaller amounts of adsorbed Pb2+ (0.32 mmol Pb/g magadiite and 0.34 mmol Pb/g octosilicate), confirming the important role of pH on the surface charge of the layered silicates in terms of ion exchange. The adsorption of Pb2+ reached equilibrium within 5 min for magadiite while it took 60 min for octosilicate. The difference was in the particle morphology; smoother diffusion of Pb2+ was possible through flower-shaped aggregates of particles of magadiite.
Automated Full-Pattern Summation of X-Ray Powder Diffraction Data for High-Throughput Quantification of Clay-Bearing Mixtures
- Benjamin M. Butler, Stephen Hillier
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 38-51
-
- Article
- Export citation
-
X-ray powder diffraction (XRPD) is found consistently to be the most accurate analytical technique for quantitative analysis of clay-bearing mixtures based on results from round-robin competitions such as the Reynolds Cup (RC). A range of computationally intensive approaches can be used to quantify phase concentrations from XRPD data, of which the ‘full-pattern summation of prior measured standards’ (FPS) has proven accurate and parsimonious. Despite its proven utility, the approach often requires time-consuming selection of appropriate pure reference patterns to use for a given sample. As such, applying FPS to large and mineralogically diverse datasets is challenging. In the present work, the accuracy of an automated FPS algorithm implemented within the powdR package for the R Language and Environment for Statistical Computing was tested on a set of 27 samples from nine RC contests. The samples represent challenging and diverse clay-bearing mixtures with known concentrations, with the added advantage of allowing the accuracy of the algorithm to be compared with results submitted to previous contests. When supplied with a library of 201 reference patterns representing a comprehensive range of phases that may be encountered in natural clay-bearing mixtures, the algorithm selected appropriate phases and achieved a mean absolute bias of 0.57% for non-clay minerals (n = 275), 2.37% for clay minerals (n = 120), and 4.43% for amorphous phases (n = 14). This accuracy would be sufficient for top-3 placings in all nine RC contests held to date (RC1 = 2nd, RC2 = 2nd, RC3 = 1st; RC4 = 2nd; RC5 = 1st; RC6 = 3rd; RC7 = 3rd; RC8 = 1st; RC9 = 2nd). The comparatively low values of absolute bias in combination with the competitive placings in all RC contests tested is particularly promising for the future of automated quantitative phase analyses by XRPD.
Incorporation of Mo6+ in Ferrihydrite, Goethite, and Hematite
- Marcel G. Görn, Ralph M. Bolanz, Stephen Parry, Jörg Göttlicher, Ralph Steininger, Juraj Majzlan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 188-204
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Among all iron oxides, hematite (α-Fe2O3), goethite (α-FeOOH), and ferrihydrite (FeOOH⋅nH2O) are the most common mineral species. While immobilization of Mo6+ by surface adsorption on ferric oxides has been studied extensively, the mechanisms of incorporation in their structure have been researched little. The objective of this study was to investigate the relation between Mo content and its structural incorporation in hematite, goethite, and six-line ferrihydrite by a combination of X-ray absorption spectroscopy (XAS), powder X-ray diffraction (pXRD), and inductively-coupled plasma optical emission spectrometry (ICP-OES). Synthesized in the presence of Mo, the hematite, goethite, and six-line ferrihydrite phases incorporated up to 8.52, 0.03, and 17.49 wt. % Mo, respectively. For hematite and goethite, pXRD analyses did not indicate the presence of separate Mo phases. Refined unit-cell parameters correlated with increasing Mo concentration in hematite and goethite. The unit-cell parameters indicated an increase in structural disorder within both phases and, therefore, supported the structural incorporation of Mo in hematite and goethite. Analysis of pXRD measurements of Mo-bearing six-line ferrihydrites revealed small amounts of coprecipitated akaganéite. X-ray absorption near edge structure (XANES) measurements at the Mo L3-edge indicated a strong distortion of the MoO6 octahedra in all three phases. Fitting of extended X-ray absorption fine structure (EXAFS) spectra of the Mo K-edge supported the presence of such distorted octahedra in a coordination environment similar to the Fe position in the investigated specimen. Incorporation of Mo6+ at the Fe3+-position for both hematite and goethite resulted in the formation of one Fe vacancy in close proximity to the newly incorporated Mo6+ and, therefore, charge balance within the hematite and goethite structures.
Interfacial X-Ray Scattering From Small Surfaces: Adapting Mineral-Fluid Structure Methods for Microcrystalline Materials
- Joanne E. Stubbs, Anna K. Wanhala, Peter J. Eng
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 688-701
-
- Article
- Export citation
-
Crystal truncation rod (CTR) X-ray diffraction is an invaluable tool for measuring mineral surface and adsorbate structures, and has been applied to several environmentally and geochemically important systems. Traditionally, the method has been restricted to single crystals with lateral dimensions >3 mm. Minerals that meet this size criterion represent a minute fraction of those that are relevant to interfacial geochemistry questions, however. Crystal screening, data collection, and CTR measurement methods have been developed for crystals of <0.3 mm in lateral size using the manganese oxide mineral chalcophanite (ZnMn3O7·3H2O) as a case study. This work demonstrates the feasibility of applying the CTR technique to previously inaccessible surfaces, opening up a large suite of candidate substrates for future study.
Processing and Characterization of Low-Thermal Conductivity, Clay-Based, Ceramic Membranes for Filtering Drinking Water
- Mohamed Mouafon, Gisèle Laure Lecomte-Nana, Nicolas Tessier-Doyen, André Njoya, Dayirou Njoya, Daniel Njopwouo
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 339-353
-
- Article
- Export citation
-
Drinking-water supply remains a significant challenge in tropical areas; to help meet this challenge, the purpose of the present study was to manufacture low-thermal conductivity ceramic membranes suitable for the retention/removal of particles found in non-potable water. These membranes with significant chemical and mechanical resistances were developed from Cameroonian clays, cassava starch, and bovine bone ash. Up to 30% of Cassava starch and bovine bone ash were added to the membrane as porogens (materials used to make pores in membranes). Membranes were manufactured by uniaxial pressing, drying at 105°C, and sintering at 1150°C for 2 h. The effects of various types of porogen on the thermal behavior, microstructure, flexural strength, porosity, and permeability of ceramic membranes were investigated to determine possible applications of those membranes for water filtration in the tropics. The thermal conductivity of membranes produced without a pore-forming agent (SM0) was greater (0.54 Wm–1K–1) than those produced with starch (SM1 and SM3) (0.45–0.40 Wm–1K–1) or bovine bone ash (SM2) (0.49 Wm–1K–1). The total porosity of SM0s (30.72%) was less than those of starch and bovine bone membranes (37.87–45.99%). The average pore size (0.04 μm) of SM2 membranes was the smallest: SM0 (0.09 μm), SM1 (0.10 μm), and SM3 (0.07 μm). The maximum pore size was 0.37 μm, indicating that membranes contain mesopores and macropores. The flexural strengths of SM1 and SM3 membranes (8.85 and 6.97 MPa, respectively) were less than those of SM2 (10.53 MPa) and SM0 (10.28 MPa), and water permeability from 108 L/h·m2 bar to 2198 L/h·m2 bar. Filtered water properties showed that pH values were upgraded from 5.9 to 7, the turbidity reduction rates and levels were >94% and <0.65 NTU. Particle-size distributions moved from 1150–39,000 nm in polluted water to <2 nm in filtered water. Judging by the sizes of particles present in filtered waters, these membranes may be suitable for elimination of viruses, pigments, proteins, colloids, and bacteria.