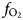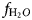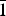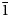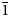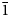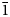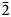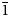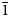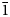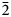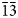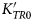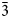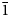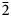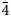Volume 83 - December 2019
Article
Magma chamber evolution of the Ardestan pluton, Central Iran: evidence from mineral chemistry, zircon composition and crystal size distribution
- Shahrouz Babazadeh, Tanya Furman, John M. Cottle, Davood Raeisi, Ianna Lima
-
- Published online by Cambridge University Press:
- 01 July 2019, pp. 763-780
-
- Article
- Export citation
-
The Oligo–Miocene Ardestan quartz diorite to tonalite is part of widespread Cenozoic magmatism within the Urumieh–Dokhtar Magmatic Assemblage of Iran. The Ardestan pluton is composed mainly of varying proportions of plagioclase feldspar (normally zoned from bytownite to andesine), amphibole (magnesio-hornblende) and biotite. Biotite exhibits a range of Al values (~2–2.8 apfu) over very restricted Fe# ratios (0.42–0.56) which are characteristic of continental arc magmatic suites. High Ti2O contents of biotite (<6.1 wt.%) suggest a magmatic origin. Ti-in-biotite geothermometery gives a mean crystallisation temperature of 730 ± 56°C, slightly higher than calculated TZr.Ti°C (716 ± 50°C) and similar to the average TZr.sat°C (735 ± 26°C). These results are consistent with the low bulk-rock SiO2 contents, which provide minimum estimates of temperature and indicate zircon crystallised from a fractionated magma. Zircons from the Ardestan pluton have high (Sm/La)N (>10) ratios suggesting a magmatic origin. T–
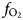 $f_{{\rm O}_{\rm 2}}$ calculations of oxygen fugacity between –13.6 to –16.9 indicate oxidising crystallisation conditions between the Ni–NiO (NNO) and Fe2O3–Fe3O4 (HM) buffers. Tight linear trends of log (XF/XOH), log (XCl/XOH) and log (XCl/XOH) vs. XMg represent a narrow range of
$f_{{\rm O}_{\rm 2}}$ calculations of oxygen fugacity between –13.6 to –16.9 indicate oxidising crystallisation conditions between the Ni–NiO (NNO) and Fe2O3–Fe3O4 (HM) buffers. Tight linear trends of log (XF/XOH), log (XCl/XOH) and log (XCl/XOH) vs. XMg represent a narrow range of 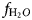 $f_{{\rm H}_2O}$, fHF and fHCl, clearly indicating that physico-chemical conditions were essentially constant throughout the formation of magmatic biotite. The shape of crystal size distribution curves along with the medium Al and Mg contents in amphibole and biotite, respectively, are consistent with a history of magma mixing involving injections of basic magma into the evolving felsic chamber. Calculated residence time for Ardestan plagioclase crystals of ~630 years support field evidence that these plutons were emplaced at shallow depths.
$f_{{\rm H}_2O}$, fHF and fHCl, clearly indicating that physico-chemical conditions were essentially constant throughout the formation of magmatic biotite. The shape of crystal size distribution curves along with the medium Al and Mg contents in amphibole and biotite, respectively, are consistent with a history of magma mixing involving injections of basic magma into the evolving felsic chamber. Calculated residence time for Ardestan plagioclase crystals of ~630 years support field evidence that these plutons were emplaced at shallow depths.
Mineral extinction
- Stuart J. Mills, Andrew G. Christy
-
- Published online by Cambridge University Press:
- 20 September 2019, pp. 621-625
-
- Article
-
- You have access Access
- HTML
- Export citation
-
‘Mineral evolution’ has attracted much attention in the last decade as a counterpart of the long-established biological concept, but is there a corresponding ‘mineral extinction’? We present new geochronological data from uranium-bearing secondary minerals and show that they are relatively recent, irrespective of the age of their primary uranium sources. The secondary species that make up much of the diversity of minerals appear to be ephemeral, and many may have vanished from the geological record without trace. Nevertheless, an ‘extinct’ mineral species can recur when physiochemical conditions are appropriate. This reversibility of ‘extinction’ highlights the limitations of the ‘evolution’ analogy. Mineral occurrence may be time-dependent but does not show the unique contingency between precursor and successor species that is characteristic of biological evolution.
Feynmanite, a new sodium uranyl sulfate mineral from Red Canyon, San Juan County, Utah, USA
- Anthony R. Kampf, Travis A. Olds, Jakub Plášil, Joe Marty, Samuel N. Perry
-
- Published online by Cambridge University Press:
- 28 May 2018, pp. 153-160
-
- Article
- Export citation
-
The new mineral feynmanite, Na(UO2)(SO4)(OH)·3.5H2O, was found in both the Blue Lizard and Markey mines, San Juan County, Utah, USA, where it occurs as a secondary phase on pyrite-rich asphaltum in association with chinleite-(Y), gypsum, goethite, natrojarosite, natrozippeite, plášilite, shumwayite (Blue Lizard) and wetherillite (Markey). The mineral is pale greenish yellow with a white streak and fluoresces bright greenish white under a 405 nm laser. Crystals are transparent with a vitreous lustre. It is brittle, with a Mohs hardness of ~2, irregular fracture and one perfect cleavage on {010}. The calculated density is 3.324 g cm–3. Crystals are thin needles or blades, flattened on {010} and elongate on [100], exhibiting the forms {010}, {001}, {101} and {10
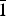 $\bar{1}$}, and are up to ~0.1 mm in length. Feynmanite is optically biaxial (–), with α = 1.534(2), β = 1.561(2) and γ = 1.571(2) (white light); 2Vmeas. = 62(2)°; no dispersion; and optical orientation: X = b, Y ≈ a,Z ≈ c. It is weakly pleochroic: X = colourless, Y = very pale green yellow and Z = pale green yellow (X < Y < Z). Electron microprobe analyses (WDS mode) provided (Na0.84Fe0.01)(U1.01O2)(S1.01O4)(OH)·3.5H2O. The five strongest powder X-ray diffraction lines are [dobs Å(I)(hkl)]: 8.37(100)(010), 6.37(33)(
$\bar{1}$}, and are up to ~0.1 mm in length. Feynmanite is optically biaxial (–), with α = 1.534(2), β = 1.561(2) and γ = 1.571(2) (white light); 2Vmeas. = 62(2)°; no dispersion; and optical orientation: X = b, Y ≈ a,Z ≈ c. It is weakly pleochroic: X = colourless, Y = very pale green yellow and Z = pale green yellow (X < Y < Z). Electron microprobe analyses (WDS mode) provided (Na0.84Fe0.01)(U1.01O2)(S1.01O4)(OH)·3.5H2O. The five strongest powder X-ray diffraction lines are [dobs Å(I)(hkl)]: 8.37(100)(010), 6.37(33)(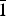 $\bar{1}$01,101), 5.07(27)(
$\bar{1}$01,101), 5.07(27)(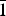 $\bar{1}$11,111), 4.053(46)(004,021) and 3.578(34)(120). Feynmanite is monoclinic, has space group P2/n, a = 6.927(3), b = 8.355(4), c = 16.210(7) Å, β = 90.543(4)°, V = 938.1(7) Å3 and Z = 4. The structure of feynmanite (R1 = 0.0371 for 1879 Io > 2σI) contains edge-sharing pairs of pentagonal bipyramids that are linked by sharing corners with SO4 groups, yielding a [(UO2)2(SO4)2(OH)2]2– sheet based on the phosphuranylite anion topology. The sheet is topologically identical to those in deliensite, johannite and plášilite. The dehydration of feynmanite to plášilite results in interlayer collapse involving geometric reconfiguration of the sheets and the ordering of Na.
$\bar{1}$11,111), 4.053(46)(004,021) and 3.578(34)(120). Feynmanite is monoclinic, has space group P2/n, a = 6.927(3), b = 8.355(4), c = 16.210(7) Å, β = 90.543(4)°, V = 938.1(7) Å3 and Z = 4. The structure of feynmanite (R1 = 0.0371 for 1879 Io > 2σI) contains edge-sharing pairs of pentagonal bipyramids that are linked by sharing corners with SO4 groups, yielding a [(UO2)2(SO4)2(OH)2]2– sheet based on the phosphuranylite anion topology. The sheet is topologically identical to those in deliensite, johannite and plášilite. The dehydration of feynmanite to plášilite results in interlayer collapse involving geometric reconfiguration of the sheets and the ordering of Na.
Editorial
A new layout and update for Mineralogical Magazine in 2019
- Stuart J. Mills, Roger H. Mitchell, Helen C. Kerbey
-
- Published online by Cambridge University Press:
- 11 March 2019, pp. 1-2
-
- Article
-
- You have access Access
- HTML
- Export citation
The 51st Hallimond Lecture
Time's arrow, time's cycle: Granulite metamorphism and geodynamics
- Michael Brown, Tim Johnson
-
- Published online by Cambridge University Press:
- 12 April 2019, pp. 323-338
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Although the thermal evolution of the mantle before c. 3.0 Ga remains unclear, since c. 3.0 Ga secular cooling has dominated over heat production—this is time's arrow. By contrast, the thermal history of the crust, which is preserved in the record of metamorphism, is more complex. Heat to drive metamorphism is generated by radioactive decay and viscous dissipation, and is augmented by the influx of heat from the mantle. Notwithstanding that reliable data are sparse before the Neoarchean, we use a dataset of temperature (T), pressure (P) and thermobaric ratio (T/P at the metamorphic ‘peak’), and age of metamorphism (t, the timing of the metamorphic ‘peak’) for rocks from 564 localities ranging in age from the Cenozoic to the Eoarchean eras to interrogate the crustal record of metamorphism as a proxy for the heat budget of the crust through time. On the basis of T/P, metamorphic rocks are classified into three natural groups: high T/P type (T/P >775°C/GPa, mean T/P ~1105°C/GPa), including common and ultrahigh-temperature granulites, intermediate T/P type (T/P between 775 and 375°C/GPa, mean T/P ~575°C/GPa), including high-pressure granulites and medium- and high-temperature eclogites, and low T/P type (T/P <375°C/GPa, mean T/P ~255°C/GPa), including blueschists, low-temperature eclogites and ultrahigh-pressure metamorphic rocks. A monotonic increase in the P of intermediate T/P metamorphism from the Neoarchean to the Neoproterozoic reflects strengthening of the lithosphere during secular cooling of the mantle—this is also time's arrow. However, temporal variation in the P of intermediate T/P metamorphism and in the moving means of T and T/P of high T/P metamorphism, combined with the clustered age distribution, demonstrate the cyclicity of collisional orogenesis and cyclic variations in the heat budget of the crust superimposed on secular cooling since c. 3.0 Ga—this is time's cycle. A first cycle began with the widespread appearance/survival of intermediate T/P and high T/P metamorphism in the Neoarchean rock record coeval with amalgamation of dispersed blocks of lithosphere to form protocontinents. This cycle was terminated by the fragmentation of the protocontinents into cratons in the early Paleoproterozoic, which signalled the start of a new cycle. The second cycle continued with the progressive amalgamation of the cratons into the supercontinent Columbia and extended until the breakup of the supercontinent Rodinia in the Neoproterozoic. This cycle represented a period of relative tectonic and environmental stability, and perhaps reduced subduction during at least part of the cycle. During most of the Proterozoic the moving means for both T and T/P of high T/P metamorphism exceeded the arithmetic means, reflecting insulation of the mantle beneath the quasi-integrated lithosphere of Columbia and, after a limited reorganisation, Rodinia. The third cycle began with the steep decline in thermobaric ratios of high T/P metamorphism to their lowest value, synchronous with the breakup of Rodinia and the formation of Pannotia, and the widespread appearance/preservation of low T/P metamorphism in the rock record. The thermobaric ratios for high T/P metamorphism rise to another peak associated with the Pan-African event, again reflecting insulation of the mantle. The subsequent steep decline in thermobaric ratios of high T/P metamorphism associated with the breakup of Pangea at c. 0.175 Ga may indicate the start of a fourth cycle. The limited occurrence of high and intermediate T/P metamorphism before the Neoarchean suggests either that suitable tectonic environments to generate these types of metamorphism were not widely available before then or that the rate of survival was low. We interpret the first cycle to record stabilisation of subduction and the emergence of a network of plate boundaries in a plate tectonics regime once the balance between heat production and heat loss changed in favour of secular cooling, possibly as early as c. 3.0 Ga in some areas. This is inferred to have been a globally linked system by the early Paleoproterozoic, but whether it remained continuous to the present is unclear. The second cycle was characterised by stability from the formation of Columbia to the breakup of Rodinia, generating higher than average T and T/P of high T/P metamorphism. The third cycle reflects colder collisional orogenesis and deep subduction of the continental crust, features that are characteristic of modern plate tectonics, which became possible once the average temperature of the asthenospheric mantle had declined to <100°C warmer than the present day after c. 1.0 Ga.
Article
New arsenate minerals from the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. X. Edtollite, K2NaCu5Fe3+O2(AsO4)4, and alumoedtollite, K2NaCu5AlO2(AsO4)4
- Igor V. Pekov, Natalia V. Zubkova, Atali A. Agakhanov, Dmitry A. Ksenofontov, Leonid A. Pautov, Evgeny G. Sidorov, Sergey N. Britvin, Marina F. Vigasina, Dmitry Y. Pushcharovsky
-
- Published online by Cambridge University Press:
- 02 October 2018, pp. 485-495
-
- Article
- Export citation
-
Two new isostructural minerals edtollite K2NaCu5Fe3+O2(AsO4)4 and alumoedtollite K2NaCu5AlO2(AsO4)4 have been found in the Arsenatnaya fumarole, Second scoria cone of the Northern Breakthrough of the Great Tolbachik Fissure Eruption, Tolbachik volcano, Kamchatka, Russia. They are associated with sylvite, tenorite, dmisokolovite, shchurovskyite, johillerite, bradaczekite, and orthoclase. Edtollite forms prismatic crystals up to 0.02 mm × 0.1 mm; alumoedtollite forms long-prismatic crystals up to 0.01 mm × 0.1 mm. Both minerals have a semi-metallic lustre. Edtollite is brown–black to black and alumoedtollite is bronze coloured. Dcalc. = 4.26 (edtollite) and 4.28 (alumoedtollite) g cm–3. In reflected light, both minerals are grey, with distinct anisotropy. Reflectance values [edtollite/alumoedtollite: R1–R2, % (λ, nm)] are: 8.3–8.2/8.7–7.7 (470); 7.7–7.4/8.3–7.4 (546); 7.1–6.9/8.3–7.4 (589); and 6.3–6.3/7.6–7.2 (650). Chemical data are: (edtollite/alumoedtollite, wt.%, electron-microprobe): Na2O 3.13/2.58, K2O 8.12/9.09, Rb2O 0.00/0.11, CaO 0.00/0.52, CuO 36.55/38.35, ZnO 0.46/0.00, Al2O3 0.00/3.48, Fe2O3 7.34/1.79, TiO2 0.27/0.00, As2O5 43.57/43.66, total 99.44/99.58. The empirical formulae, based on 18 O apfu, for edtollite is: K1.83Na1.07Cu4.88Zn0.06Fe3+0.98Ti0.04As4.03O18; and for alumoedtollite is: K2.02Rb0.01Na0.87Ca0.10Cu5.06Al0.72Fe3+0.24As3.99O18. Both minerals are triclinic, P
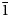 $\bar{1}$; unit-cell parameters (edtollite/alumoedtollite) are: a = 5.1168(6)/5.0904(11), b = 9.1241(12)/9.0778(14), c = 9.6979(14)/9.6658(2) Å, α = 110.117(13)/110.334(17), β = 102.454(12)/102.461(19), γ = 92.852(11)/92.788(15)°, V = 411.32(9)/404.88(14) Å3 and Z = 1/1. The strongest reflections in the powder X-ray diffraction pattern [d,Å(I)(hkl)] are for edtollite: 8.79(92)(001), 7.63(41)(0
$\bar{1}$; unit-cell parameters (edtollite/alumoedtollite) are: a = 5.1168(6)/5.0904(11), b = 9.1241(12)/9.0778(14), c = 9.6979(14)/9.6658(2) Å, α = 110.117(13)/110.334(17), β = 102.454(12)/102.461(19), γ = 92.852(11)/92.788(15)°, V = 411.32(9)/404.88(14) Å3 and Z = 1/1. The strongest reflections in the powder X-ray diffraction pattern [d,Å(I)(hkl)] are for edtollite: 8.79(92)(001), 7.63(41)(0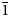 $\bar{1}$1), 5.22(44)(011), 3.427(100)(012), 3.148(64)(0
$\bar{1}$1), 5.22(44)(011), 3.427(100)(012), 3.148(64)(0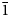 $\bar{1}$3), 2.851(65)(
$\bar{1}$3), 2.851(65)(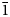 $\bar{1}$03) and 2.551(40)(
$\bar{1}$03) and 2.551(40)(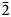 $\bar{2}$01); and for alumoedtollite: 8.78(81)(001), 7.62(67)(0
$\bar{2}$01); and for alumoedtollite: 8.78(81)(001), 7.62(67)(0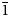 $\bar{1}$1), 3.418(100)(012), 3.147(52)(0
$\bar{1}$1), 3.418(100)(012), 3.147(52)(0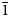 $\bar{1}$3), 2.558(58)(
$\bar{1}$3), 2.558(58)(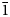 $\bar{1}$22), 2.544(65)(
$\bar{1}$22), 2.544(65)(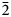 $\bar{2}$01) and 2.528(52)(
$\bar{2}$01) and 2.528(52)(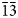 $\bar{1}\bar{3}$2). The crystal structures [single-crystal X-ray diffraction, R = 0.0773 (edtollite) and 0.0826 (alumoedtollite); 1504 and 1046 unique reflections, respectively] represent a novel structure type. It is based upon a heteropolyhedral pseudo-framework with the column formed by Cu2+-centred octahedra and square pyramids, octahedra MO6 (M = Fe3+, Al3+ or Cu2+) and AsO4 tetrahedra as the main building unit. K+ and Na+ are located in wide and narrow channels, respectively. Edtollite is named after the Russian geologist and Arctic explorer Eduard Vasilievich Toll (1858–1902), alumoedtollite is its analogue with Al prevailing among trivalent cations.
$\bar{1}\bar{3}$2). The crystal structures [single-crystal X-ray diffraction, R = 0.0773 (edtollite) and 0.0826 (alumoedtollite); 1504 and 1046 unique reflections, respectively] represent a novel structure type. It is based upon a heteropolyhedral pseudo-framework with the column formed by Cu2+-centred octahedra and square pyramids, octahedra MO6 (M = Fe3+, Al3+ or Cu2+) and AsO4 tetrahedra as the main building unit. K+ and Na+ are located in wide and narrow channels, respectively. Edtollite is named after the Russian geologist and Arctic explorer Eduard Vasilievich Toll (1858–1902), alumoedtollite is its analogue with Al prevailing among trivalent cations.
On the application of the IMA−CNMNC dominant-valency rule to complex mineral compositions
- Ferdinando Bosi, Frédéric Hatert, Ulf Hålenius, Marco Pasero, Ritsuro Miyawaki, Stuart J. Mills
-
- Published online by Cambridge University Press:
- 23 August 2019, pp. 627-632
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Mineral species should be identified by an end-member formula and by using the dominant-valency rule as recommended by the IMA–CNMNC. However, the dominant-end-member approach has also been used in the literature. These two approaches generally converge, but for some intermediate compositions, significant differences between the dominant-valency rule and the dominant end-member approach can be observed. As demonstrated for garnet-supergroup minerals, for example, the end-member approach is ambiguous, as end-member proportions strongly depend on the calculation sequence. For this reason, the IMA–CNMNC strongly recommends the use of the dominant-valency rule for mineral nomenclature, because it alone may lead to unambiguous mineral identification. Although the simple application of the dominant-valency rule is successful for the identification of many mineral compositions, sometimes it leads to unbalanced end-member formulae, due to the occurrence of a coupled heterovalent substitution at two sites along with a heterovalent substitution at a single site. In these cases, it may be useful to use the site-total-charge approach to identify the dominant root-charge arrangement on which to apply the dominant-constituent rule. The dominant-valency rule and the site-total-charge approach may be considered two procedures complementary to each other for mineral identification. Their critical point is to find the most appropriate root-charge and atomic arrangements consistent with the overriding condition dictated by the end-member formula. These procedures were approved by the IMA−CNMNC in May 2019.
Constraints on the Equations of State of stiff anisotropic minerals: rutile, and the implications for rutile elastic barometry
- Gabriele Zaffiro, Ross J. Angel, Matteo Alvaro
-
- Published online by Cambridge University Press:
- 22 April 2019, pp. 339-347
-
- Article
- Export citation
-
We present an assessment of the thermo-elastic behaviour of rutile based on X-ray diffraction data and direct elastic measurements available in the literature. The data confirms that the quasi-harmonic approximation is not valid for rutile because rutile exhibits substantial anisotropic thermal pressure, meaning that the unit-cell parameters change significantly along isochors. Simultaneous fitting of both the diffraction and elasticity data yields parameters of KTR0= 205.14(15) GPa, KSR0= 207.30(14) GPa,
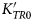 $K_{TR0}^{\prime} $= 6.9(4) in a 3rd-order Birch-Murnaghan Equation of State for compression, αV0= 2.526(16) × 10–5 K–1, Einstein temperature θE = 328(12) K, Anderson-Grüneisen parameter δT = 7.6(6), with a fixed thermal Grüneisen parameter γ = 1.4 to describe the thermal expansion and variation of bulk modulus with temperature at room pressure. This Equation of State fits all of the available data up to 7.3 GPa at room temperature, and up to 1100 K at room pressure within its uncertainties. We also present a series of formulations and a simple protocol to obtain thermodynamically consistent Equations of State for the volume and the unit-cell parameters for stiff materials, such as rutile. In combination with published data for garnets, the Equation of State for rutile indicates that rutile inclusions trapped inside garnets in metamorphic rocks should exhibit negative residual pressures when measured at room conditions.
$K_{TR0}^{\prime} $= 6.9(4) in a 3rd-order Birch-Murnaghan Equation of State for compression, αV0= 2.526(16) × 10–5 K–1, Einstein temperature θE = 328(12) K, Anderson-Grüneisen parameter δT = 7.6(6), with a fixed thermal Grüneisen parameter γ = 1.4 to describe the thermal expansion and variation of bulk modulus with temperature at room pressure. This Equation of State fits all of the available data up to 7.3 GPa at room temperature, and up to 1100 K at room pressure within its uncertainties. We also present a series of formulations and a simple protocol to obtain thermodynamically consistent Equations of State for the volume and the unit-cell parameters for stiff materials, such as rutile. In combination with published data for garnets, the Equation of State for rutile indicates that rutile inclusions trapped inside garnets in metamorphic rocks should exhibit negative residual pressures when measured at room conditions.
Distribution of Cr3+ between octahedral and tetrahedral sites in synthetic blue and green (CaMgSi2O6)95(CaCrAlSiO6)5 diopsides
- Masahide Akasaka, Yohei Takasu, Makoto Handa, Mariko Nagashima, Maki Hamada, Terumi Ejima
-
- Published online by Cambridge University Press:
- 12 February 2019, pp. 497-505
-
- Article
- Export citation
-
The distribution of Cr3+ ions in blue and green diopsides crystallised from a glass with the composition [CaMgSi2O6 (Di)]95[CaCrAlSiO6 (CrAlTs)]5 (mol.%) was determined using Rietveld refinement of X-ray diffraction data in order to evaluate published results by optical spectroscopic analysis, and to clarify the influence of Cr3+–Al3+ distribution between the octahedral M1 and tetrahedral T sites on the crystal structure. The starting material was Di95CrAlTs5-diopside crystallised from glass at 800°C for 2 days. After another 19 days at 800°C and 1000°C for 7 days, the diopsides remained blue. The blue diopside gradually changed to bluish green by heating at 1200°C for 3 days and to green after 7 days. The stoichiometric compositions of the synthesised phases were confirmed by electron microprobe analysis. The Cr occupancies refined by the Rietveld method resulted in the site populations in the M1 and T sites: M1[Mg0.95Cr0.030(4)Al0.020]T[Si1.950Cr0.020Al0.030] and M1[Mg0.95Cr0.037(4)Al0.013]T[Si1.950Cr0.013Al0.037] (per 6 oxygens) for the blue diopsides at 800 and 1000°C, respectively: M1[Mg0.95Cr0.042(3)Al0.008]T[Si1.950Cr0.008Al0.042] for the bluish green diopside at 1200°C for 3 days; and M1[Mg0.95Cr0.049(3)Al0.001]T[Si1.950Cr0.001Al0.049] for the green diopside at 1200°C for 7 days. Such Cr and Al distributions effect the volumes and site distortions of the octahedral and tetrahedral coordination polyhedra: the TO4 tetrahedron volumes of the blue diopsides (2.251–2.258 Å3) are larger than that of the green diopside (2.237 Å3); the M1O6 octahedron volumes of the former (11.74–11.77 Å3) are smaller than that of the latter (11.86 Å3); the TO4 tetrahedra in the blue diopside (<λtet> =1.006; σθ(tet)2 = 24.37–24.69) are less distorted than that of the green diopside (<λtet> = 1.007; σθ(tet)2 = 27.94); the M1O6 octahedra in the former (<λoct> =1.006; σθ(oct)2 = 20.39–21.13) are more distorted than that of the latter (<λoct> = 1.005; σθ(oct)2 = 17.58).
The geochemical evolution of Nb–Ta–Sn oxides from pegmatites of the Cape Cross–Uis pegmatite belt, Namibia
- Warrick C. Fuchsloch, Paul A. M. Nex, Judith A. Kinnaird
-
- Published online by Cambridge University Press:
- 02 October 2018, pp. 161-179
-
- Article
- Export citation
-
The Cape Cross–Uis pegmatite belt, Damara Orogen, north-central Namibia hosts multiple Ta–Nb- and Sn-oxide-bearing pegmatites. Columbite-group minerals, tapiolite, cassiterite and minor ixiolite and wodginite occur in abundance within pegmatites and display various compositional and internal structural mineralogical variations. Ta–Nb oxides display various zonation patterns indicative of multiple crystallisation phases, whereas cassiterite is dominantly homogeneous with minor euhedral columbite-group mineral inclusions. Ta–Nb oxides are mostly rich in Fe, with fractionation patterns in the columbite quadrilateral being sub parallel to the Ta/(Ta + Nb) axis; increasing Ta/(Ta + Nb) with little change in Mn/(Mn + Fe), which is consistent with classical trends in beryl-to-spodumene rare-element pegmatites. In addition, these trends suggest that co-crystallising minerals compete with Ta–Nb oxides for elements such as Mn, preventing Ta–Nb oxides from attaining Mn-rich compositions during the fractionation process. Cassiterite shows similar fractionation patterns with Fe > Mn and notable increases in the Ta content. Minor-element substitution in Ta–Nb oxides shows sharp decreases with increasing fractionation supporting the hypothesis that newly stabilised co-occurring minerals compete with columbite-group minerals for certain elements. Tapiolite shows the same minor-element trend, however, only for Sn and Ti suggesting cassiterite was a dominant competing mineral. Although crystallisation of Ta–Nb oxides from an aqueous fluid at the late-stages of pegmatite genesis is highly debated, significantly elevated Ta contents in metasomatised country rock, compared to unaltered country rock, may give new insight, suggesting that Ta may indeed partition into, and be transported by, an exsolved aqueous fluid. However, further studies of the country rock metasomatic contacts are required as currently the dataset is limited. The degree of fractionation as depicted by Ta–Nb and Sn oxides within pegmatites, indicate that a zonation from primitive to evolved pegmatites surrounding granites is not present and that pegmatites are probably not related to granites in the typical parent–daughter relationship.
Antofagastaite, Na2Ca(SO4)2·1.5H2O, a new mineral related to syngenite
- Igor V. Pekov, Vadim M. Kovrugin, Oleg I. Siidra, Nikita V. Chukanov, Dmitry I. Belakovskiy, Natalia N. Koshlyakova, Vasiliy O. Yapaskurt, Anna G. Turchkova, Gerhard Möhn
-
- Published online by Cambridge University Press:
- 12 April 2019, pp. 781-790
-
- Article
- Export citation
-
The new mineral antofagastaite, ideally Na2Ca(SO4)2·1.5H2O, was found in the oxidation zone of sulfide–quartz veins at the abandoned Coronel Manuel Rodríguez mine, Mejillones, Antofagasta Province, Antofagasta Region, Chile. It is associated with sideronatrite, metasideronatrite, aubertite, gypsum, ferrinatrite, glauberite, amarillite and an unidentified Fe phosphate. Antofagastaite occurs as prismatic crystals up to 0.5 mm × 1 mm × 5 mm, elongated along [010], typically combined in open-work aggregates up to 1 cm across. Antofagastaite is transparent and colourless, with vitreous lustre. It is brittle; the Mohs’ hardness is ca 3. Cleavage is distinct on (001). Dmeas. is 2.42(1) and Dcalc. is 2.465 g cm−3. Antofagastaite is optically biaxial (–), α = 1.489(2), β = 1.508(2), γ = 1.510(2) and 2Vmeas. = 40(10)°. The IR spectrum is reported. Chemical composition (wt.%, electron microprobe, H2O determined by gas chromatography) is: Na2O 20.85, CaO 17.42, SO3 52.56, H2O 7.93, total 98.76. The empirical formula (based on 8 O atoms belonging to sulfate anions per formula unit with all H belonging to H2O molecules) is Na2.06Ca0.95S2.01O8·1.35H2O. Antofagastaite is monoclinic, P21/m, a = 6.4596(4), b = 6.8703(5), c = 9.4685(7) Å, β = 104.580(4)°, V = 406.67(5) Å3 and Z = 2. The strongest reflections of the powder XRD pattern [d, Å (I, %) (hkl)] are: 9.17 (100) (001), 5.501 (57) (011), 3.437 (59) (020), 3.058 (43) (003), 2.918 (50) (2¯11), 2.795 (35) (013) and 2.753 (50) (121, 201). The crystal structure was solved based on single-crystal X-ray diffraction data, R1 = 5.71%. The structure of antofagastaite consists of ordered and disordered blocks and is related to syngenite K2Ca(SO4)2·H2O. Incorporation of additional H2O molecules in the syngenite-type structure results in disorder of the one of the two tetrahedral sulfate groups occurring in antofagastaite. In addition to the above-reported type material, antofagastaite together with syngenite and blödite occurs in the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia.
Special category: Foundations in mineralogy and crystallography
A structure hierarchy for silicate minerals: sheet silicates
- Part of:
- Frank C. Hawthorne, Yulia A. Uvarova, Elena Sokolova
-
- Published online by Cambridge University Press:
- 09 November 2018, pp. 3-55
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The structure hierarchy hypothesis states that structures may be ordered hierarchically according to the polymerisation of coordination polyhedra of higher bond-valence. A hierarchical structural classification is developed for sheet-silicate minerals based on the connectedness of the two-dimensional polymerisations of (TO4) tetrahedra, where T = Si4+ plus As5+, Al3+, Fe3+, B3+, Be2+, Zn2+ and Mg2+. Two-dimensional nets and oikodoméic operations are used to generate the silicate (sensu lato) structural units of single-layer, double-layer and higher-layer sheet-silicate minerals, and the interstitial complexes (cation identity, coordination number and ligancy, and the types and amounts of interstitial (H2O) groups) are recorded. Key aspects of the silicate structural unit include: (1) the type of plane net on which the sheet (or parent sheet) is based; (2) the u (up) and d (down) directions of the constituent tetrahedra relative to the plane of the sheet; (3) the planar or folded nature of the sheet; (4) the layer multiplicity of the sheet (single, double or higher); and (5) the details of the oikodoméic operations for multiple-layer sheets. Simple 3-connected plane nets (such as 63, 4.82 and 4.6.12) have the stoichiometry (T2O5)n (Si:O = 1:2.5) and are the basis of most of the common rock-forming sheet-silicate minerals as well as many less-common species. Oikodoméic operations, e.g. insertion of 2- or 4-connected vertices into 3-connected plane nets, formation of double-layer sheet-structures by (topological) reflection or rotation operations, affect the connectedness of the resulting sheets and lead to both positive and negative deviations from Si:O = 1:2.5 stoichiometry. Following description of the structural units in all sheet-silicate minerals, the minerals are arranged into decreasing Si:O ratio from 3.0 to 2.0, an arrangement that reflects their increasing structural connectivity. Considering the silicate component of minerals, the range of composition of the sheet silicates completely overlaps the compositional ranges of framework silicates and most of the chain-ribbon-tube silicates.
Article
Schmidite and wildenauerite, two new schoonerite-group minerals from the Hagendorf-Süd pegmatite, Oberpfalz, Bavaria
- Ian E. Grey, Erich Keck, Anthony R. Kampf, John D. Cashion, Colin M. MacRae, Alexander M. Glenn, Yesim Gozukara
-
- Published online by Cambridge University Press:
- 29 June 2018, pp. 181-190
-
- Article
- Export citation
-
Schmidite, Zn(Fe3+0.5Mn2+0.5)2ZnFe3+(PO4)3(OH)3(H2O)8 and wildenauerite, Zn(Fe3+0.5Mn2+0.5)2Mn2+Fe3+(PO4)3(OH)3(H2O)8 are two new oxidised schoonerite-group minerals from the Hagendorf-Süd pegmatite, Hagendorf, Oberpfalz, Bavaria, Germany. Schmidite occurs as radiating sprays of orange–brown to copper-red laths on and near to altered phosphophyllite in a corroded triphylite nodule, whereas wildenauerite forms dense compacts of red laths, terminating Zn-bearing rockbridgeite. The minerals are biaxial (+) with α = 1.642(2), β = 1.680(1), γ = 1.735(2) and 2Vmeas = 81.4(8)° for schmidite, and with α = 1.659(3), β = 1.687(3), γ = 1.742(3) and 2Vmeas = 73(1)° for wildenauerite. Electron microprobe analyses, with H2O from thermal analysis and FeO/Fe2O3 from Mössbauer spectroscopy, gave FeO 0.4, MgO 0.3, Fe2O3 23.5, MnO 9.0, ZnO 15.5, P2O5 27.6, H2O 23.3, total 99.6 wt.% for schmidite, and FeO 0.7, MgO 0.3, Fe2O3 25.2, MnO 10.7, ZnO 11.5, P2O5 27.2, H2O 24.5, total 100.1 wt.% for wildenauerite. The empirical formulae, scaled to 3 P and with OH– adjusted for charge balance are Zn1.47Mn2+0.98Mg0.05Fe2+0.04Fe3+2.27(PO4)3(OH)2.89(H2O)8.54 for schmidite and Zn1.11Mn2+1.18Mg0.05Fe2+0.08Fe3+2.47(PO4)3(OH)3.25(H2O)9.03 for wildenauerite. The two minerals have orthorhombic symmetry, space group Pmab and Z = 4. The unit-cell parameters from refinement of powder X-ray diffraction data are a = 11.059(1), b = 25.452(1) and c = 6.427(1) Å for schmidite, and a = 11.082(1), b = 25.498(2) and c = 6.436(1) Å for wildenauerite. The crystal structures of schmidite and wildenauerite differ from that of schoonerite in having minor partitioning of Zn from the [5]Zn site to an adjacent vacant tetrahedral site [4]Zn, separated by ~1.0 Å from [5]Zn. The two minerals are distinguished by the cation occupancies in the octahedral M1 to M3 sites. Schmidite has M1 = M2 = (Fe3+0.5Mn2+0.5) and M3 = Zn and wildenauerite has M1 = M2 = (Fe3+0.5Mn2+0.5) and M3 = Mn2+.
Magnesioleydetite and straβmannite, two new uranyl sulfate minerals with sheet structures from Red Canyon, Utah
- Anthony R. Kampf, Jakub Plášil, Anatoly V. Kasatkin, Barbara P. Nash, Joe Marty
-
- Published online by Cambridge University Press:
- 28 May 2018, pp. 349-360
-
- Article
- Export citation
-
Magnesioleydetite (IMA2017-063), Mg(UO2)(SO4)2·11H2O, and straβmannite (IMA2017-086), Al(UO2)(SO4)2F·16H2O, are two new minerals from mines in Red Canyon, San Juan County, Utah, USA. Magnesioleydetite occurs in the Markey mine and straβmannite occurs in both the Markey and Green Lizard mines. Both minerals are secondary phases found in efflorescent crusts on the surfaces of mine walls. Magnesioleydetite occurs in irregular aggregates (to ~0.5 mm) of blades (to ~0.2 mm) exhibiting the following properties: transparent to translucent; pale green–yellow colour; vitreous lustre; white streak; non-fluorescent; brittle; Mohs hardness ≈ 2; irregular fracture; one perfect cleavage on {001}; and calculated density = 2.463 g/cm3. Straβmannite occurs in irregular aggregates (to ~0.5 mm) of equant crystals (to ~0.2 mm) exhibiting the following properties: transparent; light yellow–green colour; vitreous to greasy lustre; nearly white streak; bright greenish-blue fluorescence; somewhat brittle, Mohs hardness ≈ 1½; irregular fracture; one good cleavage on {001}; measured and calculated densities of 2.20(2) and 2.173 g/cm3, respectively; optically biaxial (–); α = 1.477(2), β = 1.485(2) and γ = 1.489(2) (white light); 2Vmeas. = 72(2)°; dispersion r > v (slight); orientation Y = b, X ∧ c = 20° (in obtuse β); pleochroism with X = nearly colourless, Y = pale green–yellow and Z = light green–yellow (X < Y < Z). The empirical formulas for magnesioleydetite and straβmannite are (Mg0.56Fe0.26Zn0.11Mn0.01)Σ0.94(U0.99O2)(S1.015O4)2·11H2O and Al1.00Na0.16(U0.99O2)(S1.00O4)2[F0.58(OH)0.42]·16H2O, respectively. Magnesioleydetite is monoclinic, C2/c, a = 11.3513(3), b = 7.7310(2), c = 21.7957(15) Å, β = 102.387(7)°, V = 1868.19(16) Å3 and Z = 4. Straβmannite is monoclinic, C2/c, a = 11.0187(5), b = 8.3284(3), c = 26.6727(19) Å, β = 97.426(7)°, V = 2427.2(2) and Z = 4. The structures of magnesioleydetite (R1 = 0.016 for 2040 I > 2σI reflections) and straβmannite (R1 = 0.0343 for 2220 I > 2σI reflections) each contain uranyl-sulfate sheets based on the protasite-anion topology.
New arsenate minerals from the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. XI. Anatolyite, Na6(Ca,Na)(Mg,Fe3+)3Al(AsO4)6
- Igor V. Pekov, Inna S. Lykova, Vasiliy O. Yapaskurt, Dmitry I. Belakovskiy, Anna G. Turchkova, Sergey N. Britvin, Evgeny G. Sidorov, Katharina S. Scheidl
-
- Published online by Cambridge University Press:
- 22 February 2019, pp. 633-638
-
- Article
- Export citation
-
The new mineral anatolyite Na6(Ca,Na)(Mg,Fe3+)3Al(AsO4)6 was found in the Arsenatnaya fumarole, Tolbachik volcano, Kamchatka, Russia. It is associated with potassic feldspar, hematite, tenorite, cassiterite, johillerite, tilasite, ericlaxmanite, lammerite, arsmirandite, sylvite, halite, aphthitalite, langbeinite, anhydrite, wulffite, krasheninnikovite, fluoborite, pseudobrookite and fluorophlogopite. Anatolyite occurs as aggregates (up to 2 mm across) of rhombohedral–prismatic, equant or slightly elongated along [001] crystals up to 0.2 mm. The mineral is transparent, pale brownish–pinkish, with vitreous lustre. It is brittle, cleavage was not observed and the fracture is uneven. The Mohs’ hardness is ca 4½. Dcalc is 3.872 g cm–3. Anatolyite is optically uniaxial (–), ω = 1.703(4) and ε = 1.675(3). Chemical composition (wt.%, electron microprobe) is: Na2O 16.55, K2O 0.43, CaO 2.49, MgO 5.80, MnO 0.16, CuO 0.69, ZnO 0.55, Al2O3 5.01, Fe2O3 7.94, TiO2 0.18, SnO2 0.17, SiO2 0.04, P2O5 0.55, As2O5 60.75, SO3 0.03, total 101.34. The empirical formula based on 24 O apfu is (Na5.90K0.10)Σ6.00(Ca0.50Na0.13Zn0.08Mn0.03)Σ0.74(Mg1.63Fe3+1.12Al0.15Cu0.10)Σ3.00(Al0.96Ti0.03Sn0.01)Σ1.00(As5.97P0.09Si0.01)Σ6.07O24. Anatolyite is trigonal, R
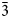 $\bar{3}$c, a = 13.6574(10), c = 18.2349(17) Å, V = 2945.6(4) Å3 and Z = 6. The strongest reflections of the powder XRD pattern [d,Å(I)(hkl)] are: 7.21(33)(012), 4.539(16)(113), 4.347(27)(211), 3.421(20)(220), 3.196(31)(214), 2.981(17)(223), 2.827(100)(125) and 2.589(18)(410). The crystal structure was solved from single-crystal XRD data to R = 4.77%. The structure is based on a 3D heteropolyhedral framework formed by M4O18 clusters [M1 = Al and M2 = (Mg,Fe3+)] linked with AsO4 tetrahedra. (Ca,Na) and Na cations centre A1O6 and A2O8 polyhedra in voids of the framework. Anatolyite is isostructural with yurmarinite. The new mineral is named in honour of the outstanding Russian crystallographer, mineralogist and mathematician Anatoly Kapitonovich Boldyrev (1883–1946).
$\bar{3}$c, a = 13.6574(10), c = 18.2349(17) Å, V = 2945.6(4) Å3 and Z = 6. The strongest reflections of the powder XRD pattern [d,Å(I)(hkl)] are: 7.21(33)(012), 4.539(16)(113), 4.347(27)(211), 3.421(20)(220), 3.196(31)(214), 2.981(17)(223), 2.827(100)(125) and 2.589(18)(410). The crystal structure was solved from single-crystal XRD data to R = 4.77%. The structure is based on a 3D heteropolyhedral framework formed by M4O18 clusters [M1 = Al and M2 = (Mg,Fe3+)] linked with AsO4 tetrahedra. (Ca,Na) and Na cations centre A1O6 and A2O8 polyhedra in voids of the framework. Anatolyite is isostructural with yurmarinite. The new mineral is named in honour of the outstanding Russian crystallographer, mineralogist and mathematician Anatoly Kapitonovich Boldyrev (1883–1946).
The discreditation of oboyerite and a note on the crystal structure of plumbotellurite
- Owen P. Missen, Michael S. Rumsey, Anthony R. Kampf, Stuart J. Mills, Malcolm E. Back, John Spratt
-
- Published online by Cambridge University Press:
- 04 October 2019, pp. 791-797
-
- Article
- Export citation
-
The mineral ‘oboyerite’, first described in 1979 from the Grand Central mine, Tombstone, Cochise County, Arizona, USA, has been re-examined. The type specimen from the Natural History Museum, London and a specimen from the Natural History Museum of Los Angeles County (traceable to S. A Williams, who first described ‘oboyerite’) were analysed in this study. The discreditation of ‘oboyerite’ as a valid mineral species has been approved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (Proposal 19-D). Single-crystal X-ray diffraction, powder X-ray diffraction, electron probe microanalysis and scanning electron microscopy were all employed to show that ‘oboyerite’ is formed of at least two distinct phases, including the lead–tellurium oxysalt minerals ottoite and plumbotellurite. During the course of the discreditation, plumbotellurite was confirmed to be identical to the synthetic compound α-Pb2+Te4+O3. Previously, in some mineralogical literature plumbotellurite was described as orthorhombic with no known crystal structure.
Quantitative Raman calibration of sulfate-bearing polymineralic mixtures: a S quantification in sedimentary rocks on Mars
- Chloé Larre, Yann Morizet, Catherine Guillot-Deudon, Fabien Baron, Nicolas Mangold
-
- Published online by Cambridge University Press:
- 14 September 2018, pp. 57-69
-
- Article
- Export citation
-
The NASA 2020 Mars mission is a Curiosity-type rover whose objective is to improve the knowledge of the geological and climatic evolution of Mars and to collect rock samples for return to Earth. The new rover has a payload of seven instruments including the SuperCam instrument which consists of four tools including a Raman spectrometer. This Raman device will be non-destructive and will analyse the surface remotely in order to determine the mineralogy of rocks and, by extent, to detect and quantify major elements such as sulfur. Sulfur has been detected as sulfate (Ca,Mg,Fe-sulfates) in sedimentary rocks. This element is difficult to quantify using the laser ablation tool of the ChemCam instrument on-board the Curiosity rover.
We propose a Raman calibration to constrain the sulfur abundance in polymineralic mixtures. We acquired Raman signatures on binary and ternary mechanical mixtures containing Ca and Mg sulfates, mixed with natural silicate minerals supposed to be relevant to basaltic-sedimentary rocks at the surface of Mars: olivine, clinopyroxene, orthopyroxene and plagioclase. Using the Voigt function to process the Raman spectra from samples extracted from our mixtures allows us to calculate the initial proportions of our preparations of Ca and Mg sulfates. From these simulations, calibration equations have been provided allowing us to determine sulfate proportions (CaSO4 and MgSO4) in a mixture with basaltic minerals. With the presented calibration, S can be quantified at a lower limit of 0.7 wt.% in Martian soil.
Twinning and incommensurate modulation in baumoite, Ba0.5[(UO2)3O8Mo2(OH)3](H2O)~3, the first natural Ba uranyl molybdate
- Peter Elliott, Jakub Plášil, Václav Petříček, Jiří Čejka, Luca Bindi
-
- Published online by Cambridge University Press:
- 12 April 2019, pp. 507-514
-
- Article
- Export citation
-
Baumoite, Ba0.5[(UO2)3O8Mo2(OH)3](H2O)~3, is a new mineral found near Radium Hill, South Australia, where it occurs in a granite matrix associated with baryte, metatorbernite, phurcalite and kaolinite. Baumoite forms thin crusts of yellow to orange–yellow tabular to prismatic crystals. The mineral is translucent with a vitreous lustre and pale yellow streak. Crystals are brittle, the fracture is uneven and show one excellent cleavage. The Mohs hardness is ~2½. The calculated density is 4.61 g/cm3. Optically, baumoite crystals are biaxial (–), with α = 1.716(4), β = 1.761(4), γ = 1.767(4) (white light); and 2Vcalc = 42.2°. Electron microprobe analyses gave the empirical formula Ba0.87Ca0.03Al0.04U2.97Mo2.02P0.03O22H11.99, based on 22 O atoms per formula unit. The eight strongest lines in the powder X-ray diffraction pattern are [dobs Å (I) (hkl)]: 9.175(39)(12
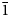 ${\bar 1}$), 7.450(100)(020), 3.554(20)(221), 3.365(31)(004, 202), 3.255(31)(123, 30
${\bar 1}$), 7.450(100)(020), 3.554(20)(221), 3.365(31)(004, 202), 3.255(31)(123, 30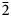 ${\bar 2}$), 3.209(28)(12
${\bar 2}$), 3.209(28)(12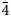 ${\bar 4}$), 3.067(33)(30
${\bar 4}$), 3.067(33)(30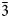 ${\bar 3}$, 222, 32
${\bar 3}$, 222, 32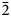 ${\bar 2}$) and 2.977(20)(142). Single-crystal X-ray studies (R1 = 5.85% for 1892 main reflections) indicate that baumoite is monoclinic, superspace group X2/m(a0g)0s with X = (0,½,0,½), with unit-cell parameters: a = 9.8337(3), b = 15.0436(5), c = 14.2055(6) Å, β = 108.978(3)°, V = 1987.25(13) Å3 and Z = 4. The crystal structure is twinned and incommensurately modulated and is based upon sheets of U6+ and Mo6+ polyhedra of unique topology. Four independent cationic sites partially occupied by Ba atoms are located between the sheets, together with H2O molecules.
${\bar 2}$) and 2.977(20)(142). Single-crystal X-ray studies (R1 = 5.85% for 1892 main reflections) indicate that baumoite is monoclinic, superspace group X2/m(a0g)0s with X = (0,½,0,½), with unit-cell parameters: a = 9.8337(3), b = 15.0436(5), c = 14.2055(6) Å, β = 108.978(3)°, V = 1987.25(13) Å3 and Z = 4. The crystal structure is twinned and incommensurately modulated and is based upon sheets of U6+ and Mo6+ polyhedra of unique topology. Four independent cationic sites partially occupied by Ba atoms are located between the sheets, together with H2O molecules.
Gem amphiboles from Mogok, Myanmar: crystal-structure refinement, infrared spectroscopy and short-range order–disorder in gem pargasite and fluoro-pargasite
- Maxwell C. Day, Frank C. Hawthorne, Umberto Susta, Giancarlo Della Ventura, George E. Harlow
-
- Published online by Cambridge University Press:
- 14 September 2018, pp. 361-371
-
- Article
- Export citation
-
The crystal structures of six gem-quality pargasites and fluoro-pargasites from Mogok, Myanmar, space group C2/m, Z = 2, have been refined to R1 indices of 2.20–2.90% using MoKα X-radiation. The unit formulae were calculated from the results of electron-microprobe analysis, and were used with the refined site-scattering values and the observed mean bond lengths to assign site populations. TAl occurs at both the T(1) and T(2) sites but is strongly ordered at T(1). [6]Al is partly disordered over the M(2) and M(3) sites but does not occur at the M(1) site. ANa is split between the A(2) and A(m) sites and K occurs at the A(m) site. The infrared spectra in the principal OH-stretching region were measured and the fine structure was fit to component bands. The component bands were assigned to short-range ion arrangements over the configuration symbol M(1)M(1)M(3)–O(3)–A–O(3):T(1)T(1) using the refined site-populations and the expected frequencies from previously assigned spectra in more simple amphibole compositions, and correspond to the local arrangements: (1) MgMgMg–OH–Na–OH:SiAl; (2) MgMgMg–OH–Na–F:SiAl; (3) MgMgAl–OH–Na–OH:SiAl and (4) MgMgAl–OH–Na–F:SiAl.
Grain-scale distribution of molybdenite polytypes versus rhenium contents: μXRD and EBSD data
- Olga Y. Plotinskaya, Vladimir V. Shilovskikh, Jens Najorka, Elena V. Kovalchuk, Reimar Seltmann, John Spratt
-
- Published online by Cambridge University Press:
- 30 July 2019, pp. 639-644
-
- Article
- Export citation
-
Molybdenite from two porphyry copper mineralisation sites within the South Urals was studied by electron microprobe (EMPA), micro x-ray diffraction (μXRD) and electron backscattered diffraction (EBSD) methods. Elevated contents of rhenium (0.2 to 0.4, sometimes up to 1.1 wt.%) form linear zones from several to tens of micrometres wide and up to hundreds of micrometres long parallel to the elongation of molybdenite flakes. In most cases Re-rich zones are composed of the rhombohedral (3R) polytype of molybdenite, while the rest of the molybdenite flakes with ca. 0.1 wt.% of Re consist of hexagonal (2H) molybdenite. In rare cases Re-rich zones are confined to grain boundaries of molybdenite-2H. It is shown that both μXRD and EBSD are the most appropriate tools to distinguish different polytypes within a single grain of molybdenite.