Volume 81 - Issue 2 - April 2017
Research Article
The REE- and HFSE-bearing phases in the Itatiaia alkaline complex (Brazil) and geochemical evolution of feldspar-rich felsic melts
- Leone Melluso, Vincenza Guarino, Michele Lustrino, Vincenzo Morra, Roberto de' Gennaro
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 217-250
-
- Article
- Export citation
-
The Late Cretaceous Itatiaia complex is made up of nepheline syenite grading to peralkaline varieties, quartz syenite and granite, emplaced in the metamorphic rocks of the Serra do Mar, SE Brazil. The nepheline syenites are characterized by assemblages with alkali feldspar, nepheline, Fe-Ti oxides, clinopyroxene, amphibole, apatite and titanite, while the peralkaline nepheline syenites have F-disilicates (rinkite, wöhlerite, hiortdahlite, låvenite), britholite and pyrophanite as the accessory phases. The silica-oversaturated rocks have alkali feldspar, plagioclase, quartz, amphibole, clinopyroxene and Fe-Ti oxides; the chevkinite-group minerals are the featured accessory phases and are found with allanite, fluorapatite, fluorite, zircon, thorite, yttrialite, zirconolite, pyrochlore and yttrocolumbite. The major- and trace-element composition of the Itatiaia rocks have variations linked to the amount of accessory phases, have smooth, enriched chondritenormalized rare-earth element (REE) distribution patterns in the least-evolved nepheline syenites and convex patterns in the most-evolved nepheline syenites. The REE distribution patterns of the quartz syenites and granites show a typical pattern caused by fractional crystallization of feldspar and amphibole, in an environment characterized by relatively high oxygen fugacity (>NiNiO buffer) and high concentrations of H2O and F, supporting the crystallization of hydrous phases, fluorite and F-disilicates. The removal of small amounts of titanite in the transition from the least-evolved to the most-evolved nepheline syenites stems from petrogenetic models involving REE, and is shown to be a common feature of the magmatic evolution of many other syenitic/ trachytic/ phonolitic complexes of the Serra do Mar and elsewhere.
Structure refinement and crystal chemistry of tokkoite and tinaksite from the Murun massif (Russia)
- M. Lacalamita, E. Mesto, E. Kaneva, F. Scordari, G. Pedrazzi, N. Vladykin, E. Schingaro
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 251-272
-
- Article
- Export citation
-
The structures of tokkoite, K2Ca4[Si7O18OH](OH,F) and tinaksite, K2Ca2NaTi[Si7O18OH]O from the Murun massif (Russia) were refined from single-crystal X-ray diffraction data in the triclinic space group P1̄. Average crystallographic data are a ≈ 10.423, b ≈ 12.477, c ≈ 7.112 Å, α ≈ 89.92°, β ≈ 99.68°, γ ≈ 92.97°, V ≈ 910.5 Å3 for tokkoite; a ≈ 10.373, b ≈ 12.176, c ≈ 7.057 Å, α ≈ 90.82°, β ≈ 99.22°, γ ≈ 92.80°, V ≈ 878.5 Å3 for tinaksite. The substantial similarities between the geometrical parameters of the tokkoite and tinaksite structures led us to conclude that the two minerals are isostructural. However, major differences of tokkoite with respect to tinaksite are larger lattice constants, especially concerning the b parameter, longer <M–O> distances, especially <M1–O>; larger values of the M1–M3 and O20–O2 bond lengths, and a stronger distortion of the M1 polyhedron. Mössbauer analysis showed that significant trivalent iron is present, VIFe3+ 40.0(7)% in tokkoite and 12.8(3)% in tinaksite. It is confirmed that 2Ca(M1+M2)2+ + (F,OH)(O20)–↔ Ti(M1)4+ + Na(M2)+ + O(O20)– is the exchange reaction that describes the relation between tokkoite and tinaksite. In addition, this exchange reaction causes local stress involving mainly the M1 site and its interaction with the M2 and M3 sites.
Shumwayite, [(UO2)(SO4)(H2O)2]2·H2O, a new uranyl sulfate mineral from Red Canyon, San Juan County, Utah, USA
- Anthony R. Kampf, Jakub Plášil, Anatoly V. Kasatkin, Joe Marty, Jiří Čejka, Ladislav Lapčák
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 273-285
-
- Article
- Export citation
-
The new mineral shumwayite (IMA2015-058), [(UO2)(SO4)(H2O)2]2·H2O, was found in the Green Lizard and Giveaway-Simplot mines, White Canyon district, San Juan County, Utah, USA, where it occurs as a secondary alteration phase. At the Green Lizard mine, it is found in association with calcite, gypsum, plášilite, pyrite, rozenite and sulfur; at the Giveaway-Simplot mine, shumwayite is associated with rhomboclase and römerite. The mineral occurs as pale greenish-yellow monoclinic prisms, elongated on [100], up to ∼0.3 mm long and commonly in subparallel to random intergrowths. The mineral is transparent with a vitreous lustre and has a white streak. It fluoresces bright greenish white under both longwave and shortwave ultraviolet radiation. The Mohs hardness is ∼2. Crystals are brittle with perfect {011} cleavage and irregular fracture. The mineral is slightly deliquescent and is easily soluble in room temperature H2O. The calculated density is 3.844 g cm–3. Optically, shumwayite is biaxial (+/–), with α = 1.581(1), β= 1.588(1), γ = 1.595(1) (measured in white light). The measured 2Vx based on extinction data collected on a spindle stage is 89.8(8)°; the calculated 2Vx is 89.6°. Dispersion is strong, but the sense is not defined because the optic sign is ambiguous. No pleochroism was observed. The optical orientation is X = b, Y = c, Z = a. Energy-dispersive spectrometer analyses (with H2O based on the crystal structure) yielded the empirical formula U2.01S1.99O12.00·5H2O.Shumwayite is monoclinic, P21/c, a = 6.74747(15), b = 12.5026(3), c = 16.9032(12) Å, β = 90.919(6)°, V = 1425.79(11) Å3 and Z = 4. The crystal structure (R1 = 1.88% for 2936 F > 4σF) contains UO7 pentagonal bipyramids and SO4 tetrahedra that link by corner-sharing to form [(UO2)(SO4)(H2O)2] chains along [100]. The chains and isolated H2O groups between them are linked together only by hydrogen bonds. The mineral is named in honour of the Shumway family, whose members account for the discovery and mining of hundreds of uranium deposits on the Colorado Plateau, including the Green Lizard mine.
Wilhelmgümbelite, [ZnFe2+Fe33+(PO4)3(OH)4(H2O)5]·2H2O, a new schoonerite-related mineral from the Hagendorf Süd pegmatite, Bavaria
- I. E. Grey, E. Keck, A. R. Kampf, C. M. Macrae, A. M. Glenn, J. R. Price
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 287-296
-
- Article
- Export citation
-
Wilhelmgümbelite, ideally [ZnFe2+Fe33+(PO4)3(OH)4(H2O)5]·2H2O, is a new secondary phosphate mineral related closely to schoonerite, [ZnMnFe22+Fe3+(PO4)3(OH)2(H2O)7]·2H2O, from oxidized zones of the Hagendorf-Süd pegmatite, Hagendorf, Oberpfalz, Bavaria, Germany. Wilhelmgümbelite occurs as radiating sprays of needle-like rectangular laths, up to 0.2 mm long and with colour varying from light yellow brown to orange red. Cleavage is perfect parallel to {010}. The mineral is associated closely with an oxidized pseudomorph of phosphophyllite, recently named steinmetzite. Other associated minerals are albite, apatite, chalcophanite, jahnsite, mitridatite, muscovite and quartz. The calculated density of wilhelmgümbelite is 2.82 g cm–3. It is optically biaxial (+) with α = 1.560(2), β = 1.669(2), γ = 1.718(2), 2V(meas) = 63(1)° and 2V(calc.) = 65°. Dispersion is weak with r > v, orientation X = b, Y = c, Z = a. Pleochroism is weak, with coloursZ = orange brown, Y = yellow brown, X = light yellow brown, Z >> Y > X. Electron microprobe analyses (average of seven analyses, seven crystals) with H2O and FeO/Fe2O3 calculated on structural grounds, gave FeO 5.8, Fe2O3 25.0, MnO 2.6, ZnO 16.4, P2O5 28.7, H2O 23.4, total 101.9 wt.%. The empirical formula, scaled to 3 P and OH– adjusted for charge balance is Zn1.50Mn0.272+Fe0.602+Fe2.333+(PO4)3·(OH)2.73(H2O)8.27. The structural formula is [Zn(Mn0.27Fe0.733+)∑1.0(Zn0.25Fe0.152+Fe0.603+)∑1.0(Zn0.25Fe0.452+)∑0.7Fe3+(PO4)3(OH,H2O)9]·2H2O.Wilhelmgümbelite has orthorhombic symmetry, Pmab, Z = 4, with the unit-cell parameters of a = 10.987(7) Å, b = 25.378(13) Å, c = 6.387(6) Å and V = 1781(2) Å3. The strongest lines in the powder X-ray diffraction pattern are [dobs in Å(Iobs) (hkl)] 12.65 (100) (020); 8.339 (5) (120); 6.421 (14) (001); 6.228 (8) (011); 4.223 (30) (120) and 2.111 (7) (0 12 0). Wilhelmgümbelite is an oxidized form of schoonerite, with the Mn2+ replaced principally by Fe3+. Its structure differs from that of schoonerite in having the Zn partitioned between two different sites, one five-coordinated as in schoonerite and the other tetrahedrally coordinated. Wilhelmgümbelite also differs structurally from schoonerite in having partial occupation of one of the Fe sites, which appears to be correlated with the Zn partitioning.
Stoichiometric partially-protonated states in hydroxide perovskites: the jeanbandyite enigma revisited
- Mark D. Welch, Anthony R. Kampf
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 297-303
-
- Article
- Export citation
-
The original description of the hydroxide perovskite jeanbandyite gives a formula (Fe1–x3+,□x)(Sn1–y,□y) (OH)6 (□= vacancy), which implies the possibility of stoichiometric vacancies at B and B' sites. The validity of this formula has been questioned subsequently. Furthermore, jeanbandyite has metrically a cubic unit cell, but it is optically uniaxial. It is clear that a structure determination is needed to clarify the nature of this enigmatic mineral. Previous studies could find no crystals of sufficient quality for structure determination using X-ray diffractometers available at the time. Crystals of jeanbandyite from Hingston Down, Cornwall, UK and the type locality Llallagua, Bolivia, have been found that are of a quality that allows structure refinement by single-crystal X-ray diffraction. Structural data for crystals from each locality are presented that clarify the nature of jeanbandyite and raise some interesting questions concerning the significance of partially deprotonated states in perovskite-type structures. The structures of both jeanbandyite crystals are cubic with space group Pn3 and unit-cell parameters a = 7.658(2) Å (Llallagua) and 7.6427(2) Å (Hingston). The octahedral tilt system is a+a+a+ and corresponds to that of the aristotype of BB'(OH)6 hydroxide double perovskites. Structure determination demonstrates that B is very Fe3+-rich and B' is filled by Sn, thereby requiring revision of the general jeanbandyite formula to Fex3+Fe(1–x)2+Sn(OH)(6–x)Oxfor 1≥ × > 0.5, with an ideal end-member formula Fe3+Sn(OH)5O. As such, jeanbandyite corresponds to oxidized natanite with partially deprotonated oxygen sites. This stoichiometry cannot be represented in space group Pn3̄ for the observed unit cell as it implies more than one non-equivalent oxygen atom. Consequently, it is inferred that there is no long-range ordering of deprotonated oxygen sites. It is, however, conceivable that the uniaxial optical character of jeanbandyite is linked to the local short-range order of deprotonated domains.
As-bearing new mineral species from Valletta mine, Maira Valley, Piedmont, Italy: III. Canosioite, Ba2Fe3+(AsO4)2(OH), description and crystal structure
- F. Cámara, E. Bittarello, M. E. Ciriotti, F. Nestola, F. Radica, F. Massimi, C. Balestra, R. Bracco
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 305-317
-
- Article
- Export citation
-
The new mineral species canosioite, ideally Ba2Fe3+(AsO4)2(OH), has been discovered in the dump of Valletta mine, Maira Valley, Cuneo Province, Piedmont, Italy. Its origin is probably related to the reaction between ore minerals and hydrothermal fluids. It occurs in reddish-brown granules, subhedral millimetre-size crystals, with a pale yellow streak and vitreous lustre. Canosioite is associated with aegirine, baryte, calcite, hematite, bronze Mn-bearing muscovite, unidentified Mn oxides and unidentified arsenates. Canosioite is biaxial (+) with a 2Vmeas = 84(2)°. It is weakly pleochroic with X = brownish yellow, Y = brown, Z = reddish brown, Z > Y > X. Canosioite is monoclinic, P21/m, with a = 7.8642(4), b = 6.1083(3), c = 9.1670(5) Å, β = 112.874(6)°, V = 405.73(4) Å3 and Z = 2. Calculated density is 4.943 g cm–3. The seven strongest diffraction lines of the observed powder X-ray diffraction pattern are [d in Å, (I) (hkl)]: 3.713 (18)(111), 3.304 (100)(211̄), 3.058 (31)(020), 3.047 (59)(103̄), 2.801 (73)(112), 2.337 (24)(220), 2.158 (24)(123̄). Electron microprobe analyses gave (wt.%): Na2O 0.06, MgO 0.43, CaO 0.02, NiO 0.02, CuO 0.03, SrO 0.42, BaO 49.36, PbO 1.69, Al2O3 1.25, Mn2O3 3.89, Fe2O3 6.95, Sb2O3 0.01, SiO2 0.03, P2O5 0.02, V2O5 10.88, As2O5 24.64, SO3 0.01, F 0.02, H2O1.61 was calculated on the basis of 1 (OH,F,H2O) group per formula unit. Infrared spectroscopy confirmed the presence of OH. The empirical formula calculated on the basis of 9 O apfu, is (Ba1.92Pb0.05Sr0.02Na0.01)∑2.00(Fe0.523+Mn0.293+Al0.15Mg0.06)∑1.02[(As0.64V0.36)∑1.00O4]2[(OH0.92F0.01)(H2O)0.07]and the ideal formula is Ba2Fe3+(AsO4)2(OH). The crystal structure was solved by direct methods and found to be isostructural to that of arsenbrackebuschite. The structure model was refined (R1 = 2.6%) on the basis of 1245 observed reflections. Canosioite is named after the small municipality of Canosio, where the type locality, the Valletta mine, is situated. The new mineral and name were approved by the International Mineralogical Association Commission on New Minerals and Mineral Names (IMA2015-030).
Mesaite,
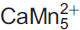 (V2O7)3·12H2O, a new vanadate mineral from the Packrat mine, near Gateway, Mesa County, Colorado, USA
(V2O7)3·12H2O, a new vanadate mineral from the Packrat mine, near Gateway, Mesa County, Colorado, USA
- Anthony R. Kampf, Barbara P. Nash, Joe Marty, John M. Hughes
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 319-327
-
- Article
- Export citation
-
Mesaite (IMA2015-069), ideally
 (V2O7)3·12H2O, is a new mineral from the Packrat mine, Gateway district, Mesa County, Colorado, USA. Crystals of mesaite occur as orangish red blades up to 0.1 mm long and ∼10 μm thick. The streak is light pinkish orange and the lustre is vitreous, transparent. Mesaite has a brittle tenacity, {010} perfect cleavage; fracture is irregular, and no parting was observed. The mineral has a Mohs hardness ≈ 2. The measured density of mesaite is 2.74(1) g cm–3. Mesaite is biaxial (–), α = 1.760(calc), β = 1.780(5), γ = 1.795(5) in white light; the measured 2V value = 81(2)°. Dispersion is strong, r < v, and pleochroism is present in shades of brownish orange. Mesaite is monoclinic, P2/n, with a = 9.146(2), b = 10.424(3), c = 15.532(4) Å, β = 102.653(7)° and V = 1444.7(6) Å3. The strongest four diffraction lines in the powder diffraction pattern are [(dobs in Å, (Iobs), (hkl)]: 10.47 (100) (010), 2.881 (25) (132, 3̄12, 033, 310), 3.568 (24) (1̄14, 1̄23, 2̄13), 3.067 (17) (1̄24, 1̄32, 2̄23). The composition of mesaite was determined by electron microprobe, and yielded an empirical formula of Mn5.32Ca0.56Zn0.31V5.96As0.04O33H23.61 on the basis of 33 O atoms per formula unit (apfu).
(V2O7)3·12H2O, is a new mineral from the Packrat mine, Gateway district, Mesa County, Colorado, USA. Crystals of mesaite occur as orangish red blades up to 0.1 mm long and ∼10 μm thick. The streak is light pinkish orange and the lustre is vitreous, transparent. Mesaite has a brittle tenacity, {010} perfect cleavage; fracture is irregular, and no parting was observed. The mineral has a Mohs hardness ≈ 2. The measured density of mesaite is 2.74(1) g cm–3. Mesaite is biaxial (–), α = 1.760(calc), β = 1.780(5), γ = 1.795(5) in white light; the measured 2V value = 81(2)°. Dispersion is strong, r < v, and pleochroism is present in shades of brownish orange. Mesaite is monoclinic, P2/n, with a = 9.146(2), b = 10.424(3), c = 15.532(4) Å, β = 102.653(7)° and V = 1444.7(6) Å3. The strongest four diffraction lines in the powder diffraction pattern are [(dobs in Å, (Iobs), (hkl)]: 10.47 (100) (010), 2.881 (25) (132, 3̄12, 033, 310), 3.568 (24) (1̄14, 1̄23, 2̄13), 3.067 (17) (1̄24, 1̄32, 2̄23). The composition of mesaite was determined by electron microprobe, and yielded an empirical formula of Mn5.32Ca0.56Zn0.31V5.96As0.04O33H23.61 on the basis of 33 O atoms per formula unit (apfu).The atomic arrangement of mesaite was solved and refined to R1 = 0.0600. The structure is formed of zigzag octahedral chains of edge-sharing Mn2+O6 octahedra. Oxygen atoms of the octahedra are shared with V2O7 groups, which link with adjacent octahedral chains to form {010} heteropolyhedral layers. The interlayer region contains Ca atoms and H2O groups. Each Ca bonds to two O6 atoms in the heteropolyhedral layer and to two fully occupied and six partially occupied O (H2O) sites in the interlayer, resulting in an effective Ca coordination of approximately seven. Similar zigzag chains of edge-sharing MnO6 octahedra decorated with V2O7 groups are also found in the mineral fianelite. Mesaite has beenapproved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA2015-069). The name mesaite is conferred for Mesa County, Colorado, USA.
Steinmetzite, Zn2Fe3+(PO4)2(OH)·3H2O, a new mineral formed from alteration of phosphophyllite at the Hagendorf Süd pegmatite, Bavaria
- I. E. Grey, E. Keck, A. R. Kampf, W. G. Mumme, C. M. Macrae, R. W. Gable, A. M. Glenn, C. J. Davidson
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 329-338
-
- Article
- Export citation
-
Steinmetzite, ideally Zn2Fe3+(PO4)2(OH)·3H2O, is a new mineral from the Hagendorf-Süd pegmatite, Hagendorf, Oberpfalz, Bavaria, Germany. Steinmetzite was found in a highly oxidized zone of the Cornelia mine at Hagendorf-Süd. It has formed by alteration of phosphophyllite, involving oxidation of the iron and some replacement of Zn by Fe. Steinmetzite lamellae co-exist with an amorphous Fe-rich phosphate in pseudomorphed phosphophyllite crystals. The lamellae are only a few μm thick and with maximum dimension ∼50 μm. The phosphophyllite pseudomorphs have a milky opaque appearance, often with a glazed yellow to orange weathering rind and with lengths ranging from sub-mm to 1 cm. Associated minerals are albite, apatite, chalcophanite, jahnsite, mitridatite, muscovite, quartz and wilhelmgümbelite.Goethite and cryptomelane are also abundant in the oxidized zone. The calculated density is 2.96 g cm–3. Steinmetzite is biaxial (–) with measured refractive indices α = 1.642(2), β = 1.659 (calc.), γ = 1.660(2) (white light). 2V(meas) = 27(1)°; orientation is Y ≈ b, X ^c ≈ 27°, with crystals flattened on {010} and elongated on [001]. Pleochroism shows shades of pale brown; Y > X ≈ Z. Electron microprobe analyses (average of seven crystals) with Fe reported as Fe2O3 and with H2O calculated from the structure gave ZnO 31.1, MnO 1.7, CaO 0.5, Fe2O3 21.9, Al2O3 0.3, P2O5 32.9, H2O 14.1 wt.%, total 102.5%. The empirical formula based on 2 P and 12 O, with all iron as ferric and OH–adjusted for charge balance is Zn1.65Fe1.193+ Mn0.112+Ca0.03Al0.023+(PO4)2(OH)1.21·2.79H2O. The simplified formula is Zn2Fe3+(PO4)2(OH)·3H2O.Steinmetzite is triclinic, P1̄, with unit-cell parameters: a = 10.438(2), b = 5.102(1), c = 10.546(2) Å, α = 91.37(2), β = 115.93(2) and γ = 94.20(2)°. V = 502.7(3) Å3, Z = 2. The strongest lines in the powder X-ray diffraction pattern are [dobs in Å (I) (hkl)] 9.313(65) (100), 5.077(38) (010), 4.726(47) (002), 4.657(100) (200), 3.365 (55) (3̄02), 3.071(54) (11̄2) and 2.735(48) (3̄1̄2). The structure is related to that of phosphophyllite.
Single-crystal X-ray diffraction, EMPA, FTIR and X-ray photoelectron spectroscopy study of narsarsukite from Murun Massif, Russia
- E. Schingaro, E. Mesto, M. Lacalamita, F. Scordari, E. Kaneva, F. N. Vladykin
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 339-354
-
- Article
- Export citation
-
A crystal chemical study of narsarsukite from the Murun alkaline massif, Russia has been carried out combining single-crystal X-ray diffraction, electron microprobe analyses, micro-Fourier transform infrared (FTIR) spectroscopy and X-ray photoelectron spectroscopy (XPS). The narsarsukite single crystals are tetragonal (space group I4/m) with unit-cell parameters: 10.7140(1) ≤ a ≤ 10.7183(2) Å and 7.9478(1) ≤ c ≤ 7.9511(1) Å. The XPS analysis showed that Fe occurs in the mineral as Fe3+, whereas the FTIR spectrum showed that the sample studied is anhydrous. The average crystal chemical formula of the Murun narsarsukite is: Na2.04K0.01(V0.015+Ti0.74Zr0.01Al0.01Fe0.223+Mg0.01)1.00Si4.00(O10.74F0.23OH0.03)11.00. Structural disorder at octahedral and interstitial sites was modelled and also discussed in consideration of the main substitutional mechanism Ti4+ + O2– ↔ Fe3+ + (F–, OH–) active in the structure of the mineral.
The crystal structure of veenite
- Dan Topa, Emil Makovicky
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 355-368
-
- Article
- Export citation
-
The crystal structure of veenite is reported for the first time from a sample from the type locality of Madoc (Ontario, Canada). It has been solved and refined by X-ray single-crystal diffraction on the basis of 4973 observed reflections (with Fo > 4σ(Fo)) with a final R1 = 0.0396. Veenite is monoclinic P21, with unit-cell parameters a = 8.429(2), b = 26.069(5), c = 8.962(2) Å, β = 117.447(2)o. The bulk veenite composition is Ag0.15Pb16.029Sb8.836As6.99S39.95 (for Z = 1) corresponding to N = 4.09 (Me8NS8N + 8, theoretical value is 4.0), with the percentage of the Ag-(As,Sb) substituted end-member only equal to 3.51 mol.%, i.e., a nearly pure Pb-Sb-As sulfosalt. The crystal structure is typical for the N = 4 sartorite homologue, with zig-zag walls of trigonal coordination prisms of Pb which separate slabs of diagonally oriented double-layers populated by Sb and As with partial Pb substitution. Orientation of three-membered crankshaft chains formed by strong (As,Sb) – S bonds on the two surfaces of double-layers differs substantially from that in dufrénoysite, which is a pure Pb-As (N = 4) sulfosalt.
Fogoite-(Y), Na3Ca2Y2Ti(Si2O7)2OF3, a Group I TS-block mineral from the Lagoa do Fogo, the Fogo volcano, São Miguel Island, the Azores: Description and crystal structure
- F. Cámara, E. Sokolova, Y. A. Abdu, F. C. Hawthorne, T. Charrier, V. Dorcet, J. -F. Carpentier
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 369-381
-
- Article
- Export citation
-
Fogoite-(Y), Na3Ca2Y2Ti(Si2O7)2OF3, is a new mineral from the Lagoa do Fogo, São Miguel Island, the Azores. It occurs in cavities as highly elongated (on [001]) prisms, up to 2000 μm long and 50 μm× 50 μm in cross-section, associated with sanidine, astrophyllite, fluornatropyrochlore, ferrokentbrooksite, quartz and ferro-katophorite. Crystals are generally transparent and colourless, with vitreous lustre, occasionally creamy white. Fogoite-(Y) has a white streak, splintery fracture and very good {100} cleavage. Mohs hardness is ∼5. Dcalc. = 3.523 g/cm3. It is biaxial (+) with refractive indices (λ = 590 nm) α = 1.686(2), β = 1.690(2), γ = 1.702(5); 2Vmeas. = 57(1)° and 2Vcalc. = 60°. It is nonpleochroic. Fogoite-(Y) is triclinic, space group P1, a = 9.575(6), b = 5.685(4), c = 7.279(5) Å, α = 89.985(6), β = 100.933(4), γ = 101.300(5)°, V = 381.2 (7) Å3. The six strongest reflections in the powder X-ray diffraction data [d (Å), I, (hkl)] are: 2.954, 100, (1̄1̄2, 3̄10); 3.069, 42, (300, 01̄2); 2.486, 24, (310, 21̄2); 3.960, 23, (1̄1̄1, 2̄10); 2.626, 21, (2̄20); 1.820, 20, (1̄04). Electron microprobe analysis gave the following empirical formula calculated on 18 (O + F) (Na2.74Mn0.15)∑2.89Ca2[Y1.21(La0.01Ce0.03Nd0.03Sm0.02Gd0.08Dy0.08Er0.05Yb0.04Lu0.01)∑0.35Mn0.16Zr0.11Na0.09Fe0.072+Ca0.01]∑2(Ti0.76Nb0.23Ta0.01)∑1(Si4.03O14)O1.12F2.88, Z = 1. The crystal structure was refined on a twinnedcrystal to R1 = 2.81% on the basis of 2157 unique reflections (Fo > 4σFo) and is a framework of TS (Titanium Silicate) blocks, which consist of HOH sheets (H – heteropolyhedral, O – octahedral) parallel to (100). In the O sheet, the the [6]MO(1) site is occupied mainly by Ti, <MO(1)–ϕ> = 1.980 Å, and the [6]MO(2) and [6]MO(3) sites are occupied by Na and Na plus minor Mn, <MO(2)–ϕ>= 2.490 Å and <MO(3)–ϕ> = 2.378 Å. In the H sheet, the two [4]Si sites are occupied by Si, with <Si–O> = 1.623 Å; the [6]MH site is occupied by Y and rare-earth elements (Y > REE), with minor Mn, Zr, Na, Fe2+ and Ca, <MH–ϕ> = 2.271 Å and the [6]AP site is occupied by Ca, <AP–ϕ> = 2.416 Å. The MH and AP octahedra and Si2O7 groups constitute the H sheet. The ideal compositions of the O and two H sheets are Na3Ti(OF)F2 and Y2Ca2(Si2O7)2 apfu. Fogoite-(Y) is isostructural with götzenite and hainite. The mineral is named after the type locality, the Fogo volcano in the Azores.
Sulfhydrylbystrite, Na5K2Ca(Al6Si6O24)(S5)(SH), a new mineral with the LOS framework, and re-interpretation of bystrite: cancrinite-group minerals with novel extra-framework anions
- A. N. Sapozhnikov, E. V. Kaneva, L. F. Suvorova, V. I. Levitsky, L. A. Ivanova
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 383-402
-
- Article
- Export citation
-
Sulfhydrylbystrite, Na5K2Ca(Al6Si6O24)(S5)(SH), cell parameters a = 12.9567(6) Å, c = 10.7711(5) Å, space group P31c, is a new mineral belonging to the cancrinite group. It was found at Malaya Bystraya lazurite deposit, Lake Baikal area, Eastern Siberian Region, Russia, associated with lazurite, calcite, diopside, phlogopite and pyrite. The mineral develops at the margins of masses of lazurite, replacing it in some areas with the formation of nonequilibrium lazurite-diopside-sulfhydrylbystrite association. It is translucent, yellow to orange, with vitreous lustre, yellow streak and Mohs hardness of 4.5–5. The empirical formula, based on 12 (Si + Al), is Na5.17K1.87Ca0.99[Al6.01Si5.99O24](S5)0.862–(SH0.86)Cl0.07, Z = 2. The crystal structure of sulfhydrylbystrite may be described as an ABAC stacking of six-membered rings of SiO4 and AlO4 tetrahedra and extra-framework cations and anions located within structural cages. There are two type of cages, cancrinite and losod, stacked into chains at (0, 0, z) and (⅔, ⅓, z), respectively. The cancrinite cage hosts Ca2+ and (SH)– ions, whereas the (S5)2– polyanion is in the losod cage associated with Na+ and K+ cations. In addition, (SH)– and (S5)2– anions are detected in the structure of a mineral for the first time.
For comparison, a structural and compositional study of a bystrite sample from the same deposit was carried out. Bystrite is confirmed to contain pentasulfide anions in the losod cages, similar to those of sulfhydrylbystrite, in contrast to previous studies. However, bystrite has chloride in cancrinite cages, whereas sulfhydrylbystrite has hydrosulfide in that position. The unit-cell parameters are distinctly different: bystrite has a = 12.8527(6) Å, c = 10.6907(5) Å in the same P31c space group.
IMA Commission on New Minerals, Nomenclature and Classification (CNMNC) Newsletter 36
New minerals and nomenclature modifications approved in 2017
- U. Hålenius, F. Hatert, M. Pasero, S. J. Mills
-
- Published online by Cambridge University Press:
- 02 January 2018, pp. 403-409
-
- Article
-
- You have access Access
- Export citation

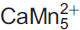 (V
(V (V
(V