Poster Presentation - Top Poster Award
Antibiotic Stewardship
Perceptions toward and practices regarding antibiotic stewardship and use among physicians at tertiary-care public hospitals in Bangladesh
- Shariful Amin Sumon, Saiful Islam, Golam Dostogir Harun
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s1
-
- Article
-
- You have access Access
- Open access
- Export citation
-
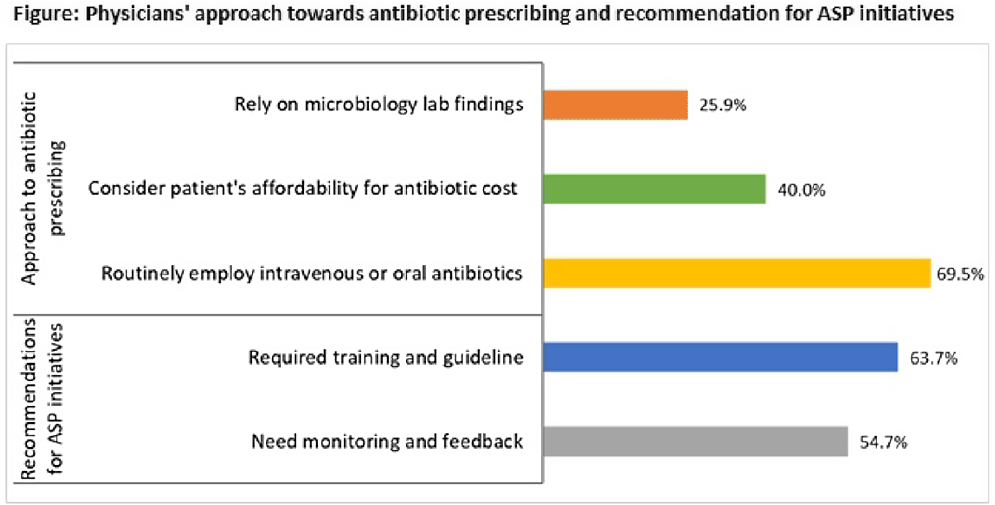
Background: The emergence of antimicrobial resistance (AMR) is affecting public health management in developing countries, including Bangladesh. Irrational and inappropriate use of antibiotics in healthcare settings has led to widespread drug resistance. To optimize antimicrobial usage to combat AMR and enhance infection treatment, the competence of healthcare workers in antibiotic prescribing is indispensable. We sought to determine the perceptions about antimicrobial resistance, antibiotic stewardship programs (ASPs), and antibiotic prescribing approaches among physicians at tertiary-care public hospitals in Bangladesh. Methods: From September to December 2020, we conducted a mixed-methods study in 9 tertiary-care public hospitals. Using a self-administered, semistructured questionnaire, we collected data on antibiotic stewardship and prescribing practices from 452 on-duty physicians. In addition, we conducted 16 key-informant interviews to explore AMR perceptions and determinants. Results: Only 43.8% of physicians were aware of the ASP, and none of the hospitals had any ASP initiative in place. Most of the participants (70.6%) recommended tailored training and antibiotic prescribing guidelines (63.7%) for effective ASP initiatives. More than half of the physicians (54.7%) preferred to receive regular monitoring and feedback on their routine antibiotic prescriptions from the stewardship program. In terms of the antibiotic prescribing approach, only 25.9% of physicians relied on microbiology lab findings, whereas 69.5% routinely employed oral or intravenous antibiotics. Also, 40.0% of physicians considered the patient’s ability to afford the antibiotic cost when recommending antibiotics. The qualitative investigation identified the use of broad-spectrum antibiotics, absence of guidelines, and inadequate laboratory support as factors contributing to AMR in the healthcare setting. Self-medication, over-the-counter dispensing, and patients’ economic instability to complete the dosage were also attributed to the irrational use of antibiotics. As a priority step, physicians advocated for intensive training on antibiotic advising, mass awareness campaigns on safe antibiotic usage and dispensing, and a restriction on the widespread sale of antibiotics from pharmacies. Conclusions: Despite favorable perceptions, the fundamental understanding of physicians regarding ASPs and rational prescribing of antibiotics needs to be improved through context-specific educational interventions and capacity building. In addition, a coherent and comprehensive policy is required for the development and implementation of antibiotic usage guidelines along with integrated ASP initiatives to combat AMR.
Funding: None
Disclosures: None
Outpatient antibiotic use for common infectious diagnoses: Patterns in telehealth during the emergence of COVID-19
- Nicole Mongilardi, Brigid Wilson, Taissa Bej, Sunah Song, Federico Jump, Federico Perez, Ukwen Akpoji
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s1-s2
-
- Article
-
- You have access Access
- Open access
- Export citation
-
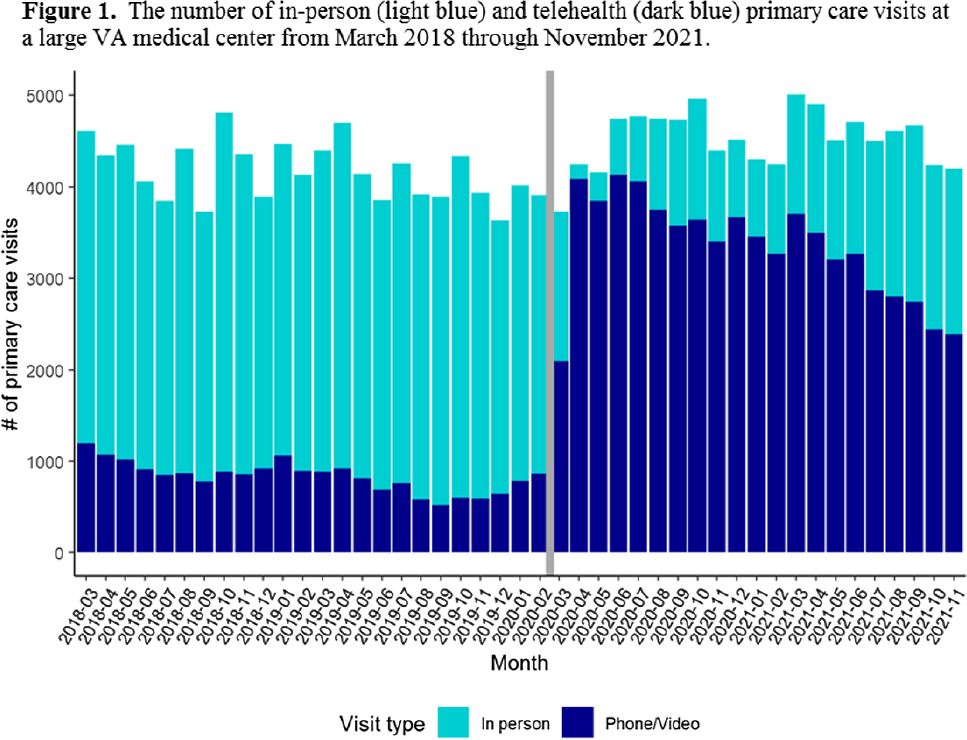
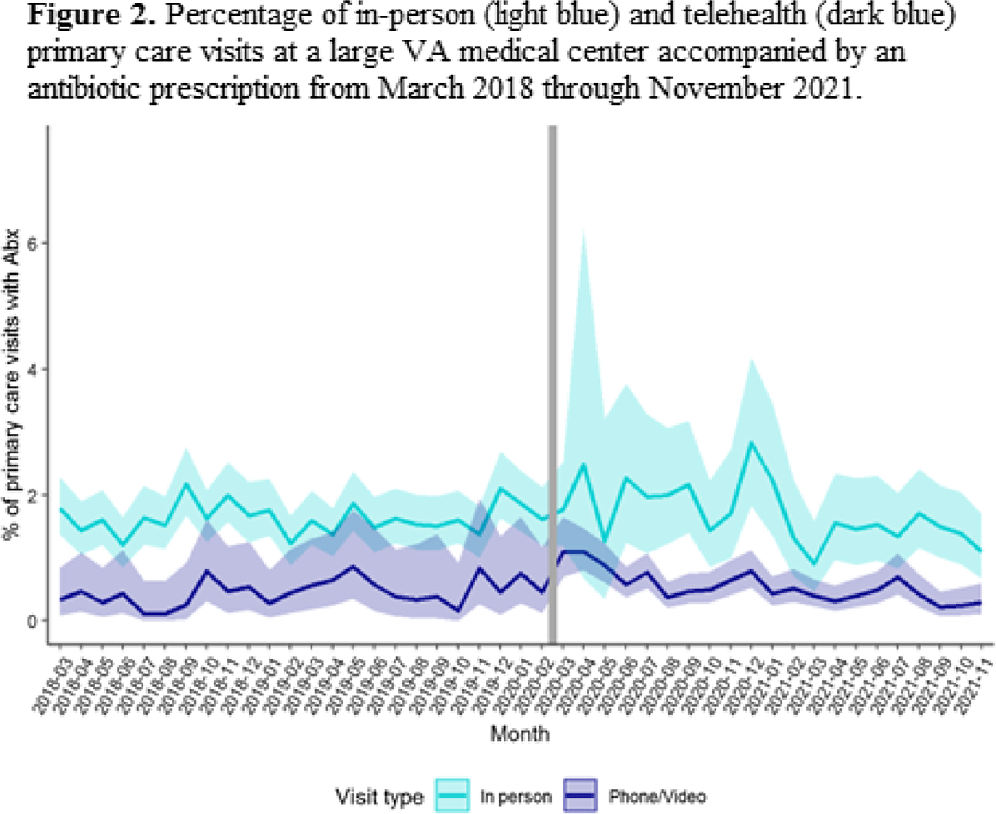
Background: The Veterans’ Affairs (VA) healthcare system has had established telehealth programs for several years. Even so, the COVID-19 pandemic led to an expansion of and changes in these services. Little is known about the influence of the increased use of telehealth due to the COVID-19 pandemic on antibiotic prescriptions in outpatient settings. Here, we report on changes in visit modality and antibiotic prescribing at primary care clinics at a large VA medical center after the emergence of the COVID-19 pandemic. Methods: Using VA administrative databases, we identified primary care visits from March 2018 to November 2019 (before the COVID-19 pandemic) and March 2020 to November 2021 (during the COVID-19 pandemic), which permitted us to account for seasonality while analyzing visit modality and antibiotic trends. For primary care visits during the pre–COVID-19 and COVID-19 periods, we have described the type of visit (in-person or telehealth), diagnostic codes for any infection, and antibiotic prescriptions. Results: The patient population was primarily men (89%) with a mean age of 62.9 years (SD, ±15.3) at first visit. The most common medical conditions were diabetes mellitus (26%) and chronic lung disease (17%). Comparing visits during the pre–COVID-19 and the COVID-19 periods, the proportions of telehealth visits were 20% (17,708 of 88,565) and 74% (69,891 of 94,937), respectively (Fig. 1). The proportions of visits with an antibiotic prescription were 1.4% (1,212 of 88,565) and 0.8% (798 of 94,396), respectively. When considered by the type of visit, the rates of antibiotics prescribed were consistent during the pre–COVID-19 and COVID-19 periods, with a lower rate for telehealth visits (Fig. 2). In both periods, >50% of antibiotic prescriptions occurred during visits without an associated infectious disease diagnosis. Conclusions: Compared to the pre–COVID-19 period, primary care providers at a large VA medical center prescribed fewer antibiotics during the COVID-19 period, and they saw most of their patients via telehealth. These results suggest that some aspects of telehealth may support clinical practices consistent with antibiotic stewardship. The prescription of an antibiotic without an associated diagnostic code also suggests opportunities to improve implementation of antibiotic stewardship principles in primary care settings.
Funding: This work was supported by the Merck Investigator Studies Program (grant no. MISP 59266 to F.P. and R.J.) and by funds and facilities provided by the Cleveland Geriatric Research.
Disclosures: None
Do hospitalists who prescribe high (risk-adjusted) rates of antibiotics do so repeatedly?
- Udodirim Onwubiko, Christina Mehta, Zanthia Wiley, Jesse Jacob, Ashley Jones, Shabir Hassan, Marybeth Sexton, Sujit Suchindran, Scott Fridkin
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s2
-
- Article
-
- You have access Access
- Open access
- Export citation
-
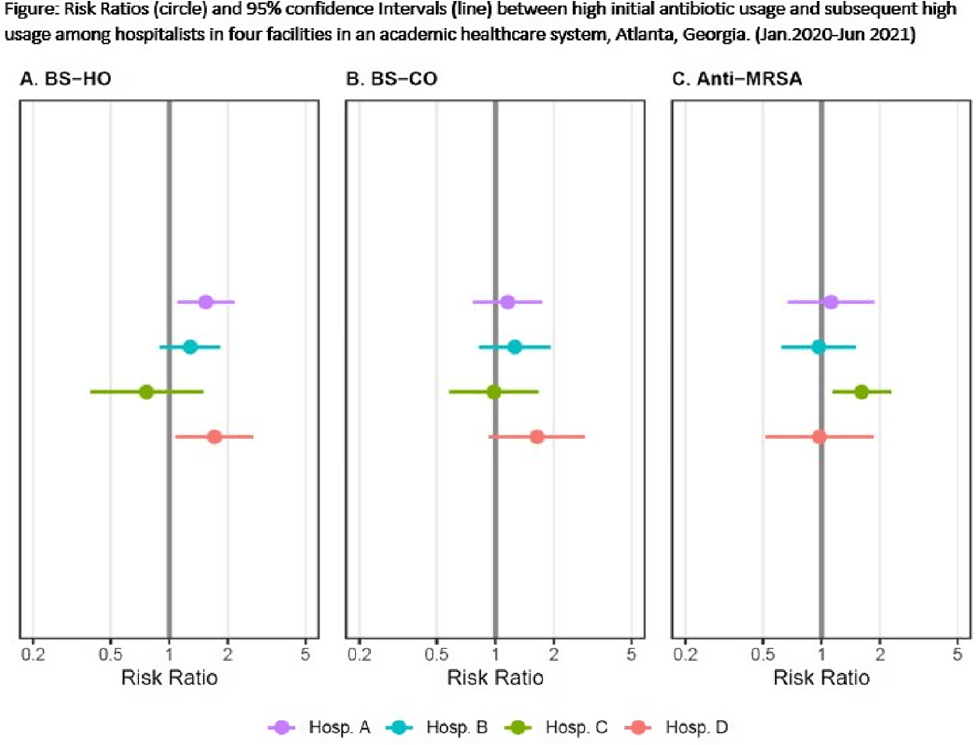
Background: Provider-specific prescribing metrics can be used for benchmarking and feedback to reduce unnecessary antibiotic use; however, metrics must be credible. To improve credibility of a recently described risk-adjusted antibiotic prescribing metric for hospital medicine service (HMS) providers, we assessed whether providers who initially prescribed excess antibiotics continued to prescribe antibiotics excessively. Methods: We linked administration and billing data among patients at 4 acute-care hospitals (1,571 beds) to calculate days of therapy (DOT) ordered by individual hospitalists for each of 3 NHSN antibiotic groupings: broad-spectrum hospital onset (BS-HO), broad-spectrum community-onset (BS-CO), or anti-MRSA for each patient day billed from January 2020 to June 2021. To incorporate repeated measures by provider, mixed models adjusted for patient-mix characteristics (eg, % encounters with urinary tract infection, etc) were used to calculate serial, bimonthly, provider-specific, observed-to-expected ratios (OERs). An OER of 1.25 indicates that the prescribing rate observed was 25% higher than predicted, adjusting for patient mix. We then used log binomial generalized estimating equations to assess whether a high prescribing rate (defined as an OER ≥ 1.25) for an individual provider in an earlier bimonthly period was associated with a persistent high rate for that provider in the following period. Results: Overall, 975 bimonthly periods were evaluated from 136 hospitalists. Most (58%) contributed data the entire 18-month study period. Median OERs were similar between hospitals: 0.94 (IQR, 0.65–1.28) for BS-HO antibiotic use, 0.99 (IQR, 0.73–1.24) for BS-CO antibiotic use, and 0.95 (IQR, 0.65–1.28) for anti-MRSA antibiotic use. At the individual prescriber level, roughly one-quarter of bimonthly OERs (range varied by group and hospital from 21% to 31%) were categorized as high. At 3 of the 4 hospitals, a provider with a high OER for either BS-HO or BS-CO antibiotic use in any bimonthly period was more likely to have a high OER in the subsequent period (Fig. 1). These observed risk ratios were statistically significant for BS-HO antibiotic use at only 2 hospitals: hospital A risk ratio (RR) was 1.54 (95% CI, 1.10–2.16); hospital B RR was 1.28 (95% CI, 0.90–1.82); hospital C RR was 0.76 (95% CI, 0.39–1.48); and ospital D RR was 1.71 (95% CI, 1.09–2.68). Conclusions: Our findings suggest that hospitalists with a higher than expected 2-month period of antibiotic prescribing are likely to continue to have elevated prescribing rates in the following period, particularly for BS-HO antibiotics. These findings increase the credibility of using a 2-month prescribing metric for BS-HO antibiotic stewardship efforts; further work is needed to evaluate utility for other antibiotic groupings.
Funding: None
Disclosures: None
Outpatient antibiotic prescribing during the COVID-19 pandemic–United States, January 2019–October 2021
- Destani Bizune, Sharon Tsay, Sarah Kabbani, Lauri Hicks
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s2-s3
-
- Article
-
- You have access Access
- Open access
- Export citation
-
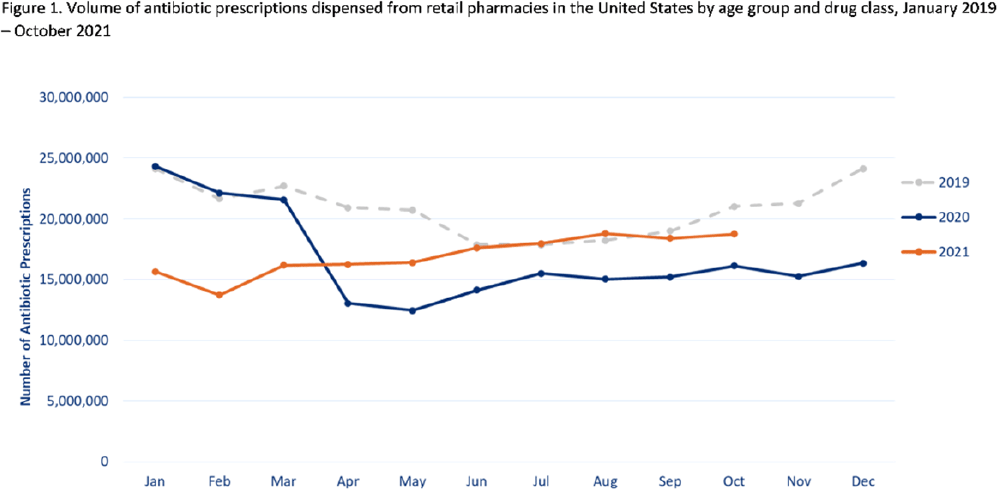
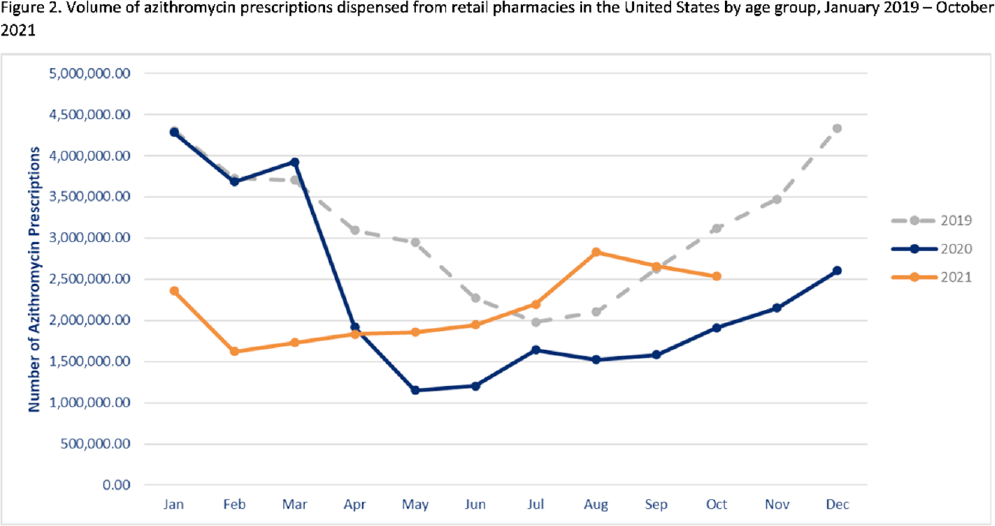
Background: Improving antibiotic use is a key strategy to combat antimicrobial resistance. Here, we have described national outpatient antibiotic prescribing trends during the COVID-19 pandemic. We compared the monthly numbers of prescriptions in 2020–2021 to those from 2019 to describe the impact of the pandemic and to highlight areas for improvement. Methods: We used the IQVIA National Prescription Audit (NPA) data set to identify all antibiotic prescriptions dispensed from US retail pharmacies during January 2019–October 2021. We calculated the percentage change in volume of prescriptions for each month during the pandemic (beginning in March 2020) compared to the baseline (defined as the corresponding month in 2019). Data were characterized by patient age group (0–19 years, 20–64 years, ≥65 years) and antibiotic class and drug, including azithromycin. Results: Antibiotic prescriptions were lower than baseline during March 2020–June 2021. The greatest decrease in antibiotic prescribing volume occurred in May 2020 (40.0% lower than May 2019) (Fig. 1), with the greatest decreases among children 0–19 years of age. However, prescribing was similar to baseline levels in July–August 2021 (Fig. 1). Specifically, azithromycin prescribing exceeded the 2019 baseline by 11.0% in July and further to a 34.5% increase in August 2021 (Fig. 2). Increases in azithromycin prescribing in August 2021 were observed across all age groups: 20–64 years (46.9% above baseline), ≥65 years (25.3% above baseline), and children 0–19 years (7.8% above baseline). Conclusions: Antibiotic prescribing volume was lower during 2020 and the first half of 2021 compared to the corresponding months in 2019. Decreases in outpatient antibiotic prescriptions during the pandemic likely reflect decreased utilization of outpatient healthcare and decreased transmission of non–COVID-19 infections secondary to nonpharmaceutical interventions (eg, masking, social distancing, school closures). However, outpatient antibiotic prescribing levels in general, and azithromycin prescribing in particular, approached or exceeded prepandemic levels in July and August 2021. Ongoing surveillance and sustained outpatient antibiotic stewardship efforts are needed to optimize antibiotic use during the COVID-19 pandemic and beyond.
Funding: None
Disclosures: None
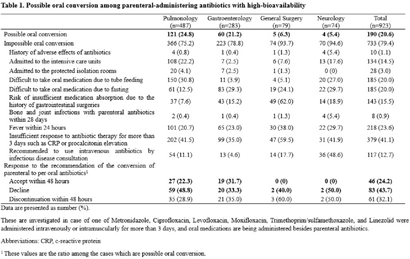
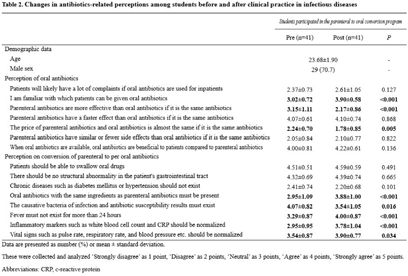
The effect of a parenteral-to-oral conversion program for high-bioavailability antibiotics use
- Wooyoung Jang, Bongyoung Kim
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s3-s4
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Appropriate conversion of antibiotics from parenteral to the oral route can lower the risk of catheter-associated infections, reduce medical costs, and shorten hospitalization. We investigated the effect of a parenteral-to-oral conversion program on high-bioavailability antibiotics conducted by medical students and the changes in perceptions of oral antibiotics after participating in the program. Methods: The parenteral-to-oral conversion program was implemented as a core clinical practice course for the fifth-year medical students in 2021 at the infectious diseases department in an affiliated hospital of a medical school in Korea. Half of the students in this class participated in the program from January to October 2021. An evaluation of the possibility of oral conversion was performed for parenterally administered, high oral-bioavailability antibiotics including ciprofloxacin, levofloxacin, moxifloxacin, metronidazole, linezolid, and trimethoprim–sulfamethoxazole. These agents are prescribed in the departments of pulmonology, gastroenterology, general surgery, and neurology. The medical students reviewed medical records for the patients treated with those antibiotics and wrote a recommendation for oral conversion for the cases with “possible oral conversion” after an infectious disease specialist confirmed their assessments. The cases without administration of any oral drugs or with the duration of parenteral antibiotic use of <3 days were excluded from the evaluation. The following cases were considered as “impossible oral conversion” and were excluded from the intervention: (1) admitted to the ICUs, (2) admitted to the protected isolation rooms, (3) difficult to take oral medication, (4) risk of insufficient medication absorption, e) bone and joint infections, (5) fever within 24 hours, (6) insufficient response to antibiotic therapy, and (7) recommended to use intravenous antibiotics by consultation with an infectious disease specialist. Furthermore, a survey was conducted on the perception of oral antibiotics in medical students before and after clinical practice to evaluate the educational effect of this program. Results: In total, 923 cases were reviewed, and 190 (20.6%) of 923 antibiotics prescriptions with high oral bioavailability were found to be administered parenterally even though they could be converted oral administration. Among these 190 antibiotics prescriptions, 46 (24.2%) were changed via a written proposal within 48 hours, 83 (43.7%) proposed changes were declined, and 61 (32.1%) antibiotics prescriptions were discontinued within 48 hours. Through this program, students have gained a better perception of oral antibiotics. Conclusions: This parenteral-to-oral conversion program showed a 24.2% acceptance rate of oral antibiotics conversions in the hospital, and it had significant educational effects on medical students regarding an appropriate perception of oral antibiotics.
Funding: None
Disclosures: None
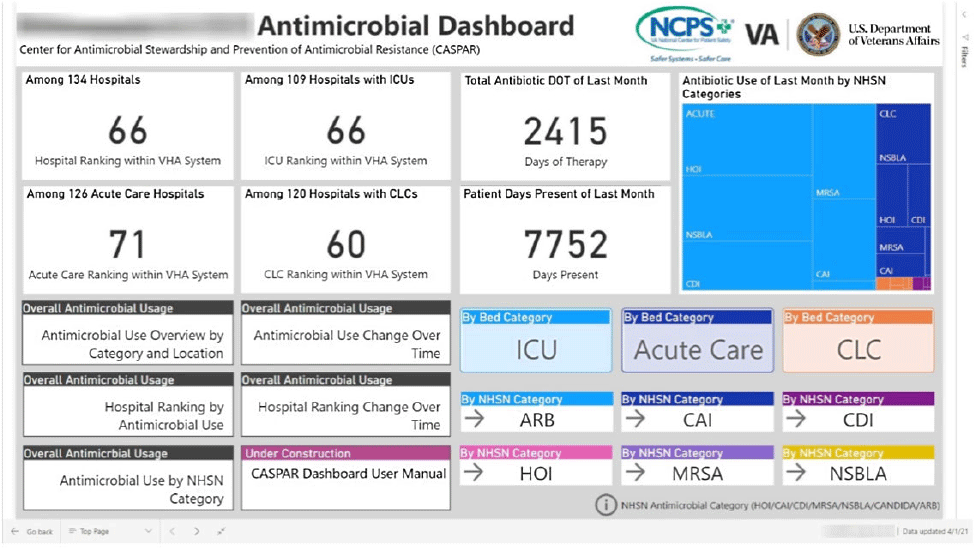
Qualitative Evaluation of an automated nationwide benchmarking antimicrobial utilization dashboard for the VHA
- DeShauna Jones, Alexandre Marra, Daniel Livorsi, Eli Perencevich, Michihiko Goto
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s4
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antimicrobial stewardship programs (ASPs) are advised to audit antimicrobial consumption as a metric to feedback to clinicians. However, many ASPs lack the tools necessary for appropriate risk adjustment and standardized data collection, which are critical for peer-program benchmarking. We evaluated the impact of the dashboard deployment that displays these metrics and its acceptance among ASP members and antimicrobial prescribers. Materials/methods: We conducted semistructured interviews of ASP stewards and antimicrobial prescribers before and after implementation of a web-based ASP information dashboard (Fig. 1) implemented in the VA Midwest Health Care Network (VISN23). The dashboard provides risk-adjusted benchmarking, longitudinal trends, and analysis of antimicrobial usage patterns at each facility. Risk-adjusted benchmarking was based on an observed-to-expected comparison of antimicrobial days of therapy at each facility, after adjusting for differences in patient case mix and facility-level variables. Respondents were asked to evaluate several aspects of the dashboard, including its ease of use, applicability to ongoing ASP activities, perceived validity and reliability, and advantages compared to other ASP monitoring systems. All interviews were digitally recorded and transcribed verbatim. The analysis was conducted using MaxQDA 2020.4 and the Consolidated Framework for Implementation Research (CFIR) constructs. Results: We completed 4 preimplementation interviews and 11 postimplementation interviews with ASP champions and antimicrobial prescribers from 6 medical centers. We derived 4 key themes from the data that map onto CFIR constructs. These themes were interconnected so that implementation of the dashboard (ie, adapting and adopting) was influenced by respondents’ perception of a facility’s size, patient population, and priority placed on stewardship (ie, structural and cultural context), the availability of dedicated stewardship staff and training needed to implement the dashboard (ie, resources needed), and how the dashboard compared to established stewardship activities (ie, relative advantage). ASP champions and antimicrobial prescribers indicated that dashboard metrics were useful for identifying antimicrobial usage and for comparing metrics among similar facilities. Respondents also specified barriers to acceptance of the risk-adjusted metric, such as disagreement regarding how antimicrobials were grouped by the current NHSN protocol, uncertainty of factors involved in risk adjustments, and difficulty developing a clear interpretation of hospital rankings. Conclusions: Given the limited resources for antimicrobial stewardship personnel, automated, risk-adjusted, antimicrobial-use dashboards provided by ASPs are an attractive method to both facilitate compliance and improve efficiency. To increase the uptake of surveillance systems in antimicrobial stewardship, our study highlights the need for clear descriptions of methods and metrics.
Funding: None
Disclosures: None
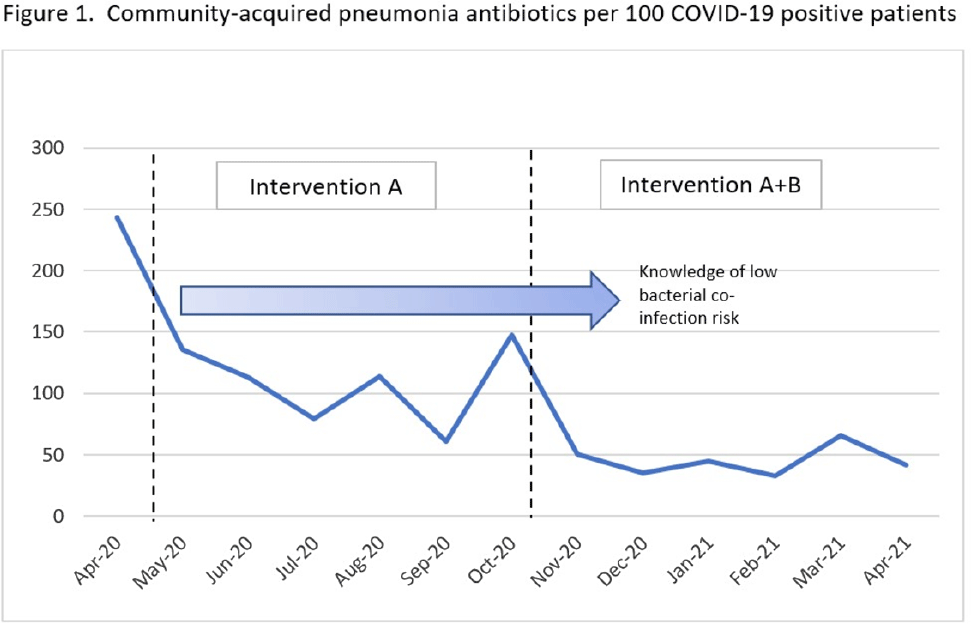
A little education goes a long way: Decreasing antibiotics for community-acquired pneumonia in COVID-19 patients
- Ravi Tripathi, Rohini Dave, Elizabeth Eden, Jacqueline Bork
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s4-s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antibiotic use was common in patients with suspected or confirmed COVID-19 infection; however, data emerged demonstrating low rates of bacterial coinfection (6%–10%). Antimicrobial stewardship best practice was challenged during this time, requiring new strategies and education to limit the inappropriate use of antibiotics. At the Veterans’ Affairs Maryland Healthcare System, we evaluated the use of community acquired pneumonia (CAP) specific antibiotics in COVID-19–positive patients after successive interventions. Methods: We conducted a pre–post evaluation of common CAP antibiotics (ceftriaxone IV/IM, cefpodoxime PO, azithromycin PO/IV, ampicillin/sulbactam IV, amoxicillin-clavulanate PO, levofloxacin) during the COVID-19 pandemic. The preintervention period was April–October 2020 and the postintervention period was November 2020–April 2021. During the preintervention period, intervention A was carried out as follows: (1) inpatient weekly virtual interdisciplinary COVID-19 rounds were led by an antimicrobial stewardship champion, (2) χprocalcitonin was implemented in clinical decision making, and (3) inpatient audit and feedback of active antibiotics was conducted by the antimicrobial stewardship team. In the postintervention period, intervention B was added as follows: (1) weekly educational COVID-19 virtual seminars were conducted for providers, and (2) targeted education was provided to emergency department and hospitalist directors. Comparisons of the proportions of antibiotics prescribed were made between the pre- and postintervention periods using X2 statistic, and data were stratified by location. The rates of CAP antibiotic prescription per 100 COVID-19–positive patients were also compared using Poisson distribution. Results: During the study period, 814 unique patients had COVID-19 infection: 182 (22.4%) patients admitted to the acute-care center, 66 (8.1%) long-term care residents, and 566 (69.5%) were managed outside the hospital. Of these 814 patients, 211 (25%) were prescribed a CAP antibiotic. Of the antibiotics prescribed, 223 (61%) were ceftriaxone, cefpodoxime, amoxicillin-clavulanate, or ampicillin-sulbactam; 123 (34%) were azithromycin; and 16 (4.4%) were levofloxacin. We observed a decrease in the frequency of all antibiotic prescriptions after intervention B was added: 32% (86 of 273) vs 23% (125 of 541) (P = .01). Decreases in antibiotic prescriptions were observed in all locations: acute care (57% vs 44%), long-term care (53% vs 41%) and outpatient care (19% vs 15%). The rates of CAP antibiotic prescribing per 100 COVID-19–positive patients were 114 in the preintervention period and 45 in the postintervention period, a rate difference of −70 antibiotics per 100 COVID-19–positive patients (pConclusions: Curbing antibiotic use for CAP indication during the COVID-19 pandemic was a challenge. A multifaceted approach focusing on education was an impactful intervention leading to significant decreases in antibiotic prescribing despite COVID-19 cases increasing.
Funding: None
Disclosures: None
Impact of different COVID-19 encounter definitions on antibiotic prescribing rates in urgent care
- Sharon Onguti, David Ha, Emily Mui, Amy Chang, Eddie Stenehjem, Adam Hersh, Marisa Holubar
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
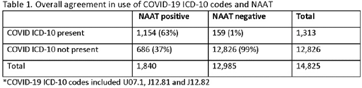
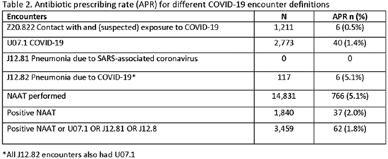
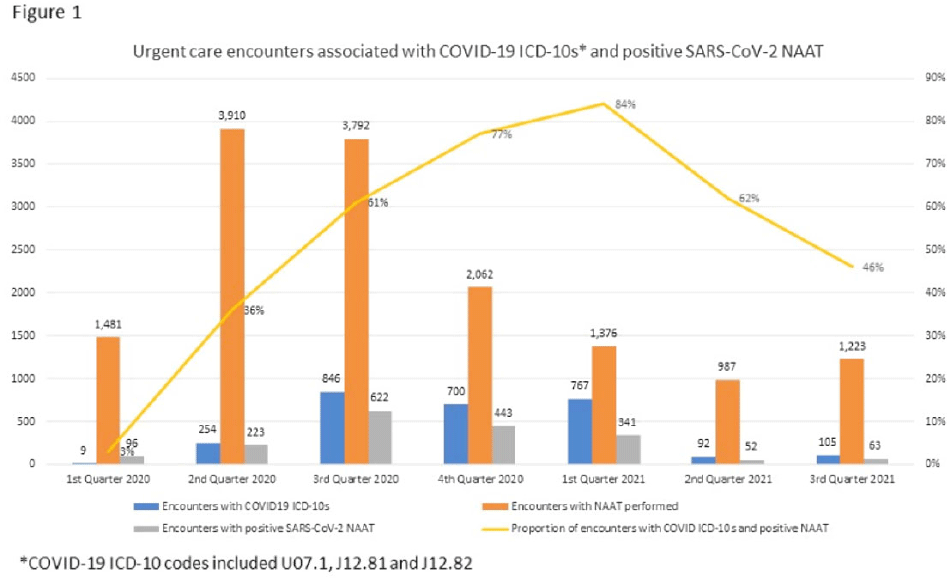
Background: Billing data have been used in the outpatient setting to identify targets for antimicrobial stewardship. However, COVID-19 ICD-10 codes are new, and the validity of using COVID-19 ICD-10 codes to accurately identify COVID-19 encounters is unknown. We investigated COVID-19 ICD-10 utilization in our urgent care clinics during the pandemic and the impact of using different COVID-19 encounter definitions on antibiotic prescribing rates (APRs). Methods: We included all telemedicine and office visits at 2 academic urgent-care clinics from January 2020 to September 2021. We extracted ICD-10 encounter codes and testing data from the electronic medical record. We compared encounters for which COVID-19 ICD-10 codes were present with encounters for which SARS-CoV-2 nucleic acid amplification testing (NAAT) was performed within 5 days of and up to 2 days after the encounter (Fig. 1). We calculated the sensitivity of the use of COVID-19 ICD-10 codes against a positive NAAT. We calculated the APR as the proportion of encounters in which an antibacterial drug was prescribed. This quality improvement project was deemed non–human-subjects research by the Stanford Panel on Human Subjects in Medical Research.
Funding: None
Disclosures: None
Tier-based antimicrobial stewardship metrics for genitourinary-related antibiotic use in Veterans’ Affairs outpatient settings
- Matthew Samore, Matthew Goetz, McKenna Nevers, Jacob Crook, Suzette Rovelsky, Ben Brintz, Kelly Echevarria, Melinda Neuhauser, Sharon Tsay, Lauri Hicks, Karl Madaras-Kelly
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s5-s6
-
- Article
-
- You have access Access
- Open access
- Export citation
-
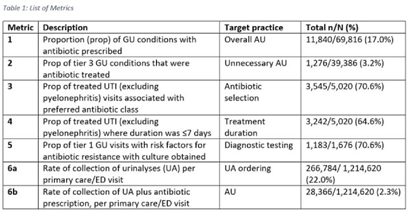
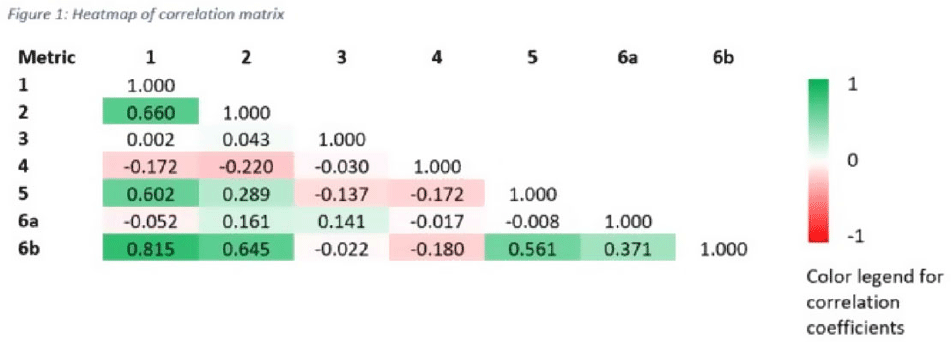
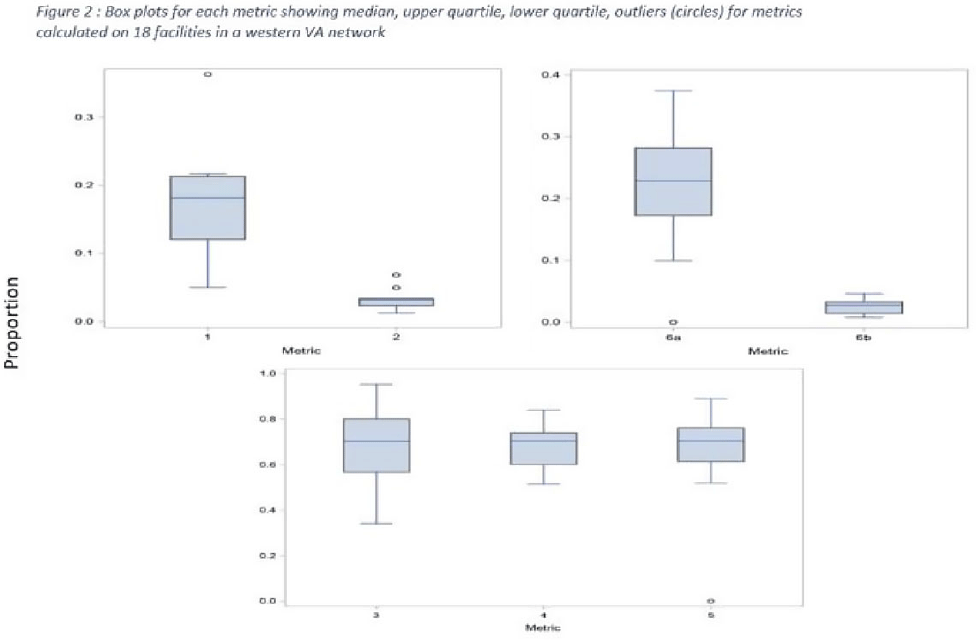
Background: Tracking antibiotic use is a core element of antimicrobial stewardship. We developed a set of metrics based on electronic health record data to support an outpatient stewardship initiative to improve management of urinary tract infections (UTIs) in Veterans’ Affairs (VA) emergency departments (EDs) and primary care clinics. Because UTI diagnostic codes only capture a portion of genitourinary (GU)-related antibiotic use, a tier-based approach was used to evaluate practices. Methods: Metrics were developed to target practices related to antibiotic prescribing and diagnostic testing (Table 1). GU conditions were divided into 3 categories: tier 1, conditions for which antibiotics are usually or always indicated; tier 2, conditions for which antibiotics are sometimes indicated; and tier 3, conditions for which antibiotics are rarely or never indicated (eg, benign prostatic hypertrophy with symptoms). Patients with visits related to urological procedures, nontarget providers, and concomitant non-GU infections were excluded. Descriptive analyses included calculation of the correlation matrix for the 7 metrics and the construction of box plots to display interfacility variability. Results: Metrics were calculated quarterly for 18 VA medical centers, including affiliated clinics, in a western VA network, from July 2018 to June 2020 (Table 1). Tier 3 GU conditions accounted for 1,276 of 11,840 (11%) of GU-related antibiotic use. Metrics 1 and 6b were strongly correlated with each other and were also positively correlated with metrics 2 and 5 (coefficients > 0.5) (Fig. 1). Substantial interfacility variation was observed (Fig. 2). Conclusions: Stewardship metrics for suspected or documented UTIs can identify opportunities for practice improvement. Broadly capturing GU conditions in addition to UTIs may enhance utility for performance feedback. Antibiotic prescribing for tier 3 GU conditions is analogous to unnecessary antibiotic use for acute, uncomplicated bronchitis and upper respiratory tract infections.
Funding: None
Disclosures: None
COVID-19
COVID-19 incidence among nonphysician healthcare workers at a tertiary care center–Iowa, 2020–2021
- Takaaki Kobayashi, John Heinemann, Alexandra Trannel, Alexandre Marra, William Etienne, Oluchi Abosi, Stephanie Holley, Mary Kukla, Angie Dains, Kyle Jenn, Holly Meacham, Beth Hanna, Bradley Ford, Melanie Wellington, Patrick Hartley, Daniel Diekema, Jorge Salinas
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s6-s7
-
- Article
-
- You have access Access
- Open access
- Export citation
-
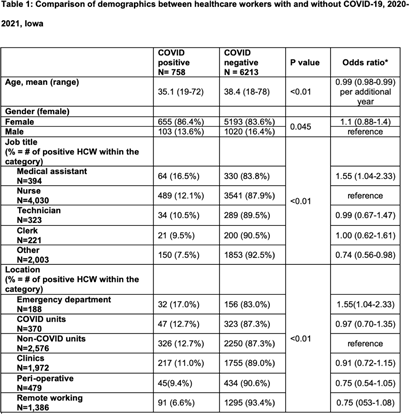
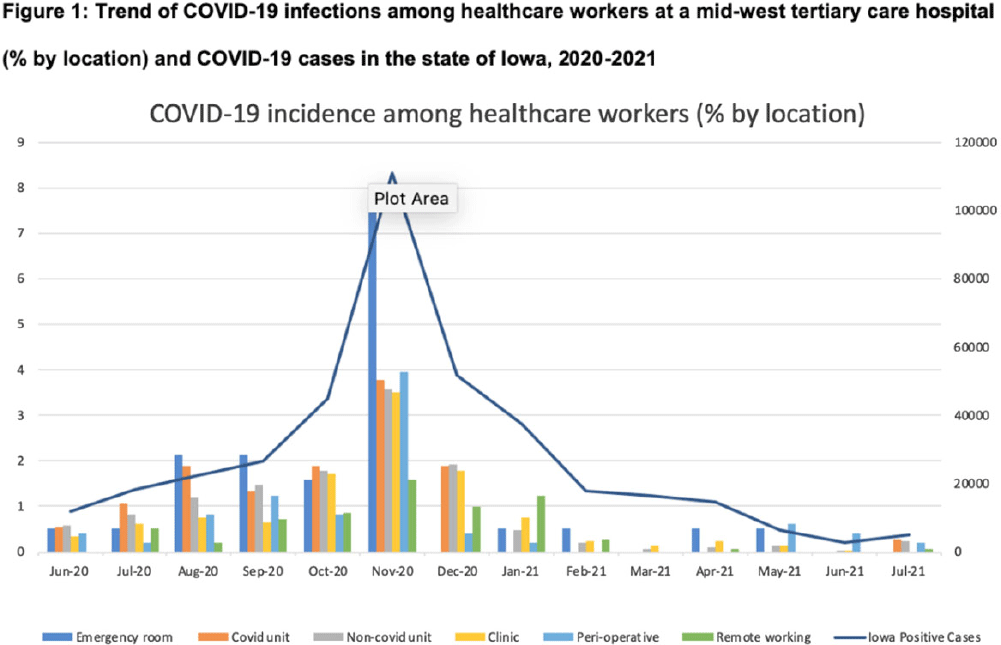
Background: Whether working on COVID-19 designated units put healthcare workers (HCWs) at higher risk of acquiring COVID-19 is not fully understood. We report trends of COVID-19 incidence among nonphysician HCWs and the association between the risk of acquiring COVID-19 and work location in the hospital. Methods: The University of Iowa Hospitals & Clinics (UIHC) is an 811-bed, academic medical center serving as a referral center for Iowa. We retrospectively collected COVID-19–associated data for nonphysician HCWs from Employee Health Clinic between June 1st 2020 and July 31th 2021. The data we abstracted included age, sex, job title, working location, history of COVID-19, and date of positive COVID-19 test if they had a history of COVID-19. We excluded HCWs who did not have a designated working location and those who worked on multiple units during the same shift (eg, medicine resident, hospitalist, etc) to assess the association between COVID-19 infections and working location. Job titles were divided into the following 5 categories: (1) nurse, (2) medical assistant (MA), (3) technician, (4) clerk, and (5) others (eg patient access, billing office, etc). Working locations were divided into the following 6 categories: (1) emergency department (ED), (2) COVID-19 unit, (3) non–COVID-19 unit, (4) Clinic, (5) perioperative units, and (6) remote work. Results: We identified 6,971 HCWs with work locations recorded. During the study period, 758 HCWs (10.8%) reported being diagnosed with COVID-19. Of these 758 COVID-19 cases, 658 (86.8%) were diagnosed before vaccines became available. The location with the highest COVID-19 incidence was the ED (17%), followed by both COVID-19 and non–COVID-19 units (12.7%), clinics (11.0%), perioperative units (9.4%) and remote work stations (6.6%, p Conclusions: Strict and special infection control strategies may be needed for HCWs in the ED, especially where vaccine uptake is low. The administrative control of HCWs working remotely may be associated with a lower incidence of COVID-19. Given that the difference in COVID-19 incidence among HCWs by location was lower and comparable after the availability of COVID-19 vaccines, facilities should make COVID-19 vaccination mandatory as a condition of employment for all HCWs, especially in areas where the COVID-19 incidence is high.
Funding: None
Disclosures: None
Which nursing home workers were at highest risk for SARS-CoV-2 infection during the November 2020–February 2021 winter surge of COVID-1?
- Joseph Kellogg, William Dube, Carly Adams, Matthew Collins, Theodore Lopman, Theodore Johnson, Avnika Amin, Joshua Weitz, Scott Fridkin
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s7
-
- Article
-
- You have access Access
- Open access
- Export citation
-
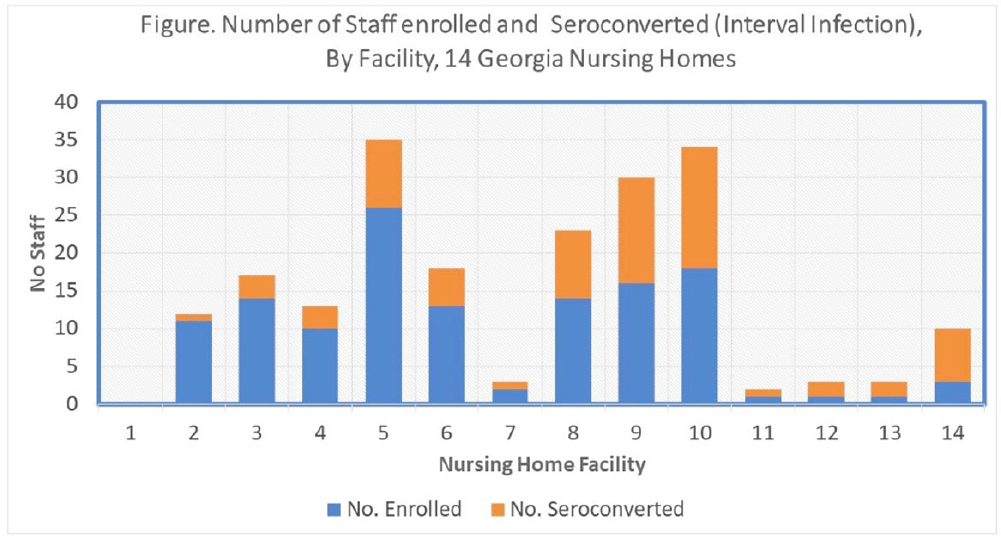
Background: Nursing home (NH) residents and staff were at high risk for COVID-19 early in the pandemic; several studies estimated seroprevalence of infection in NH staff to be 3-fold higher among CNAs and nurses compared to other staff. Risk mitigation added in Fall 2020 included systematic testing of residents and staff (and furlough if positive) to reduce transmission risk. We estimated risks for SARS-CoV-2 infection among NH staff during the first winter surge before widespread vaccination. Methods: Between February and May 2021, voluntary serologic testing was performed on NH staff who were seronegative for SARS-CoV-2 in late Fall 2020 (during a previous serology study at 14 Georgia NHs). An exposure assessment at the second time point covered prior 3 months of job activities, community exposures, and self-reported COVID-19 vaccination, including very recent vaccination (≤4 weeks). Risk factors for seroconversion were estimated by job type using multivariable logistic regression, accounting for interval community-incidence and interval change in resident infections per bed. Results: Among 203 eligible staff, 72 (35.5%) had evidence of interval seroconversion (Fig. 1). Among 80 unvaccinated staff, interval infection was significantly higher among CNAs and nurses (aOR, 4.9; 95% CI, 1.4–20.7) than other staff, after adjusting for race and interval community incidence and facility infections. This risk persisted but was attenuated when utilizing the full study cohort including those with very recent vaccination (aOR, 1.8; 95% CI, 0.9–3.7). Conclusions: Midway through the first year of the pandemic, NH staff with close or common resident contact continued to be at increased risk for infection despite enhanced infection prevention efforts. Mitigation strategies, prior to vaccination, did not eliminate occupational risk for infection. Vaccine utilization is critical to eliminate occupational risk among frontline healthcare providers.
Funding: None
Disclosures: None
Coinfections in hospitalized COVID-19 patients are associated with high mortality: need for improved diagnostic tools
- Sonya Kothadia, Brigid Wilson, Federico Perez, Robert Bonomo
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s7-s8
-
- Article
-
- You have access Access
- Open access
- Export citation
-
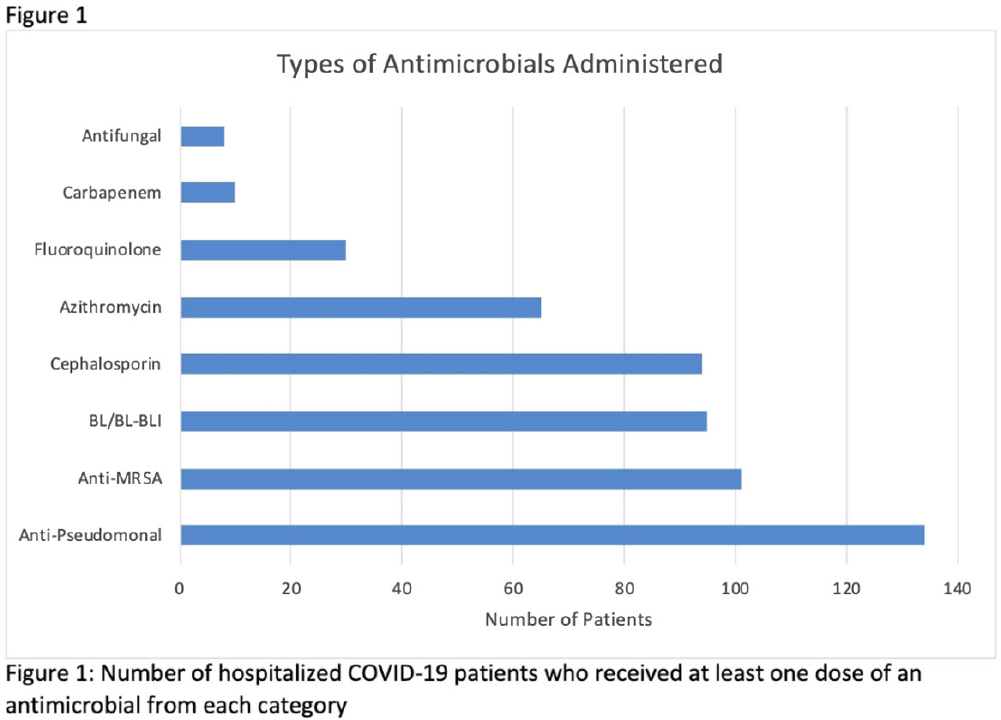
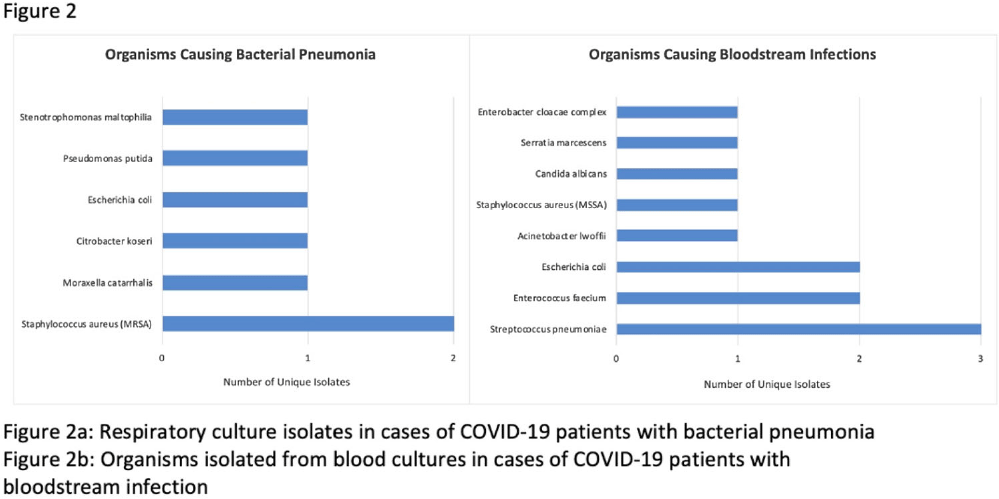
Background: Hospitalized patients with COVID-19 often receive antimicrobial therapies due to concerns for bacterial and fungal coinfections. We analyzed patients admitted with COVID-19 to our VA facility to understand antimicrobial use, frequency of coinfections, and outcomes in our population. Methods: This retrospective study included veterans who were 18 years or older and hospitalized with COVID-19 from March 10, 2020, to March 9, 2021 at the Louis Stokes VA Medical Center in Cleveland, Ohio. We identified antimicrobials administered and coinfections with bacterial or fungal pathogens. Patients were deemed to have coinfection if there was supporting microbiological data and a consistent clinical course upon review of clinical records. Urinary tract infections were excluded because of difficulty determining infection. Odds ratios (ORs) and 95% confidence intervals (CIs) for 30-day mortality were derived using multivariate logistic regression models that included age, Charlson comorbidity index (CCI), corticosteroid use, and time of infection. Results: In our cohort of 312 patients, the median age was 70 years and 97% of the patients were male. The mean CCI was 3.7 (SD, 3.0), and 111 patients (35.6%) had a score ≥5. Oxygen was administered to 250 patients (80.1%), and 20 (6.4%) required mechanical ventilation. Antimicrobials were administered to 164 patients (52.6%) (Fig. 1). Of 20 patients (6.4%) with coinfection, 11 (3.5%) had a bloodstream infection (BSI) and 9 (2.9%) had bacterial pneumonia (Fig. 2). The overall 30-day mortality rate was 12.5% (39 of 312). Among patients with coinfection, the 30-day mortality rate was 45% (9 of 20). Diagnoses of BSI (OR, 6.35; 95% CI, 1.41–26.30) and bacterial pneumonia (OR, 9.34; 95% CI, 2.01–46.34) were associated with increased mortality. Of the data available, 12 (63%) of 19 patients with coinfection had elevated procalcitonin levels (ie, >0.50). At the time of COVID-19 diagnosis, the median absolute lymphocyte count in patients who died was 0.7 K/mm3 (95% CI, 0.6–1.12) in comparison to 1 K/mm3 (95% CI, 0.7–1.4) in patients who survived at 30 days. Conclusions: Our analysis of hospitalized COVID-19 patients with advanced age and underlying comorbid conditions demonstrated that coinfections were infrequent but that they were independently associated with increased mortality. This finding highlights the need for better tools to diagnose the presence or absence of bacterial and fungal coinfection in COVID-19 patients. Our findings also emphasize the need for judicious use of antimicrobials while discerning which patients are at risk of critical illness and mortality.
Funding: None
Disclosures: None
Characteristics of healthcare personnel who reported concerns related to PPE use during care of COVID-19 patients
- Nora Chea, Stephanie Tavitian, Cedric Brown, Taniece Eure, Rebecca Alkis, Gregory Blazek, Austin Penna, Joelle Nadle, Linda Frank, Christopher Czaja, Helen Johnston, Devra Barter, Kathleen Angell, Kristen Marshall, James Meek, Monica Brackney, Stacy Carswell, Stepy Thomas, Scott Fridkin, Lucy Wilson, Ashley Fell, Sara Lovett, Sarah Lim, Ruth Lynfield, Ruth SarahShrum, Erin C. Phipps, Marla Sievers, Ghinwa Dumyati, Cate Concannon, Kathryn McCullough, Woods, Sandhya Seshadri, Christopher Myers, Rebecca Pierce, Valerie Ocampo, Judith Guzman-Cottrill, Gabriela Escutia, Monika Samper, Sandra Pena, Cullen Adre, Tiffanie Markus, Kathryn Billings, Matthew Groenewold, Ronda Sinkowitz-Cochran, Shelley Magill, Cheri Grigg, Betsy Miller
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s8-s9
-
- Article
-
- You have access Access
- Open access
- Export citation
-
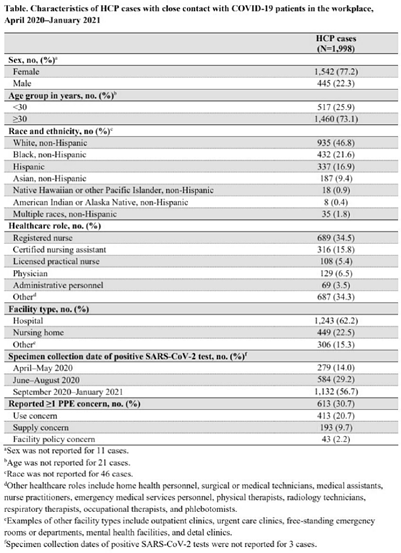
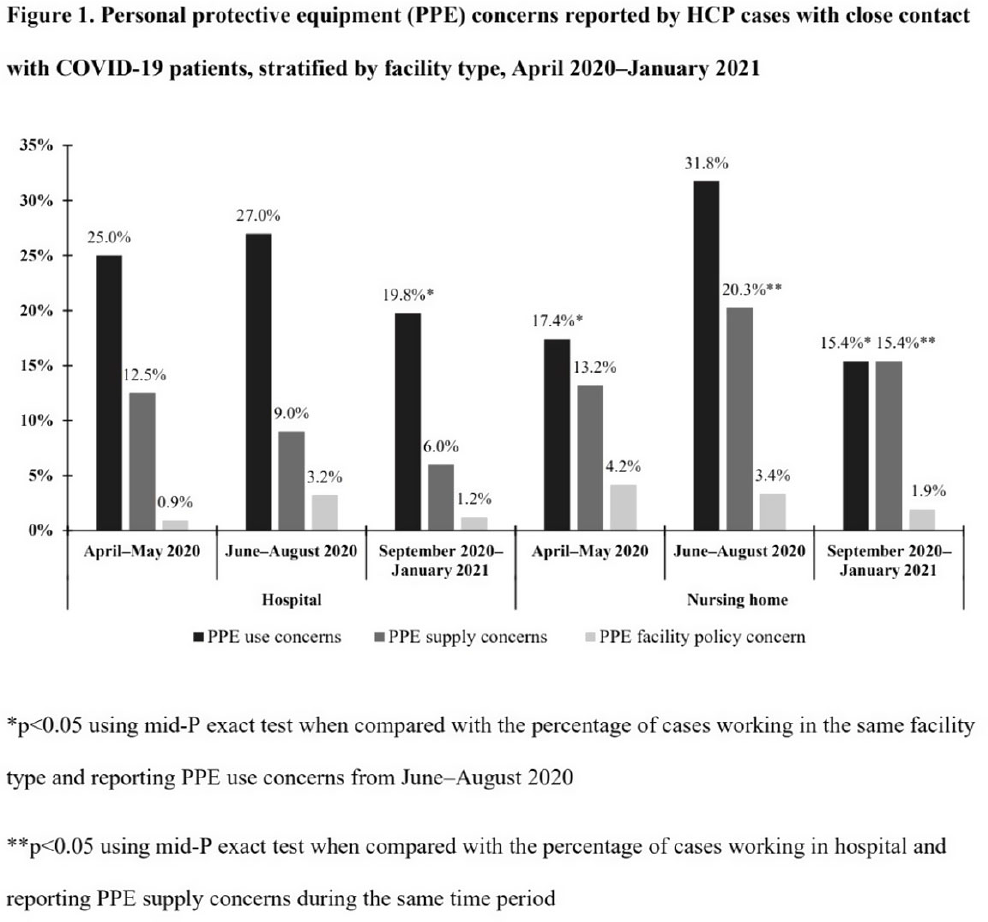
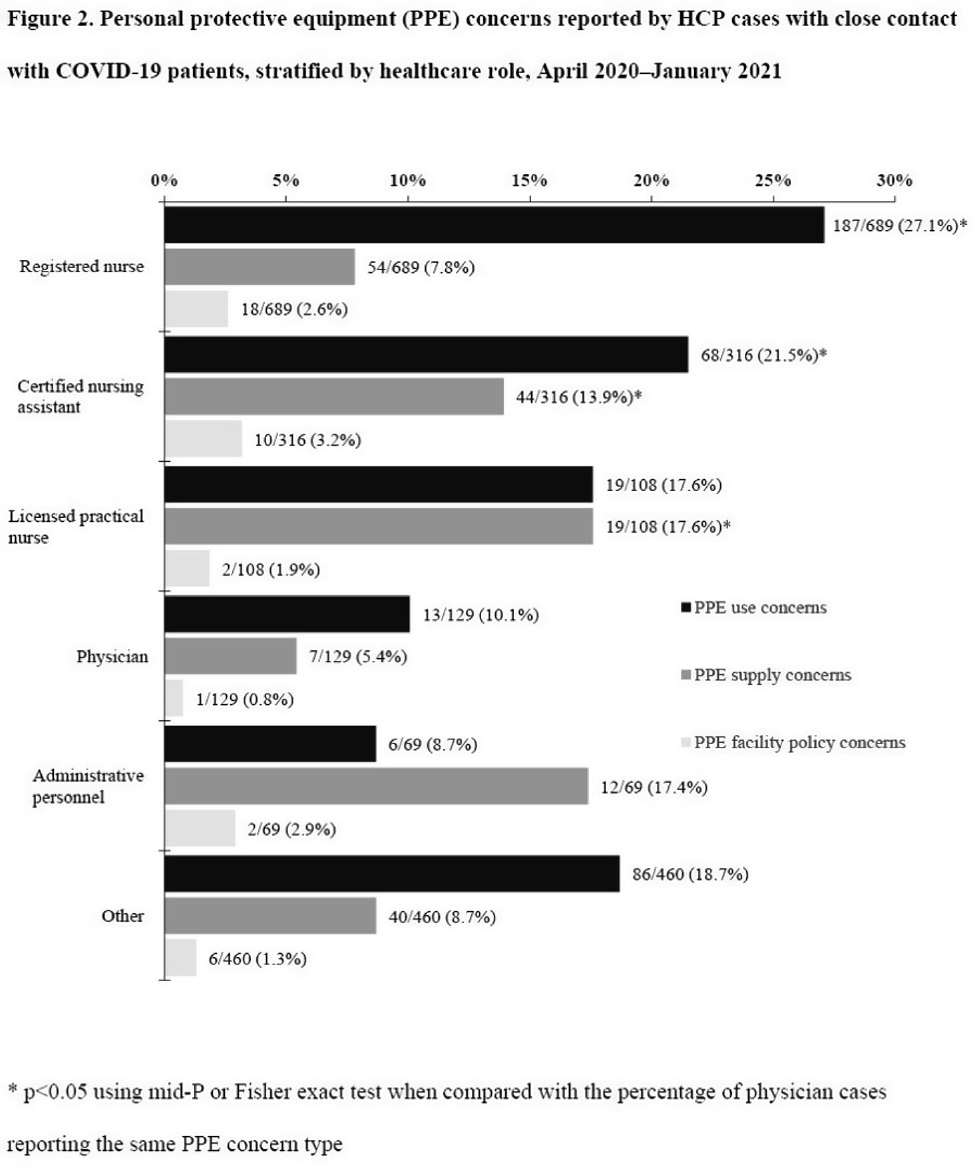
Background: Healthcare facilities have experienced many challenges during the COVID-19 pandemic, including limited personal protective equipment (PPE) supplies. Healthcare personnel (HCP) rely on PPE, vaccines, and other infection control measures to prevent SARS-CoV-2 infections. We describe PPE concerns reported by HCP who had close contact with COVID-19 patients in the workplace and tested positive for SARS-CoV-2. Method: The CDC collaborated with Emerging Infections Program (EIP) sites in 10 states to conduct surveillance for SARS-CoV-2 infections in HCP. EIP staff interviewed HCP with positive SARS-CoV-2 viral tests (ie, cases) to collect data on demographics, healthcare roles, exposures, PPE use, and concerns about their PPE use during COVID-19 patient care in the 14 days before the HCP’s SARS-CoV-2 positive test. PPE concerns were qualitatively coded as being related to supply (eg, low quality, shortages); use (eg, extended use, reuse, lack of fit test); or facility policy (eg, lack of guidance). We calculated and compared the percentages of cases reporting each concern type during the initial phase of the pandemic (April–May 2020), during the first US peak of daily COVID-19 cases (June–August 2020), and during the second US peak (September 2020–January 2021). We compared percentages using mid-P or Fisher exact tests (α = 0.05). Results: Among 1,998 HCP cases occurring during April 2020–January 2021 who had close contact with COVID-19 patients, 613 (30.7%) reported ≥1 PPE concern (Table 1). The percentage of cases reporting supply or use concerns was higher during the first peak period than the second peak period (supply concerns: 12.5% vs 7.5%; use concerns: 25.5% vs 18.2%; p Conclusions: Although lower percentages of HCP cases overall reported PPE concerns after the first US peak, our results highlight the importance of developing capacity to produce and distribute PPE during times of increased demand. The difference we observed among selected groups of cases may indicate that PPE access and use were more challenging for some, such as nonphysicians and nursing home HCP. These findings underscore the need to ensure that PPE is accessible and used correctly by HCP for whom use is recommended.
Funding: None
Disclosures: None
Improved assay for detecting SARS-CoV-2 from nonporous hospital surfaces using surrogate human coronavirus OC43
- Lisa Tran, William Furin, Geun Woo Park, Martinique Edwards, Carrie Whitworth
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s9
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Understanding SARS-CoV-2 persistence on surfaces can help inform transmission risk from surfaces in healthcare and community settings. A sensitive viral infectivity assay is crucial for the detection of infective virus in environmental investigations. The conventional cell culture-based infectivity assay is limited by the time dependence, subjectivity, and insensitivity of cytopathic effect (CPE) scoring. We validated an integrated cell-culture and reverse-transcription quantitative RT-PCR method (cc-RT-qPCR) to improve SARS-CoV-2 detection and reduce detection time. We compared cc-RT-qPCR with CPE-scored cell culture to evaluate assay sensitivity of recovered virus from stainless-steel coupons simulating nonporous healthcare surfaces. Method: Human β-coronavirus OC43, a model strain for SARS-CoV-2, was propagated on HRT-18G cells in growth medium at 33°C in a 5% CO2 incubator. The OC43 infectivity was determined by cell culture with a 10-fold dilution series of viral samples in 96-well plates, and incubation for 7 days at 33°C to confirm CPE. Plates were CPE-scored and TCID50 was calculated using the Reed-Muench method. For the cc-RT-qPCR assay, CPE-negative wells were interrogated for viral intracellular replication using RT-PCR; infectivity was based on a titer increase of ≥ 2 logs 7 days after inoculation using RT-qPCR. CPE-positive or replicative virus-harboring cells were enumerated to determine TCID50. The sensitivity of both CPE-scored cell culture and cc-RT-qPCR assays were evaluated by inoculating 105 TCID50/mL OC43 in infection media and artificial saliva matrices onto coupons and dried in an environmental chamber at 26°C and 57% relative humidity for 6 hours. Viral eluates from coupons served as test samples. Results: Low-titer infectious OC43 (0.75 log10) was detected by both methods 7 days after incubation; however, infectivity confirmation required 4 and 6 days after incubation, respectively, for cc-RT-qPCR and CPE-scored cell culture methods. When cells were inoculated with OC43 at titer range 1.75–4.75 log10, CPE presented at 4–5 days after incubation, while viral replication was already detected at 3 days after incubation via RT-PCR. Upon virus titration, cc-RT-qPCR demonstrated greater sensitivity, detecting up to 1 log10 higher of infectious OC43 than cell culture alone at 0 and 6 hours (P ≤ .05) dried in infection medium and 0 hours (P ≤ .05) in saliva. Conclusions: Our data demonstrated greater sensitivity and shorter times to detect viral replication by cc-RT-qPCR, minimizing potential for false-negative results with cell culture alone. This sensitive assay may provide investigators with quicker results for informing infection control practices to reduce risk of transmission from deposited bodily fluids on surfaces, eg, coughing and sneezing.
Funding: None
Disclosures: None
Work system factors affecting COVID-19 PPE use: A human factors approach to analysis of video recordings of emergency department clinical work
- Esosa Nosakhare, Shawna Perry, Susan Peterson, Frankie Catalfumo, Kelly Osei, Kelly Osei, Kelly Osei, Kelly Williams, Maia Bradley, Marium Sultan, Oluseyi Daodu, Nivedha Prabhu, Ayse Gurses
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s9-s10
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The effectiveness of PPE in preventing self-contamination of healthcare workers (HCWs) and transmission of pathogens (airborne and contact) in the emergency department (ED) is highly dependent on consistent, appropriate use of and other interactions (eg, storing, cleaning, etc) with the PPE. Pre–COVID-19 studies focused primarily on individual HCW contributions to incorrect or suboptimal PPE use. We conducted an analysis of ED video recordings using a human-factors engineering framework (ie, The Systems Engineering Initiative for Patient Safety, SEIPS), to identify work-system–level contributions to inappropriate PPE usage by HCWs while they provide care in their actual clinical care environment. Methods: In total, 47 video sessions (each ~15 minute) were recorded between June 2020 and May 2021 using a GoPro camera in an 8-bed pod area, designated for persons under investigation (PUI) and confirmed COVID-19–positive patients, in an ED of a large, tertiary-care, academic medical center. These recordings captured a ‘landscape view’: 2 video cameras were set up to capture the entire ED pod area and HCWs as they provided care. A team with hemorrhagic fever expertise, infection prevention and control expertise, and ED expertise reviewed each video together and extracted data using a semistructured form. Results: Guided by the 5 components of the SEIPS work system model, (ie, task, physical environment, person, organization, tools and technology), multiple work system failure points influencing HCWs appropriate use of PPE were identified. For example, under the task component, HCWs were observed not doffing and donning in recommended sequence. Also, inconsistencies with COVID-19 status signage on a patient’s door and ambiguous labelling of work areas designated as clean (donning) and dirty (doffing) sites acted as a barrier to appropriate PPE use under the physical environment section. Conclusions: Human factors–based analysis of video recordings of actual ED work identified a variety of work system factors that impede appropriate or correct use of PPE by HCWs. Future efforts to improve appropriate PPE use should focus on eliminating or mitigating the effects of these work system factors.
Funding: US CDC
Disclosures: The authors gratefully acknowledge the CDC for funding this work. This material is based upon work supported by the Naval Sea Systems Command (under contract No. N00024-13-D-6400, Task Order NH076). Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the Naval Sea Systems Command (NAVSEA) or the US CDC.
Work system barriers to & resilience strategies for COVID-19 PPE use in the emergency department: A qualitative interview study
- Oluseyi Daodu, Ayse Gurses, Patience Osei, Esosa Nosakhare, Shawna Perry, Marium Sultan, Nivedha Prabhu, Susan Peterson, Emma MacIntyre, Khue Vo, Lauren Yuan, Lauren Benishek, Jessica Li
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s10
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Emergency departments (EDs) are complex, sociotechnical, high-paced, safety-critical work systems that have been disproportionately affected by the COVID-19 pandemic. Despite training, consistent compliance with recommended PPE use during COVID-19 pandemic has been challenging. Healthcare workers (HCWs) have had adapt to overcome these challenges to ensure their own safety and patient safety. We sought to identify barriers in the work system that impede the recommended COVID-19 PPE use in EDs. Methods: We conducted semistructured, in-depth interviews over ZoomTM from August 2020–May 2021 with 45 HCWs from the ED (ie, physicians, nurses, ancillary support staff, etc) affiliated with a large, tertiary-care, academic medical center. These audio-recorded interviews were transcribed and analyzed using a hybrid (inductive and deductive) qualitative coding approach in NVivo software. The deductive portion was guided by the SEIPS work system model, a well-known human-factors conceptual framework. Results: We identified multiple work-system factors in the ED that impede compliance with the recommended COVID-19 PPE use. In addition, ED HCWs have reported making a variety of adaptations or developing strategies to overcome these barriers. Some of these adaptations were made to the PPE physically (eg, trimming portions of PPE), and others were related to the tasks and/or processes associated with PPE, such as filming their own training video demonstrating PPE donning and doffing techniques, and environment services staff checking a patient’s status with nurses prior to entering the patient’s room when there was no COVID-19 signage on the door. Conclusions: Consistent compliance with COVID-19 PPE use in ED clinical practice is challenging and can be negatively affected by a variety of work system factors. Resilience strategies developed by HCWs can provide critical information with regards to HCW needs and potential directions for innovation. Future efforts should focus on not only changing individual HCW behavior through training but also on improving the PPE and ED work system design.
Funding: US CDC
Disclosures: The authors gratefully acknowledge the US CDC for funding this work. This material is based upon work supported by the Naval Sea Systems Command (under contract no. N00024-13-D-6400, task order NH076). Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the Naval Sea Systems Command (NAVSEA) or the US CDC.
Analysis of Universal admission laboratory screening for SARS-CoV-2 asymptomatic infection across a large health system
- Jennifer Cihlar, Bryan Harris, Patty Wright, Romney Humphries, Kelly (Caroline), Brandi Cherry, Thomas Talbot
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s10-s11
-
- Article
-
- You have access Access
- Open access
- Export citation
-
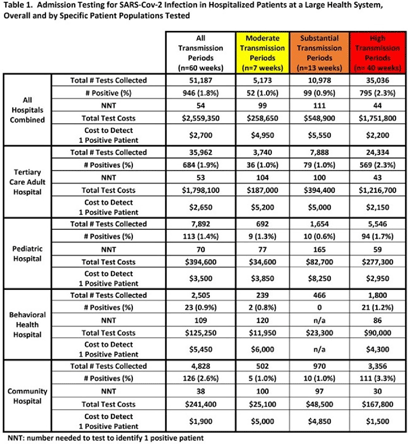
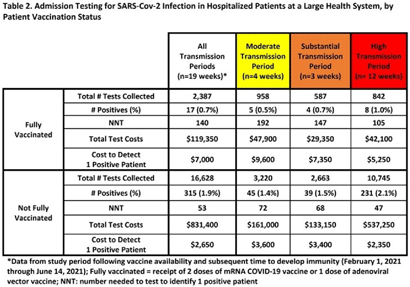
Background: Admission laboratory screening for asymptomatic COVID-19 has been utilized to mitigate healthcare-associated SARS-CoV-2 transmission. A better understanding of the impact of such testing across a variety of patient populations is needed. Methods: Beginning April 2020, every patient admitted within an academic healthcare system underwent SARS-CoV-2 PCR testing upon admission. Between April 20, 2020 through June 14, 2021, results were analyzed in asymptomatic patients across 4 inpatient facilities: a tertiary-care adult hospital, a free-standing pediatric hospital, a community-based hospital, and a behavioral health hospital. Positivity rates and the number needed to test (NNT) to identify 1 asymptomatic infected patient were calculated overall, by hospital type, by patient vaccination status, and by CDC-defined levels of community transmission. Weekly community incidence rates of COVID-19 for the system’s metropolitan service area (8 central Tennessee counties) were obtained from Tennessee Department of Health records. Weekly COVID-19 incidence rates per 100,000 people were calculated using US Census Bureau data. Using a national survey of hospital epidemiologists, a clinically meaningful NNT was identified (ie, 1 positive patient per 100 patients tested). A crude admission testing cost (covering testing supplies, reagents, and lab personnel costs) was obtained from operational data ($50 per test) to assess testing utility. Results: In total, 51,187 tests were collected during the study period with a positivity rate of 1.8%. No periods of low transmission were observed (Table 1). During high transmission periods, the NNT met the clinically relevant threshold in all populations. In addition, the NNT approached or met the 1:100 threshold for most locations during periods of less transmission, suggesting continued benefit even as infection rates decline. In all transmission periods, the NNT for non–fully vaccinated patients met the clinically meaningful threshold, in contrast to testing of fully vaccinated patients (Table 2). Discussion: Implementing an asymptomatic patient admission testing program can provide clinically relevant value based on the NNT, even during lower periods of transmission and in different patient populations. Limiting admission testing to non–fully vaccinated patients during periods of lower transmission may be a strategy to address cost and resource concerns around this practice. Further investigations into the impact of booster vaccination and newer SARS-CoV-2 variants on admission testing programs are also necessary. Although the impact of such testing on healthcare-associated COVID-19 among patients and healthcare workers could not be clearly determined, these data provide important information as facilities weigh the costs and benefits of such testing.
Funding: None
Disclosures: None
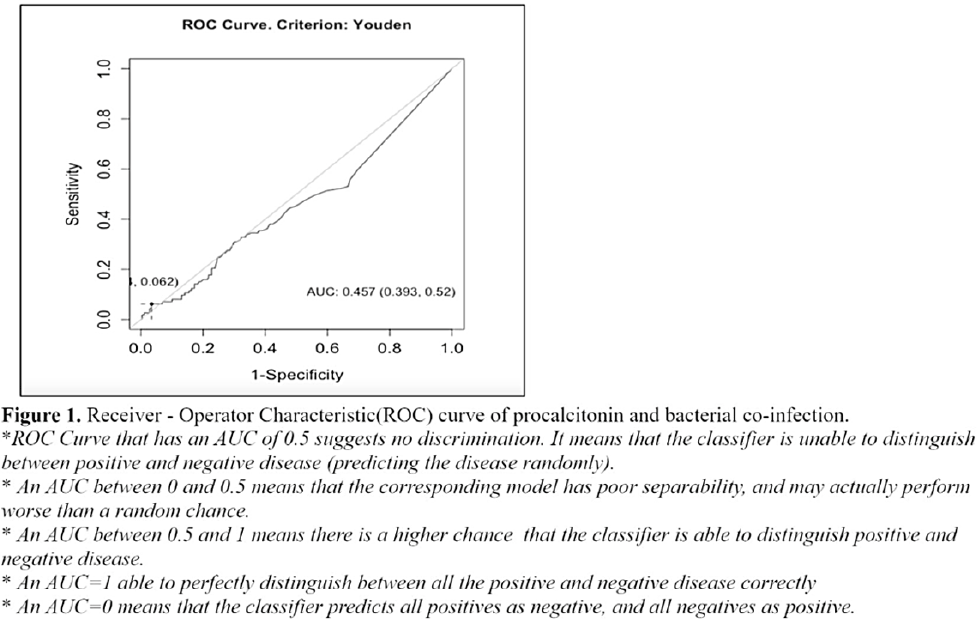
Procalcitonin as marker for bacterial coinfection among adult COVID-19 patients in a tertiary-care hospital in the Philippines
- Angelie Lorraine Pesebre, Cybele Lara Abad, Jan Jorge Francisco, Justin Allister Ong
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s11
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antimicrobials are often given to patients with COVID-19 despite the absence of a bacterial coinfection. Procalcitonin (PCT), when elevated, often indicates the presence of a bacterial infection and is used to guide empiric antibiotic therapy. We sought to determine the utility of PCT and the optimal cutoff value of PCT among patients with COVID-19. Methods: We retrospectively reviewed all COVID-19 confirmed ca-ses hospitalized in our institution from March to December 2020. Of 729 cases, we included 403 (55.3%) who had baseline PCT and blood or respiratory tract specimens (eg, sputum, endotracheal aspirate) within 48 hours of admission. Participants were classified according to PCT levels and COVID-19 severity. A receiver operating characteristic (ROC) curve analysis was performed. The area under the curve (AUC) obtained was used to compute the possible optimal cutoff value using the Youden index. A χ2 test was used to define association between groups according to the characteristics of variables. Results: Of a total cohort of 403, 245 (57%) were male, with an overall median age of 60 years (range, 22–94). Overall, 28 presented with mild COVID-19, 194 presented with moderate COVID-19, and 181 presented with severe or critical COVID-19. Moreover, 363 (90%) were given antibiotics. Of 28 with mild COVID-19, 22 (79%) received empiric antibiotics. The rate of bacterial coinfection was high at 28% (113 of 403). Klebsiella pneumoniae was the most commonly identified microorganism: 52 (19.5%) of 266 patients. Based on the ROC curve, the optimal cutoff for PCT was 4.72 ng/mL, with 97% specificity and only 6% sensitivity. Only 17 participants had PCT > 4.72 ng/mL. Of these, 1 was mild, 5 were moderate COVID-19, 8 had severe COVID-19, and 3 had critical COVID-19; all received antibiotic therapy. Conclusions: In our cohort, the rate of bacterial coinfection was high. A PCT of >4.72 ng/mL increased the likelihood of a coinfection. However, PCT had poor sensitivity and may not detect the presence of bacterial coinfection, especially when used alone. Serial PCT monitoring, its use in conjunction with other markers, or as a prognostic tool, need to be explored further.
Funding: None
Disclosures: None
Prioritizing SARS-CoV-2 testing in a highly immunosuppressed patient population
- Jenna Shackelford, Michele Woolbert, Ninet Sinaii, Brooke Decker, Tara Palmore, Robin T Odom
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s11-s12
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The NIH Clinical Center implemented multiple testing protocols to facilitate early detection and isolation of SARS-CoV-2 infected patients and rooming-in family members (RIFMs). Beginning in February 2020, all symptomatic patients were tested; in March 2020, all patients were tested prior to aerosol-generating procedures (AGPs); and in May 2020, all patients and RIFMs were tested on admission. We sought to determine the value of SARS-CoV-2 testing practices in our hospital. Methods: Respiratory specimens collected March 2020 through June 2021 tested for SARS-CoV-2 by RT-PCR were reviewed, and corresponding patient clinical and demographic variables were collected. Repeated tests from SARS-CoV-2–positive persons were excluded from the data. Results associated with multiple testing indications were assigned the highest priority reason based on a predetermined hierarchy. Data were analyzed using the χ2 test and logistic regression. Results: Of 12,706 results from 5,704 patients, primary testing reasons were pre-AGP (n = 5,387, 43.0%), admission (n = 2,733; 21.8%), and symptomatic testing (n = 2,701; 21.6%). Overall, 159 tests (1.25%) were positive for SARS-CoV-2. Asymptomatic patients tested on admission were 1.8 times more likely to be positive than outpatients tested for any reason (P = .003) and 4.2 times more likely than asymptomatic inpatients tested prior to AGP (P = .003). Within asymptomatic pre-AGP testing, there was no difference between inpatients (0.46%) and outpatients (0.65%). Hispanic patients were 1.9 times more likely to be positive. (p Conclusions: At a hospital with a geographically broad referral base, admissions COVID-19 testing was far more fruitful than pre-AGP testing of inpatients. Pre-AGP used the most testing resources yet had the lowest yield. Admissions testing remains beneficial regardless of community transmission rates, while testing prior to AGP could be pared back when community rates of COVID-19 are low and redeployed when community rates rise. Conclusions: Our findings that Hispanic persons had higher risk and that transplant patients had lower risk of testing positive suggests differences in the extent to which each subgroup may have been able to shelter from COVID-19 in the community during this earlier phase of the pandemic. Keeping immunocompromised patients safe from COVID-19 while they undergo longitudinal care involves layered precautions in the hospital and in the community that must evolve in response to evidence and epidemiological trends.
Funding: None
Disclosures: None
Diagnostic/Microbiology
Temporal trends in urine-culture rates in the US acute-care hospitals, 2017–2020
- Sophia Kazakova, Natalie McCarthy, James Baggs, Kelly Hatfield, Babatunde Wolford, Babatunde Olubajo, John Jernigan, Sujan Reddy
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s12
-
- Article
-
- You have access Access
- Open access
- Export citation
-
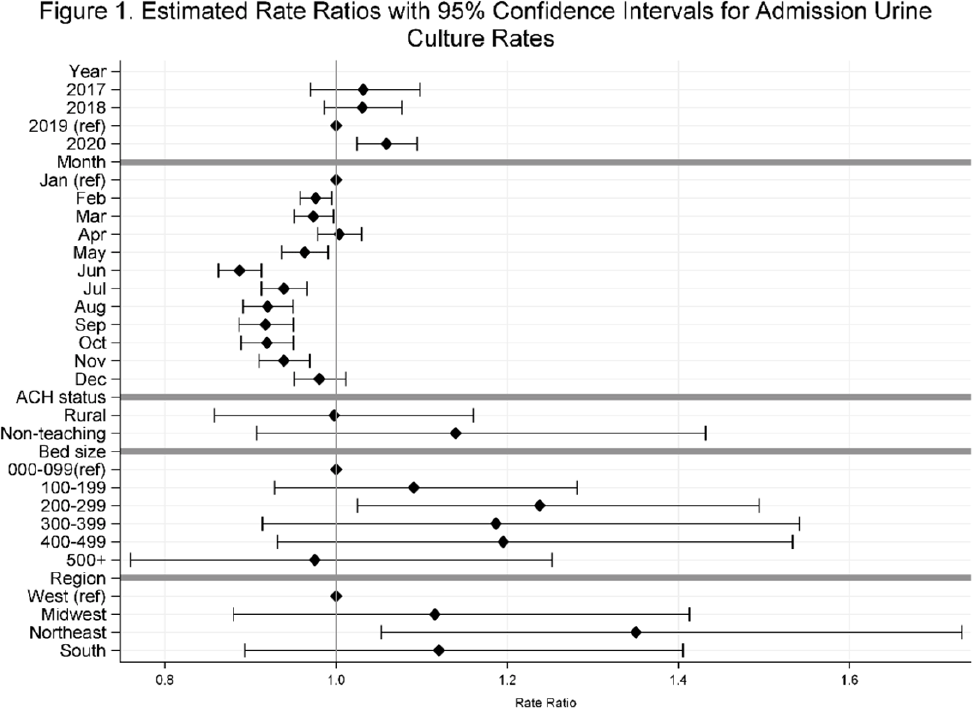
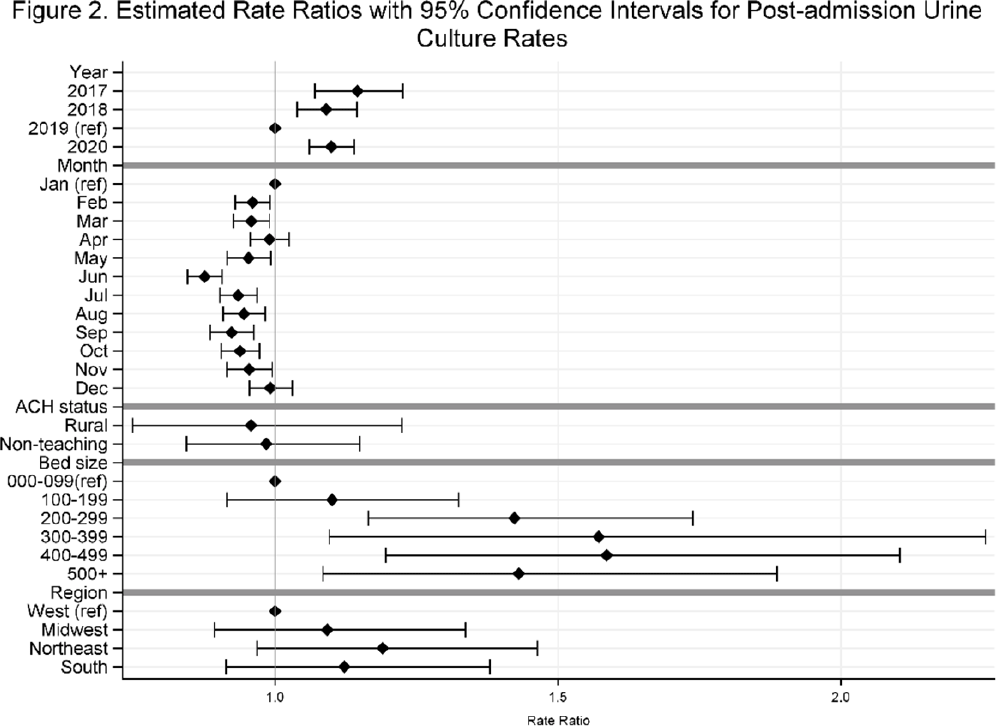
Background: Previously, we reported decreasing postadmission urine-culture rates in hospitalized patients between 2012 and 2017, indicating a possible decrease in hospital-onset urinary tract infections or changes in diagnostic practices in acute-care hospitals (ACHs). In this study, we re-evaluated the trends using more recent data from 2017–2020 to assess whether new trends in hospital urine-culturing practices had emerged. Method: We conducted a longitudinal analysis of monthly urine-culture rates using microbiology data from 355 ACHs participating in the Premier Healthcare Database in 2017–2020. All cultures from the urinary tract collected on or before day 3 were defined as admission urine cultures and those collected on day 4 or later were defined as postadmission urine cultures. We included discharges from months where a hospital reported at least 1 urine culture with microbiology and antimicrobial susceptibility test results. Annual estimates of rates of admission culture and postadmission urine-culture rates were assessed using general estimating equation models with a negative binomial distribution accounting for hospital-level clustering and adjusting for hospital bed size, teaching status, urban–rural designation, discharge month, and census division. Estimated rate for each year (2018, 2019, and 2020) was compared to previous year’s estimated rate using rate ratios (RRs) and 95% confidence intervals (CIs) generated through the multivariable GEE models. Results: From 2017 to 2020, we included 8.7 million discharges and 1,943,540 urine cultures, of which 299,013 (15.4%) were postadmission urine cultures. In 2017–2020, unadjusted admission culture rates were 20.0, 19.6, 17.9, and 18.2 per 100 discharges respectively; similarly, unadjusted postadmission urine-culture rates were 8.6, 7.8, 7.0, and 7.5 per 1,000 patient days. In the multivariable analysis, adjusting for hospital characteristics, no significant changes in admission urine-culture rates were detected during 2017–2019; however, in 2020, admission urine-culture rates increased 6% compared to 2019 (RR, 1.06; 95% CI, 1.02–1.09) (Fig. 1). Postadmission urine-culture rates decreased 4% in 2018 compared to 2017 (RR, 0.96; 95% CI, 0.91–0.99) and 8% in 2019 compared to 2018 (RR, 0.92; 95% CI, 0.87–0.96). In 2020, postadmission urine-culture rates increased 10% compared to 2019 (RR, 1.10; 95% CI, 1.06–1.14) (Fig. 2). Factors significantly associated with postadmission urine-culture rates included discharge month and hospital bed size. For admission urine cultures, discharge month was the only significant factor. Conclusions: Between 2017–2019, postadmission urine-culture rates continued a decreasing trend, while admission culture rates remained unchanged. However, in 2020 both admission and postadmission urine culture rates increased significantly in comparison to 2019.
Funding: None
Disclosures: None