Oral Presentation - Top Oral Award
COVID-19
Impact of a Black Physician Panel Discussion on Coronavirus Disease 2019 (COVID-19) Health Education
- Kenisha Evans, Jannel Lee-Allen, Donald Chinemelu Okoye, Lauren Uroda, Teena Chopra, Gini Ikwuezunma, Ijeoma Nnodim Opara, Hayley Thompson
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s1
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Coronavirus disease 19 (COVID-19) has infected >26 million Americans with >400,000 deaths. Both Pfizer and Moderna vaccines against severe acute respiratory coronavirus 2 (SARS-CoV-2) have demonstrated 95% efficacy; yet there has been growing vaccination hesitancy, especially within communities of color. To achieve herd immunity and quell the spread of SARS-CoV-2, several strategies need to be deployed. This community-based demonstration project highlights the impact of a panel of black physicians’ ability to increase vaccination intent within a social media campaign targeted toward a black audience, namely a live question-and-answer (Q&A) event on SARS-CoV-2 vaccines. Methods: The social media campaign included a flyer featuring the head shots and titles of 11 black physicians. The flyer showcased a live Q&A event via Zoom video conference software. Attendees were requested to preregister with their name, e-mail address, and country of origin. Results: The live Q&A event was attended by 251 viewers. Geographic distribution was predominantly within the United States (~88%), but a few attendees were from the United Kingdom (~11%) and Canada (<1%), Puerto Rico (<1%), and Paraguay (<1%). One hundred twenty eight questions and comments were received from attendees. Audience questions were categorized, with predominant topics as follows: Vaccine Safety, Medical Mistrust, Vaccine Safety in Pregnancy, Vaccine Efficacy, and Vaccine Development. The top five poll results revealed: 31% of audience members were not planning to vaccinate or were not sure about vaccination, but after the event are now planning to vaccinate; 93% believed their knowledge of the C19 vaccines had increased; 95% believed it was important that the information was presented by Black health experts; 90% reported that they trusted the information presented; and 96% rated the session as “good or excellent”. Conclusion: Our social media project is an example of one strategy healthcare professionals can utilize to positively influence local and global communities in the mitigation of the COVID-19 pandemic. Results of this project evaluation showed that viewers responded favorably, reporting increases in vaccine acceptance and knowledge. Most respondents also affirmed the importance of having black experts involved in communicating this information. COVID-19 has disproportionately affected black communities as a result of health inequities and institutionalized racism.1 The event amplifies the importance of utilizing social-media–based interventions and increasing black healthcare representation to aid infection control. 1. Jones C. Why Racism, Not Race, Is a Risk Factor for Dying of COVID-19. Scientific American June 12, 2020.
Funding: No
Disclosures: None
Coronavirus Disease 2019 (COVID-19) Admission Screening at a Tertiary-Care Center, Iowa 2020
- Mohammed Alsuhaibani, Takaaki Kobayashi, Alexandra Trannel, Stephanie Holley, Oluchi Abosi, Kyle Jenn, Holly Meacham, Lorinda Sheeler, William Etienne, Angie Dains, Mary Kukla, Emily Ward, Bradley Ford, Michael Edmond, Melanie Wellington, Daniel Diekema, Jorge Salinas
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s1
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Hospitalized patients may unknowingly carry severe acute respiratory coronavirus virus 2 (SARS-CoV-2), even if they are admitted for other reasons. Because SARS-CoV-2 may remain positive by reverse-transcriptase polymerase chain reaction (RT-PCR) for months after infection, patients with a positive result may not necessarily be infectious. We aimed to determine the frequency of SARS-CoV-2 infections in patients admitted for reasons unrelated to coronavirus disease 2019 (COVID-19). Methods: The University of Iowa Hospitals and Clinics is an 811-bed tertiary-care center. We use a nasopharyngeal SARS-CoV-2 RT-PCR to screen admitted patients without signs or symptoms compatible with COVID-19. Patients with positive tests undergo a repeat test to assess cycle threshold (Ct) value kinetics. We reviewed records for patients with positive RT-PCR screening admitted during July–October 2020. We used a combination of history, serologies, and RT-PCR Ct values to assess and qualify likelihood of infectiousness: (1) likely infectious, if Ct values were <29, or (2) likely not infectious, if 1 or both samples had Cts <30 with or without a positive SARS-CoV-2 antinucleocapsid IgG/IgM test or history of a positive result in the past 90 days. Contact tracing was only conducted for patients likely to be infectious. We describe the isolation duration and contact tracing data. Results: In total, 6,447 patients were tested on hospital admission for any reason (persons under investigation or admitted for reasons other than COVID-19). Of these, 240 (4%) had positive results, but 65 (27%) of these were admitted for reasons other than COVID-19. In total, 55 patients had Ct values available and were included in this analysis. The median age was 56 years (range, 0–91), 28 (51%) were male, and 12 (5%) were children. The most frequent admission syndromes were neurological (36%), gastrointestinal (16%), and trauma (16%). Our assessment revealed 23 likely infections (42%; 14 definite, 9 possible) and 32 cases likely not infectious (58%). The mean Ct for patients who were likely infectious was 22; it was 34 for patients who were likely not infectious. Mean duration of in-hospital isolation was 6 days for those who were likely infectious and 2 days for those who were likely not infectious. We detected 8 individuals (1 healthcare worker and 7 patients) who were exposed to a likely infectious patient. Conclusions: SARS-CoV-2 infection in patients hospitalized for other reasons was infrequent. An assessment of the likelihood of infectiousness including history, RT-PCR Cts, and serology may help prioritize patients in need of isolation and contact investigations.
Funding: No
Disclosures: None
Long-Term Care
Infection Hospitalization Trends Among US Home Healthcare Patients, 2013–2018
- Ashley Chastain, Jordan Harrison, Jingjing Shang, E. Yoko Furuya, Andrew Dick, Mark Sorbero, Patricia Stone
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s1-s2
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Infections are a frequent cause of hospital (re)admissions for older adults receiving home health care (HHC) in the United States. However, previous investigators have likely underestimated the prevalence of infections leading to hospitalization due to limitations of identifying infections using Outcome and Assessment Information Set (OASIS), the standardized assessment tool mandated for all Medicare-certified HHC agencies. By linking OASIS data with inpatient data from the Medicare Provider Analysis and Review (MedPAR) file, we were able to better quantify infection hospitalization trends and subsequent mortality among HHC patients. Method: After stratification (by census region, ownership, and urban or rural location) and random sampling, our data set consisted of 2,258,113 Medicare beneficiaries who received HHC services between January 1, 2013, and December 31, 2018, from 1,481 Medicare-certified HHC agencies. The 60-day HHC episodes were identified in OASIS. Hospital transfers reported in OASIS were linked with corresponding MedPAR records. Our outcomes of interest were (1) hospitalization with infection present on admission (POA); (2) hospitalization with infection as the primary cause; and (3) 30-day mortality following hospitalization with infection as the primary cause. We identified bacterial (including suspected) infections based on International Classification of Disease, Ninth Revision (ICD-9) and ICD-10 codes in MedPAR. We classified infections by site: respiratory, urinary tract, skin/soft tissue, intravenous catheter-related, and all (including other or unspecified infection site). We also identified sepsis diagnoses. Result: From 2013 through 2018, the percentage of 60-day HHC episodes with 1 or more hospital transfers ranged from 15% to 16%. Approximately half of all HHC patients hospitalized had an infection POA. Over the 6 years studied, infection (any type) was the primary cause of hospitalization in more than a quarter of all transfers (25.86%–27.57%). The percentage of hospitalizations due to sepsis increased from 7.51% in 2013 to 11.49% in 2018, whereas the percentage of hospitalizations due to respiratory, urinary tract, or skin/soft-tissue infections decreased (p <0.001). Thirty-day mortality following a transfer due to infection ranged from 14.14% in 2013 to 14.98% in 2018; mortality rates were highest following transfers caused by sepsis (23.14%-26.51%) and respiratory infections (13.07%-14.27%). Conclusion: HHC is an important source of post-acute care for those aging in place. Our findings demonstrate that infections are a persistent problem in HHC and are associated with substantial 30-day mortality, particularly following hospitalizations caused by sepsis, emphasizing the importance of infection prevention in HHC. Effective policies to promote best practices for infection prevention and control in the home environment are needed to mitigate infection risk.
Funding: No
Disclosures: None
Outbreaks
A Cluster of Coronavirus Disease 2019 (COVID-19) Cases on an Inpatient Hospital Unit Involving Multiple Modes of Transmission
- Kelsey Witherspoon, Michael Haden, Justin Smyer, Jennifer Flaherty, Heather Smith, Nora Colburn, Christina Liscynesky, James Allen, Shandra Day
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s2-s3
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The Ohio State University Wexner Medical Center identified a cluster of coronavirus disease 2019 (COVID-19) cases on an inpatient geriatric stroke care unit involving both patients and staff. The period of suspected severe acute respiratory coronavirus virus 2 (SARS-CoV-2) transmission and exposure on the unit was December 20, 2020, to January 1, 2021, with some patients and staff developing symptoms and testing positive within the 14 days thereafter. Methods: An epidemiologic investigation was conducted via chart review, staff interviews, and contact tracing to identify potential patient and staff linkages. All staff who worked on the unit were offered testing regardless of the presence of symptoms as well as all patients admitted during the outbreak period. Results: In total, 6 patients likely acquired COVID-19 in the hospital (HCA). An additional 6 patients admitted to the unit during the outbreak period subsequently tested positive but had other possible exposures outside the hospital (Fig. 1). One patient failed to undergo COVID-19 testing on admission but tested positive early in the cluster and is suspected to have contributed to patient to employee transmission. Moreover, 32 employees who worked on the unit in some capacity during this period tested positive, many of whom became symptomatic during their shifts. In addition, 18 employees elected for asymptomatic testing with 3 testing positive; these were included in the total. Some staff also identified potential community exposures. Additionally, staff reported an employee who was working while symptomatic with inconsistent mask use (index employee) early in the outbreak period. The index employee likely contributed to employee transmission but had no direct patient contact. Our epidemiologic investigation ultimately identified 12 employees felt to be linked to transmission based on significant, direct patient care provided to the patients within the outbreak period (Fig. 1). In addition, 3 employees had an exposure outside the hospital indicating likely community transmission. Conclusions: Transmission was felt to be multidirectional and included employee-to-employee, employee-to-patient, and patient-to-employee transmission in the setting of widespread community transmission. Interventions to stop transmission included widespread staff testing, staff auditing regarding temperature and symptom monitoring, and re-education on infection prevention practices. Particular focus was placed on appropriate PPE use including masking and eye protection, hand hygiene, and cleaning and disinfection practices throughout the unit. SARS-CoV-2 admission testing and limited visitation remain important strategies to minimize transmission in the hospital.
Figure 1.
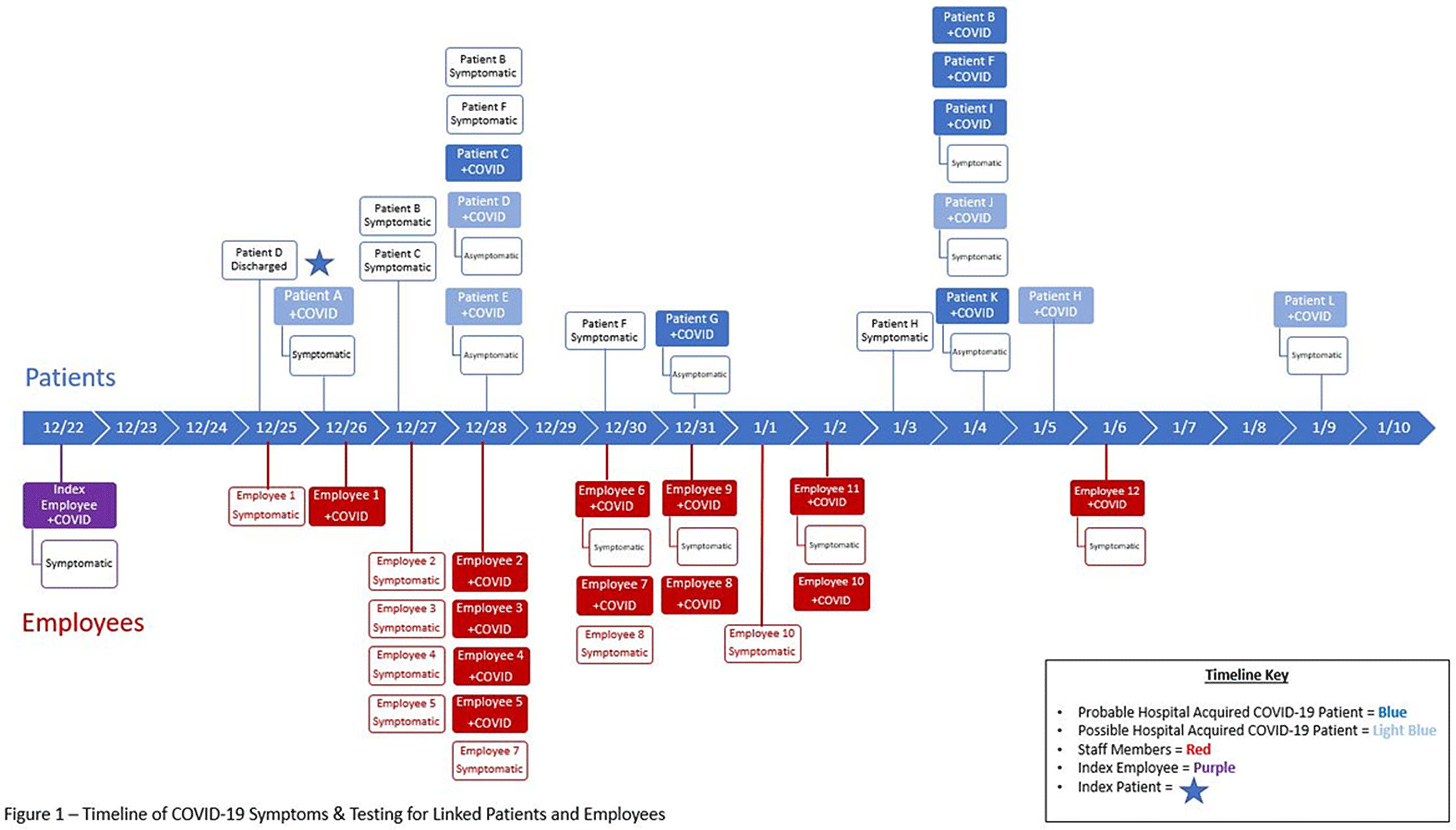
Funding: No
Disclosures: None
Surveillance/Public Health
Automation of Healthcare-Associated Infections (HAIs) Areas for Opportunity Through the Use of Technology
- Meri Pearson
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s3
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: A large healthcare system in Georgia implemented an enhanced electronic infection surveillance system that is incorporated into the electronic medical record (EMR). Prior to the implementation of this electronic infection surveillance system, the infection prevention (IP) team performed healthcare-associated infection (HAI) surveillance through a locally created system that did not interface with their EMR. Each HAI identified undergoes a robust analysis for opportunities depending on the infection type by manual abstraction from the EMR, which is stored in a spreadsheet. One disadvantage of this spreadsheet is that only 1 person can enter data at a time. The coronavirus disease 2019 (COVID-19) pandemic has presented many challenges for healthcare facilities including shifting resources from HAI prevention programs. These programs include the investigations performed to identify risk factors that can aid in preventing future infections. Due to the necessity to increase efficiency in the current pandemic, the IP team proposed using technology to automate our HAI investigation process. Method: The IP team and the business intelligence (BI) team met to determine whether data completed in the electronic infection surveillance system could flow into an interactive data visualization software that is currently used for other HAI prevention dashboards. The existing spreadsheet was reviewed to select variables essential for HAI investigations and for which the data existed in the EMR. The BI team worked to find the correct data tables within the EMR so that the data could automatically refresh daily in the data visualization software. Result: The BI team was able to correctly identify variables used in the previously manual HAI investigation process within the EMR to interface with the data visualization software. This automation of investigations allows quick analysis of trends and areas of opportunity to prevent future HAIs. Conclusion: This utilization of technology can be applied to other healthcare facilities with similar software systems to streamline IP workflows. The automation of quickly and efficiently recognizing areas of opportunity allows IPs more time to facilitate the prevention of HAIs in other ways.
Funding: No
Disclosures: None
Poster Presentation - Top Poster Award
Antibiotic Stewardship
Implementation of an Antibiotic Timeout at Veterans’ Affairs Medical Centers (VAMC): COVID-19 Facilitators and Barriers
- Jorie Butler, Joshua Judd, Cassie Goedken, Vanessa Stevens, Nui Brown, Michael Rubin, Matthew Goetz
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s3
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Effective stewardship strategies such as an “antibiotic timeout” to encourage prescriber reflection on the use of broad-spectrum antibiotics are critical to reduce the threat of multidrug-resistant organisms. We sought to understand the facilitators and barriers of the implementation of the Antibiotic Self-Stewardship Timeout Program (SSTOP), which used a template note integrated into the electronic health record system to guide decision making regarding anti- methicillin-resistant S. aureus (MRSA) therapy after 3 days of hospitalization. We conducted interviews at 10 Veterans’ Affairs medical centers (VAMCs) during the preimplementation period (N = 16 antibiotic stewards) and postimplementation (N = 13 antibiotic stewards) ~12 months after program initiation. Preimplementation interviews focused on current stewardship programs, whereas postimplementation interviews addressed the implementation process and corresponding challenges. We also directly asked about the impact of COVID-19 on stewardship activities at each facility. Interviews were transcribed and analyzed using consensus-based inductive and deductive coding. Codes were iteratively combined into barrier and facilitator groupings. Barriers identified in the preimplementation interviews included challenges with staffing, the difficulties of changing prescribing culture, and academic affiliates (eg, rotating physician trainees). Facilitators included intellectual support (eg, providers who understand the concept of stewardship), facility support, individual strengths of antibiotic stewards (eg, diplomacy, strong relationships with surgeons), and resources such as VA policies mandating stewardship. By the postimplementation phase, all sites reported a high volume of COVID-19 cases. Additional demands were placed on infectious disease providers who comprise the antibiotic stewardship teams, which complicated the implementation of SSTOP. Many barriers and facilitators mentioned were similar to those identified during preimplementation interviews. Staffing problems and specific providers not “getting it [stewardship activities]” continued, whereas facilitators centered around strong institutional support. Specific pandemic-related barriers included slow down or stoppage of stewardship activities including curbing of regular MRSA screening practices, halting weekly stewardship rounds, and delaying stewardship committee planning. Pandemic-specific staffing problems occurred due to the need for “all hands on deck” and challenges with staff working from home, as well as being pulled in multiple directions, (eg, writing COVID-19 policies). Furthermore, an increase in antibiotic use was also reported at sites during COVID-19 surges. Our findings indicate that SSTOP implementation met with barriers at most times; however, pandemic-specific barriers were particularly powerful. Sites with strong staffing resources were better equipped to deal with these challenges. Understanding how the program evolves with subsequent COVID-19 surges will be important to support the broad implementation of SSTOP.
Funding: No
Disclosures: None
Chronic Antibiotic Suppression for Nonstaphylococcal Prosthetic Joint Infections Treated with Debridement or Implant Retention
- Poorani Sekar, Rajeshwari Nair, Brice Beck, Bruce Alexander, Kelly Miell, Aaron Tande, Kimberly Dukes, Julia Friberg, Marin Schweizer, Andrew Pugely
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s3-s4
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Early postoperative and acute prosthetic joint infection (PJI) may be managed with debridement, antibiotics, and implant retention (DAIR). Among patients with nonstaphylococcal PJI, an initial 4–6-week course of intravenous or highly bioavailable oral antibiotics is recommended in the Infectious Diseases Society of America (IDSA) guidelines, with disagreement among committee members on the need for subsequent chronic oral antimicrobial suppression (CAS). We aimed to characterize patients with nonstaphylococcal PJI who received CAS and to compare them to those who did not receive CAS. Methods: This retrospective cohort study included patients admitted to Veterans’ Affairs (VA) hospitals from 2003 to 2017 who had a PJI caused by nonstaphylococcal bacteria, underwent DAIR, and received 4–6 weeks of antimicrobial treatment. PJI was defined by Musculoskeletal Infection Society (MSIS) 2011 criteria. CAS was defined as at least 6 months of oral antibiotics following initial treatment of the PJI. Patients were followed for 5 years after debridement. We used χ2 tests and t tests were used to compare patients who received CAS with those who did not receive CAS. Results: Overall, 561 patients had a nonstaphylococcal PJI treated with DAIR, and 80.6% of patients received CAS. The most common organisms causing PJI were streptococci. We detected no significant differences between patients who received CAS and those who did not receive CAS, except that modified Acute Physiology and Chronic Health Evaluation (mAPACHE) scores were higher among patients who did not receive CAS (Table 1). Conclusion: Patients not on CAS were more severely ill (by mAPACHE) than those on CAS. Otherwise, the 2 groups were not different. This finding was contrary to our hypothesis that patients with multiple comorbidities or higher mAPACHE scores would be more likely to get CAS. A future analysis will be conducted to assess treatment failure in both groups. We hope to find a specific cohort who may benefit from CAS and hope to deimplement CAS in others who may not benefit from it.
Funding: No
Disclosures: None
Table 1. 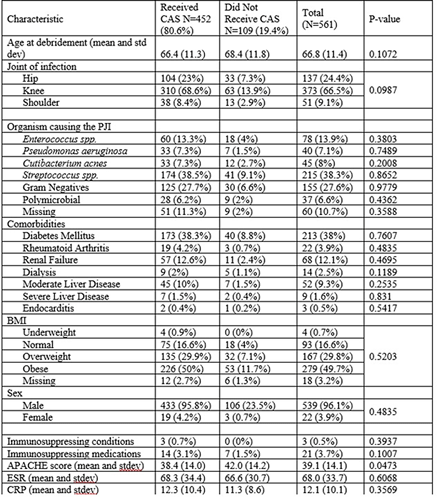
Reductions in Postdischarge Clostridioides difficile Infection after an Inpatient Health System Fluoroquinolone Stewardship
- K. Ashley Jones, Zanthia Wiley, Julianne Kubes, Mary Elizabeth Sexton, Benjamin Albrecht, Jesse Jacob, Jessica Howard-Anderson, Scott Fridkin, Udodirim Onwubiko
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s4
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Effective inpatient stewardship initiatives can improve antibiotic prescribing, but impact on outcomes like Clostridioides difficile infections (CDIs) is less apparent. However, the effect of inpatient stewardship efforts may extend to the postdischarge setting. We evaluated whether an intervention targeting inpatient fluoroquinolone (FQ) use in a large healthcare system reduced incidence of postdischarge CDI. Methods: In August 2019, 4 acute-care hospitals in a large healthcare system replaced standalone FQ orders with order sets containing decision support. Order sets redirected prescribers to syndrome order sets that prioritize alternative antibiotics. Monthly patient days (PDs) and antibiotic days of therapy (DOT) administered for FQs and NHSN-defined broad-spectrum hospital-onset (BS-HO) antibiotics were calculated using patient encounter data for the 23 months before and 13 months after the intervention (COVID-19 admissions in the previous 7 months). We evaluated hospital-onset CDI (HO-CDI) per 1,000 PD (defined as any positive test after hospital day 3) and 12-week postdischarge (PDC- CDI) per 100 discharges (any positive test within healthcare system <12 weeks after discharge). Interrupted time-series analysis using generalized estimating equation models with negative binomial link function was conducted; a sensitivity analysis with Medicare case-mix index (CMI) adjustment was also performed to control for differences after start of the COVID-19 pandemic. Results: Among 163,117 admissions, there were 683 HO-CDIs and 1,009 PDC-CDIs. Overall, FQ DOT per 1,000 PD decreased by 21% immediately after the intervention (level change; P < .05) and decreased at a consistent rate throughout the entire study period (−2% per month; P < .01) (Fig. 1). There was a nonsignificant 5% increase in BS-HO antibiotic use immediately after intervention and a continued increase in use after the intervention (0.3% per month; P = .37). HO-CDI rates were stable throughout the study period, with a nonsignificant level change decrease of 10% after the intervention. In contrast, there was a reversal in the trend in PDC-CDI rates from a 0.4% per month increase in the preintervention period to a 3% per month decrease in the postintervention period (P < .01). Sensitivity analysis with adjustment for facility-specific CMI produced similar results but with wider confidence intervals, as did an analysis with a distinct COVID-19 time point. Conclusion: Our systemwide intervention using order sets with decision support reduced inpatient FQ use by 21%. The intervention did not significantly reduce HO-CDI but significantly decreased the incidence of CDI within 12 weeks after discharge. Relying on outcome measures limited to inpatient setting may not reflect the full impact of inpatient stewardship efforts and incorporating postdischarge outcomes, such as CDI, should increasingly be considered.
Funding: No
Disclosures: None
Figure 1.
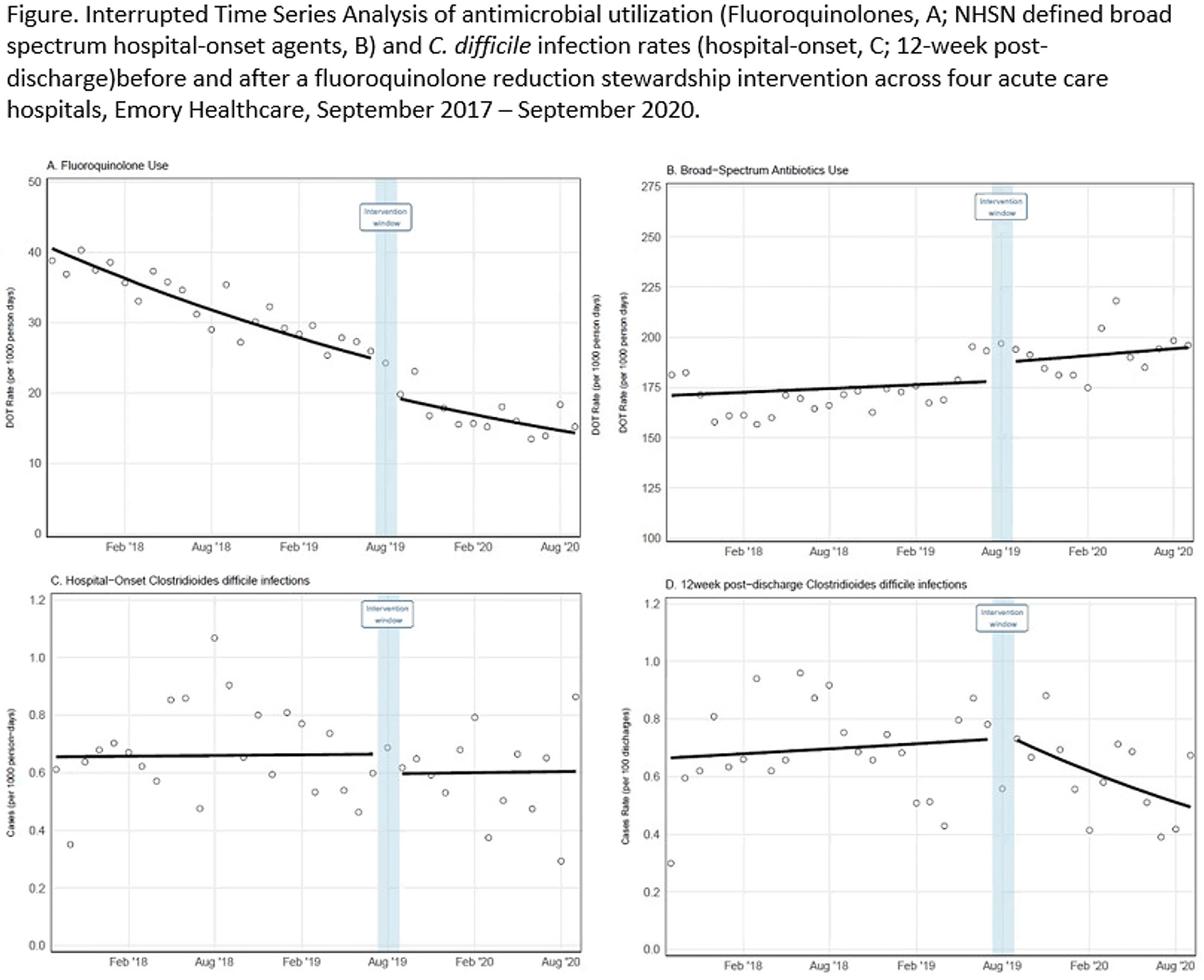
C. difficile
Efficacy and Safety of Investigational Microbiome Drug SER-109 for Treatment of Recurrent Clostridioides difficile Infection
- Barbara McGovern, Mathew Sims, Colleen Kraft, Elaine Wang, Kelly Brady, Christopher Ford, O’Brien Edward, Mary-Jane Lombardo, Jennifer Wortman, Kevin Litcofsky, Jennifer Mahoney, Christopher McChalicher, Jonathan Winkler, Sarah Garant, John Aunins, Matthew Henn, Lisa von Moltke
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antibiotics targeted against Clostridioides difficile bacteria are necessary, but insufficient, to achieve a durable clinical response because they have no effect on C. difficile spores that germinate within a disrupted microbiome. ECOSPOR-III evaluated SER-109, an investigational, biologically derived microbiome therapeutic of purified Firmicute spores for treatment of rCDI. Herein, we present the interim analysis in the ITT population at 8 and 12 weeks. Methods: Adults ≥18 years with rCDI (≥3 episodes in 12 months) were screened at 75 US and CAN sites. CDI was defined as ≥3 unformed stools per day for <48 hours with a positive C. difficile assay. After completion of 10–21 days of vancomycin or fidaxomicin, adults with symptom resolution were randomized 1:1 to SER-109 (4 capsules × 3 days) or matching placebo and stratified by age (≥ or <65 years) and antibiotic received. Primary objectives were safety and efficacy at 8 weeks. Primary efficacy endpoint was rCDI (recurrent toxin+ diarrhea requiring treatment); secondary endpoints included efficacy at 12 weeks after dosing. Results: Overall, 287 participants were screened and 182 were randomized (59.9% female; mean age, 65.5 years). The most common reason for screen failure was a negative C. difficile toxin assay. A significantly lower proportion of SER-109 participants had rCDI after dosing compared to placebo at week 8 (11.1% vs 41.3%, respectively; relative risk [RR], 0.27; 95% confidence interval [CI], 0.15–0.51; p-value <0.001). Efficacy rates were significantly higher with SER-109 vs placebo in both stratified age groups (Figure 1). SER-109 was well-tolerated with a safety profile similar to placebo. The most common treatment-emergent adverse events (TEAEs) were gastrointestinal and were mainly mild to moderate. No serious TEAEs, infections, deaths, or drug discontinuations were deemed related to study drug. Conclusions: SER-109, an oral live microbiome therapeutic, achieved high rates of sustained clinical response with a favorable safety profile. By enriching for Firmicute spores, SER-109 achieves high efficacy while mitigating risk of transmitting infectious agents, beyond donor screening alone. SER-109 represents a major paradigm shift in the clinical management of patients with recurrent CDI. Clinicaltrials.gov Identifier NCT03183128. These data were previously presented as a late breaker at American College of Gastroenterology 2020.
Funding: Seres Therapeutics
Disclosures: None
Figure 1.
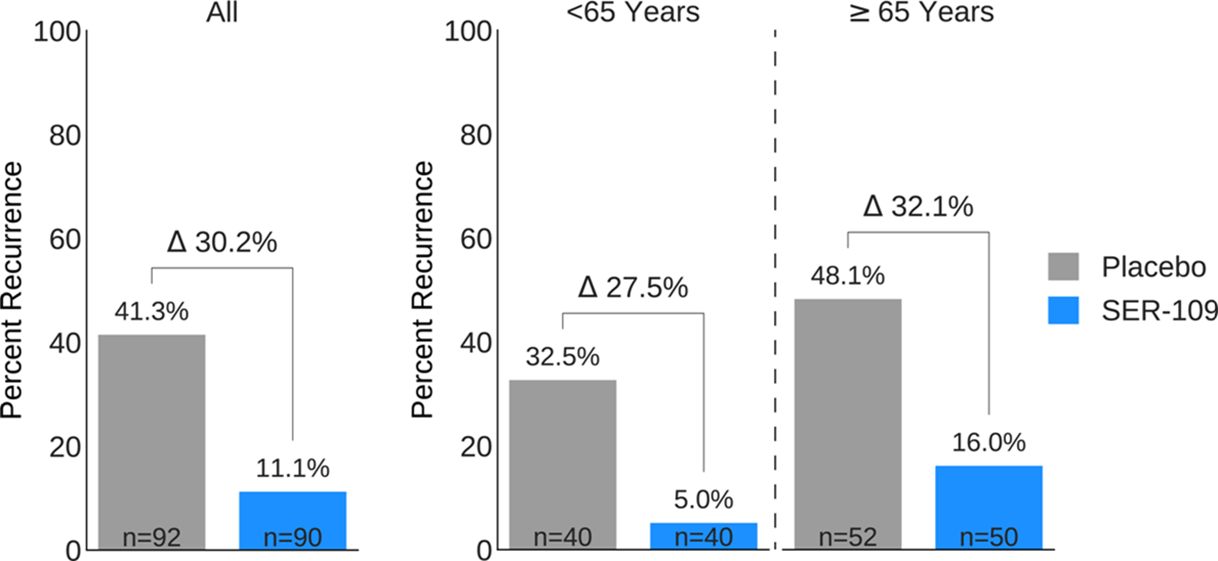
CAUTI
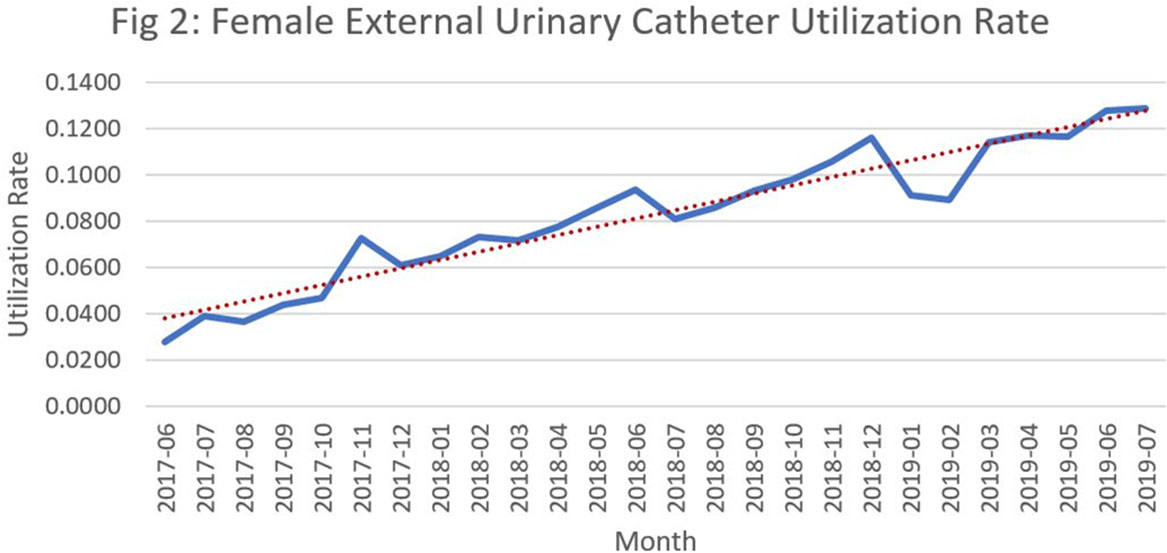
Impact of Female External Urinary Catheter on Indwelling Catheter Use and Catheter-Associated Urinary Tract Infection Rates
- Lea Monday, Geehan Suleyman, George Alangaden, Stephanie Schuldt, Catherine Jackman, Christine Halash
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s5
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Catheter-associated urinary tract infections (CED: TIs) are one of the most prevalent healthcare-associated infections. They can lead to bacteremia and increased length of stay, healthcare costs, and mortality. Indwelling urinary catheter (IUC) prevention bundles, nurse-driven removal protocols, and the use of external catheters can help reduce CED: TIs. However, female external urinary catheters (FEUCs) have only recently become widely available. FEUCs were introduced at our institution in July 2017. The purpose of this study was to evaluate the impact of FEUC on IUC utilization ratio and overall CED: TI rate in an 844-bed teaching hospital in southeastern Michigan. Methods: We retrospectively evaluated the utilization ratio of FEUCs (female FEUC days per patient days ×1,000) and female IUCs (IUC days per patient days ×1,000), and labia hospital-acquired pressure injury (HAPI) rate due to FEUC from July 2017 through June 2019. We compared the overall (male and female) CED: TI rate per 1,000 IUC days in the preintervention period (January 2016 to June 2017) to the postintervention period (July 2017 to June 2019). Results: In total, 4,013 FEUCs were placed during the intervention period. The utilization ratio of FEUC increased by 59% and the utilization ratio of female IUC decreased by 13% over the course of the 2 years. Only 1 HAPI was reported during the observation period at a rate of 0.025% (1 of 4,013). The overall CED: TI rate decreased from 1.60 to 1.40 (P = .372). Conclusion: Introduction of a FEUC was associated with a decrease in the IUC utilization ratio in female patients with minimal adverse events; however, there was no significant difference in the overall CED: TI rate.
Funding: No
Disclosures: None
Figure 1.
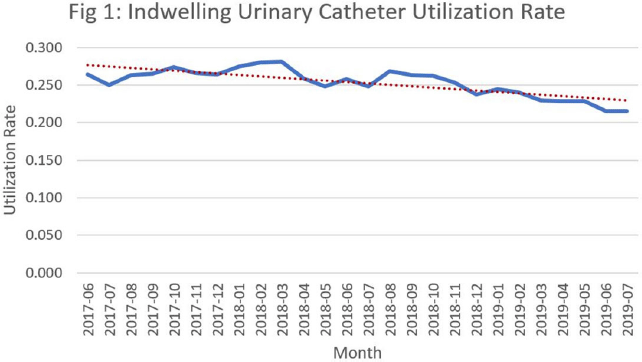
Figure 2.
Differences in the Clinical Outcome of Community-Acquired APN According to the Appropriateness of Antibiotic Use
- Bongyoung Kim, Choseok Yoon, Se Yoon Park, Ki Tae Kwon, Seong-yeol Ryu, Seong-Heon Wie, Hyun-uk Jo, Jieun Kim, Kyung-Wook Hong, Hye In Kim, Hyun ah Kim, Mi-Hee Kim, Mi-Hyun Bae, Yong-Hak Sohn, Jieun Kim, Yangsoon Lee, Hyunjoo Pai
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s6
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The purpose of this study was to find out the relationship between appropriateness of antibiotic prescription and clinical outcomes in patients with community-acquired acute pyelonephritis (CA-APN). Methods: A multicenter prospective cohort study was performed in 8 Korean hospitals from September 2017 to August 2018. All hospitalized patients aged ≥19 years diagnosed with CA-APN at admission were recruited. Pregnant women and patients with insufficient data were excluded. In addition, patients with prolonged hospitalization due to medical problems that were not associated with APN treatment were excluded. The appropriateness of empirical and definitive antibiotics was divided into “optimal,” “suboptimal,” and “inappropriate,” and optimal and suboptimal were regarded as appropriate antibiotic use. The standard for the classification of empirical antibiotics was defined reflecting the Korean national guideline for the antibiotic use in urinary tract infection 2018. The standards for the classification of definitive antibiotics were defined according to the result of in vitro susceptibility tests of causative organisms. Clinical outcomes including clinical failure (mortality or recurrence) rate, hospitalization days, and medical costs were compared between patients who were prescribed antibiotics appropriately and those who were prescribed them inappropriately. Results: In total, 397 and 318 patients were eligible for the analysis of the appropriateness of empirical and definitive antibiotics, respectively. Of these, 10 (2.5%) and 18 (5.7%) were inappropriately prescribed empirical and definitive antibiotics, respectively, and 28 (8.8%) were prescribed either empirical or definitive antibiotics inappropriately. Patients who were prescribed empirical antibiotics appropriately showed a lower mortality rate (0 vs 10%; P = .025), shorter hospitalization days (9 vs 12.5 days; P = .014), and lower medical costs (US$2,333 vs US$4,531; P = .007) compared to those who were prescribed empirical antibiotics “inappropriately.” In comparison, we detected no significant differences in clinical outcomes between patients who were prescribed definitive antibiotics appropriately and those who were prescribed definitive antibiotics inappropriately. Patients who were prescribed both empirical and definitive antibiotics appropriately showed a lower clinical failure rate (0.3 vs 7.1%; P = .021) and shorter hospitalization days (9 vs 10.5 days; P = .041) compared to those who were prescribed either empirical or definitive antibiotics inappropriately. Conclusions: Appropriate use of antibiotics leads patients with CA-APN to better clinical outcomes including fewer hospitalization days and lower medical costs.
Funding: No
Disclosures: None
Table 1. 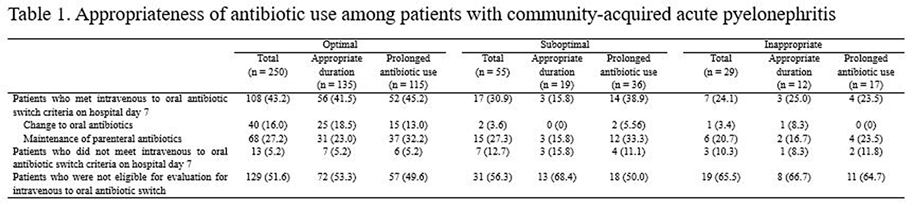
Table 2. 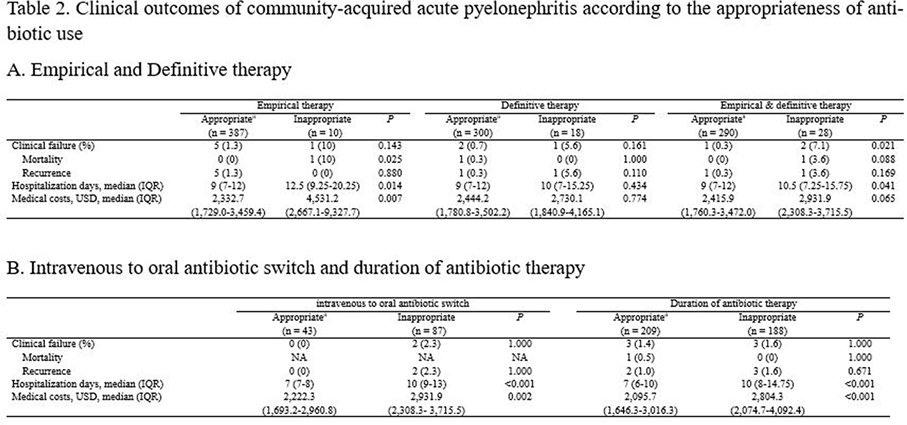
CLABSI
Effectiveness of a Multidisciplinary Team-Approach on Central-Line–Associated Bloodstream Infections
- Geehan Suleyman, Melissa Ahrens, Ann Keegan
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s7
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Although there has been a significant reduction in central-line–associated bloodstream infection (CLABSI) rates in the past decade with the implementation of evidence-based practices, an estimated 30,100 CLABSI occur each year in acute-care facilities. CLABSIs are associated with increased length of stay, cost, morbidity, and mortality, and they are preventable. In this study, we assessed the impact of a multidisciplinary team approach on CLABSI rates at a 319-bed teaching hospital in northwestern Ohio. Methods: In this before-and-after retrospective study, we compared the CLABSI rate per 1,000 central-line days, standardized infection ratio (SIR), and standardized utilization ratio (SUR) in the preintervention period (January 1, 2016, to December 31, 2018) to those of the intervention period (January 1, 2019, to December 31, 2020). Despite hospital-wide nursing education focusing on central-line maintenance in 2017, our SIR and SUR remained above the national benchmark. Starting in August 2018, we began to focus on insertion practices and physician education. An infection preventionist observed resident central-line insertion training and noted that there was no emphasis on infection prevention measures. There was a best practice knowledge gap. Thus, the indications for central-line use were updated, the insertion checklist was standardized, and the vascular access policy was revised to limit femoral and internal jugular vein use. Infection prevention training was provided to all providers involved in central-line insertions. Nurses were tasked with observing insertion of every central line and stopping the procedure if there is was an observed break in sterile technique. A central-line report listing indications and duration was developed and was sent to the nursing directors who assessed daily need with providers and prompted removal of unnecessary lines. The infection prevention medical director provided CLABSI prevention education to providers. Results: The CLABSI rate per 1,000 central-line days decreased from 0.90 in the preintervention period to 0.34 in the postintervention period, resulting in a 62% reduction in CLABSI rate. The SIR decreased from 0.886 to 0.323 (p-value <0.05), yielding a 64% reduction. The SUR also decreased from 1.156 to 0.874 (p-value <0.001) with a 24% reduction. Conclusion: A multidisciplinary team-approach with emphasis on standardized insertion checklist to ensure adherence to sterile technique and prompt removal of unnecessary central lines, and physician insertion training focusing on IP practices may potentially reduce CLABSI rates.
Funding: No
Disclosures: None
Sustainable Neonatal CLABSI Surveillance: Consensus Toward New Criteria in the Netherlands
- Ilja Heijting, Joost Hopman, Marije Hogeveen, Willem de Boode, Alma Tostmann, Tim Antonius
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s7-s8
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Group Name: Working Group on Neonatal Infectious Diseases of the Section of Neonatology of the Dutch Paediatric Society
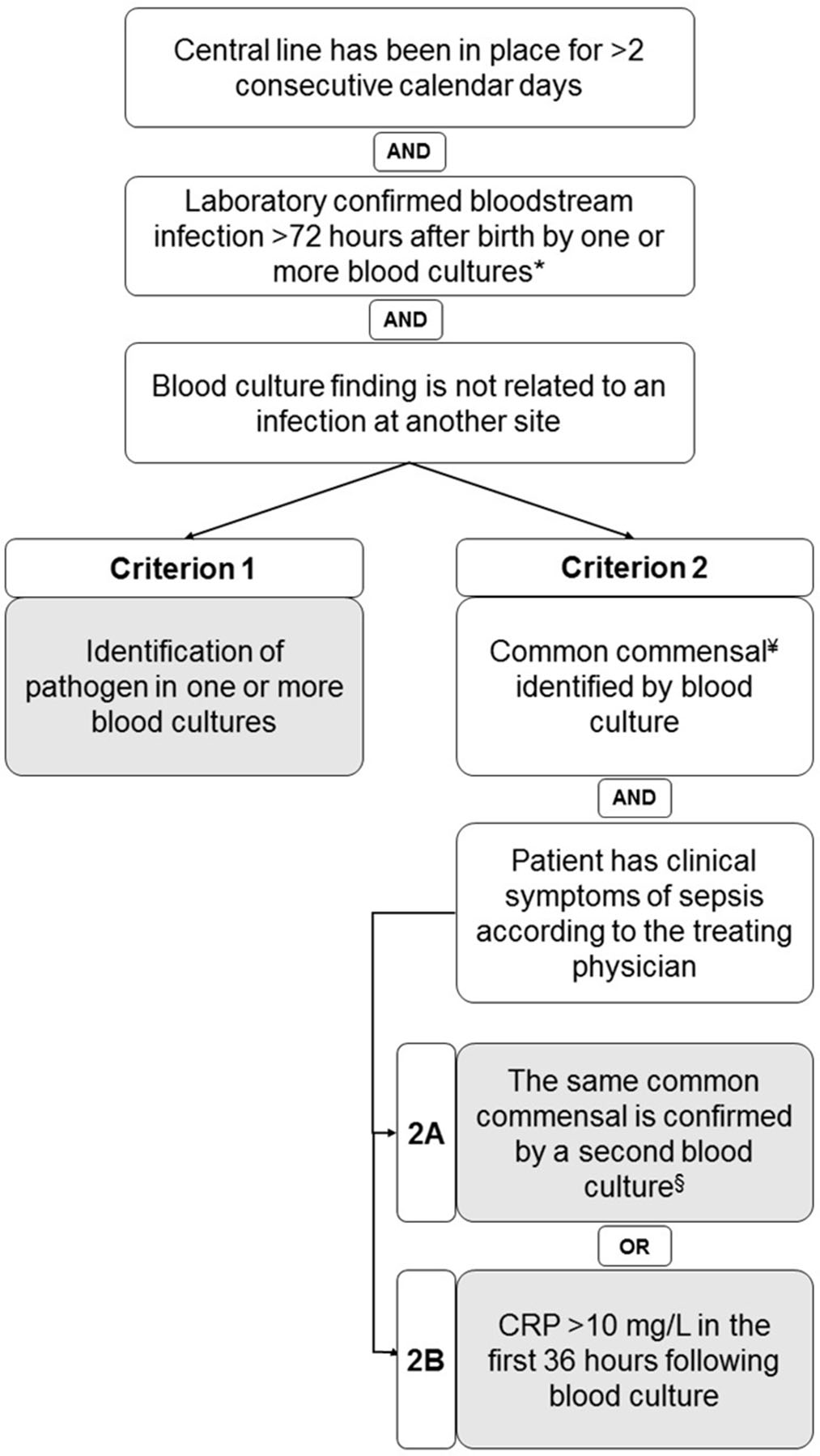
Background: Central-line–associated bloodstream infections (CLABSIs) are a main focus of infection prevention and control initiatives in neonatal care. Standardized surveillance of neonatal CLABSI enables intra- and interfacility comparisons, which can contribute to quality improvement. To date, there is no national registration system for CLABSI in neonatal care in the Netherlands. Across neonatal intensive care units (NICUs), several different sets of CLABSI criteria and surveillance methods are used for local monitoring of CLABSI incidence rates. To achieve standardized CLABSI surveillance, we conducted a consensus procedure with regard to nationwide neonatal CLABSI surveillance criteria. Method: A modified Delphi consensus procedure for the development of nationwide neonatal CLABSI surveillance criteria was performed between January 2016 and January 2017 in the Netherlands. An expert panel was formed by members of the Working Group on Neonatal Infectious Diseases of the Section of Neonatology of the Dutch Paediatric Society. The consensus procedure consisted of 3 expert panel rounds. Figure 1 shows a detailed description of the consensus procedure. Result: The expert panel achieved consensus on Dutch neonatal CLABSI surveillance criteria, which are summarized in Figure 2. Neonatal CLABSI is defined as a bloodstream infection occurring >72 hours after birth, associated with an indwelling central venous or arterial line and laboratory confirmed by 1 or more blood cultures. In addition, the blood culture finding should not be related to an infection at another site and one of the following criteria can be applied: (1) a bacterial or fungal pathogen is identified from 1 or more blood cultures; (2) the patient has clinical symptoms of sepsis and (2A) a common commensal is identified in 2 separate blood cultures or (2B) a common commensal is identified by 1 blood culture and C-reactive protein (CRP) level is >10 mg/L in the first 36 hours following blood culture collection. Conclusion: The newly developed Dutch neonatal CLABSI surveillance criteria are concise, are specific to the neonatal population, and comply with a single blood-culture policy in actual neonatal clinical practice. International agreement upon neonatal CLABSI surveillance criteria is needed to identify best practices for infection prevention and control.
Funding: No
Disclosures: None
Figure 1.
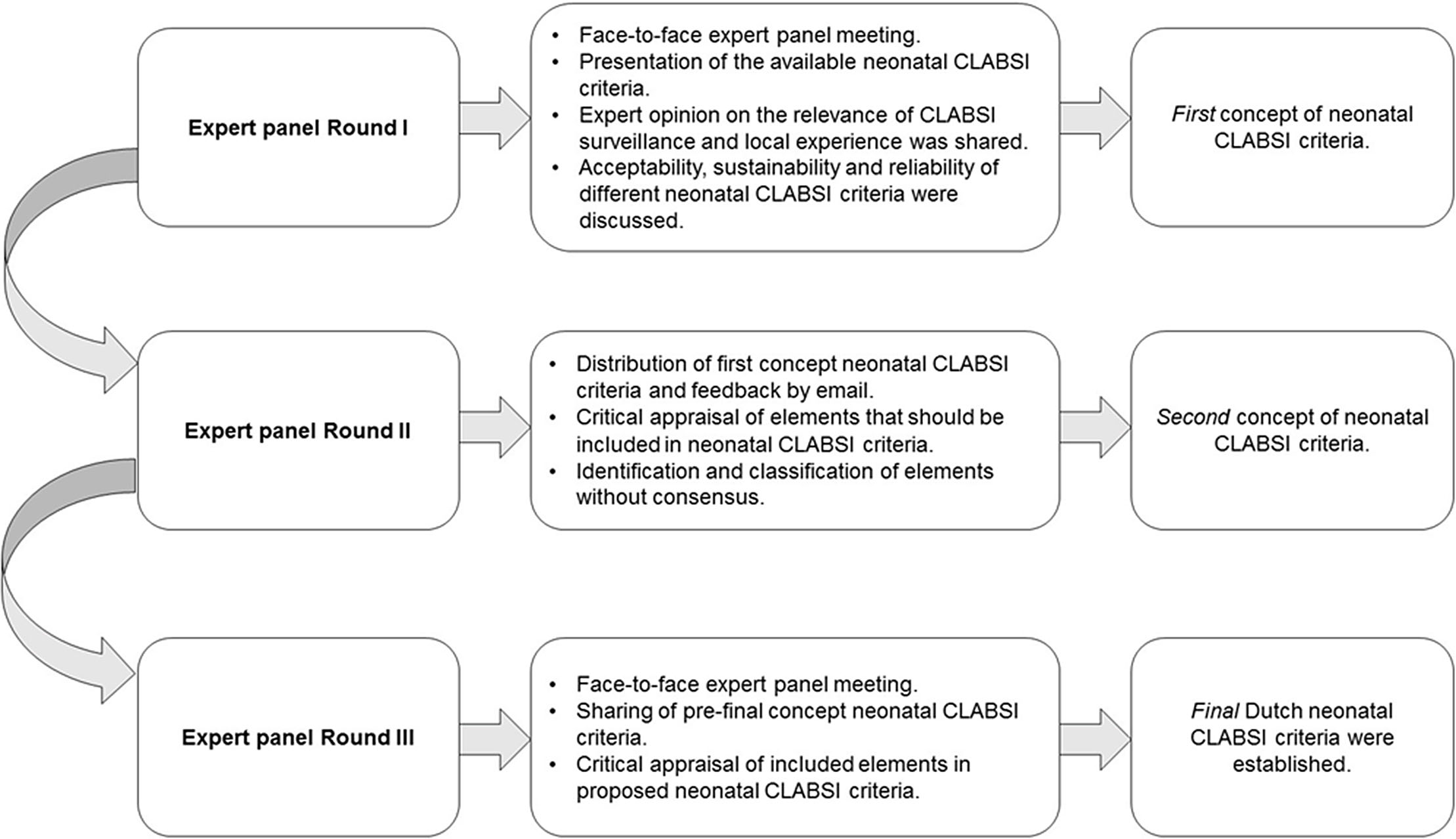
Figure 2.
COVID-19
Does Every Patient with a Positive SARS-CoV-2 RT-PCR Test Require Isolation? A Prospective Analysis
- Chanu Rhee, Meghan Baker, Sanjat Kanjilal, Robert Tucker, Vineeta Vaidya, Amy Badwaik, Elizabeth Mermel Blaeser, Cassie Coughlin, Jennifer Elloyan, Candace Hsieh, Meghan Holtzman, Ofelia Solem, Michael Klompas
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s8-s9
-
- Article
-
- You have access Access
- Open access
- Export citation
-
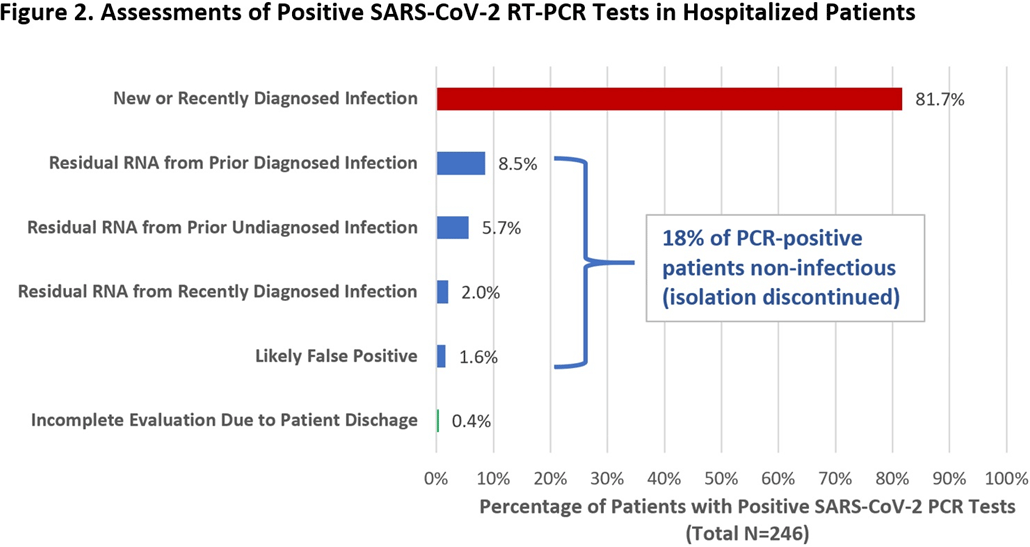
Group Name: CDC Prevention Epicenters Program Background: Reverse-transcriptase polymerase chain reaction (RT-PCR) tests are the reference standard for diagnosing SARS-CoV-2 infection, but false positives can occur and viral RNA may persist for weeks-to-months following recovery. Isolating such patients increases pressure on limited hospital resources and may impede care. Therefore, we quantified the percentage of patients who tested positive by RT-PCR yet were unlikely to be infectious and could be released from isolation. Methods: We prospectively identified all adults hospitalized at Brigham and Women’s Hospital (Boston, MA) who tested positive for SARS-CoV-2 by RT-PCR (primarily Hologic Panther Fusion or Cepheid Xpert platforms) between December 24, 2020, and January 24, 2021. Each case was assessed by infection control staff for possible discontinuation of isolation using an algorithm that incorporated the patient’s prior history of COVID-19, current symptoms, RT-PCR cycle threshold (Ct) values, repeat RT-PCR testing at least 24 hours later, and SARS-CoV-2 serologies (Figure 1). Results: Overall, 246 hospitalized patients (median age, 66 years [interquartile range, 50–74]; 131 [53.3%] male) tested positive for SARS-CoV-2 by RT-PCR during the study period. Of these, 201 (81.7%) were deemed new diagnoses of active disease on the basis of low Ct values and/or progressive symptoms. Moreover, 44 patients (17.9%) were deemed noninfectious: 35 (14.2%) had prior known resolved infections (n = 21) or unknown prior infection but positive serology (n = 14), high Ct values on initial testing, and negative or stably high Ct values on repeat testing. Also, 5 (2.0%) had recent infection but >10 days had passed since symptom onset and they were clinically improving. In addition, 4 (1.6%) results were deemed false positives based on lack of symptoms and at least 1 negative repeat RT-PCR test (Figure 2). One patient was asymptomatic with Ct value <35 but was discharged before further testing could be obtained. Among the 44 noninfectious patients, isolation was discontinued a median of 3 days (IQR, 2–4) after the first positive test. We did not identify any healthcare worker infections attributable to early discontinuation of isolation in these patients. Conclusions: During the winter COVID-19 second surge in Massachusetts, nearly 1 in 5 hospitalized patients who tested positive for SARS-CoV-2 by RT-PCR were deemed noninfectious and eligible for discontinuation of precautions. Most of these cases were consistent with residual RNA from prior known or undiagnosed infections. Active assessments of SARS-CoV-2 RT-PCR tests by infection control practitioners using clinical data, Ct values, repeat tests, and serologies can safely validate the release many patients from isolation and thereby conserve resources and facilitate patient care.
Funding: No
Disclosures: None
Figure 1.
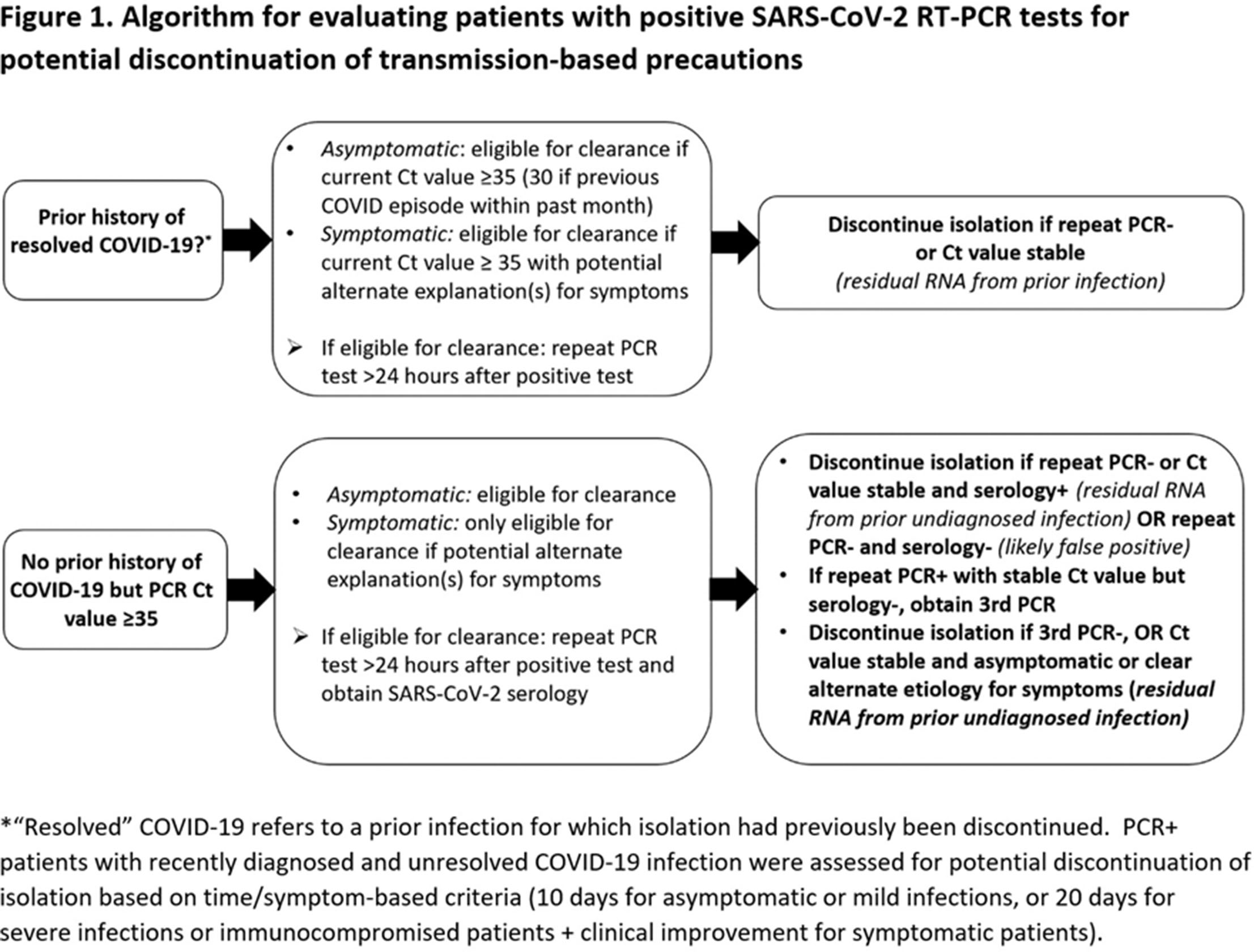
Figure 2.
Model Input and Optimization: Improving the Speed and Accuracy of Our COVID-19 Hospitalization Forecasts
- Sarah Rhea, Emily Hadley, Kasey Jones, Alexander Preiss, Marie Stoner, Caroline Kery, Peter Baumgartner, Alex Giarrocco
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s9
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: During the COVID-19 pandemic, public-health decision makers have increasingly relied on hospitalization forecasts that are routinely provided, accurate, and based on timely input data to inform pandemic planning. In North Carolina, we adapted an existing agent-based model (ABM) to produce 30-day hospitalization forecasts of COVID-19 and non–COVID-19 hospitalizations for use by public-health decision makers. We sought to continually improve model speed and accuracy during forecasting. Methods: The geospatially explicit ABM included movement of agents (ie, patients) among 104 short-term acute-care hospitals, 10 long-term acute-care hospitals, 421 licensed nursing homes, and the community in North Carolina. Agents were based on a synthetic population of North Carolina residents (ie, >10.4 million agents). We assigned SARS-CoV-2 infections to agents according to county-level susceptible, exposed, infectious, recovered (SEIR) models informed by reported COVID-19 cases by county. Agents’ COVID-19 severity and probability of hospitalization were determined using agent-specific characteristics (eg, age, comorbidities). During May 2020–December 2020, we produced weekly 30-day forecasts of intensive care unit (ICU) and non-ICU bed occupancy for COVID-19 agents and non–COVID-19 agents statewide and by region under a range of SARS-CoV-2 effective reproduction numbers. During the reporting period, we identified optimizations for faster results turnaround. We evaluated the incorporation of real-time hospital-level occupancy data at model initialization on forecast accuracy using mean absolute percent error (MAPE). Results: During May 2020–December 2020, we provided 31 weekly reports of 30-day hospitalization forecasts with a 1-day turnaround time. Reports included (1) raw and smoothed 7-day average values for 42 model output variables; (2) static visuals of ICU and non-ICU bed demand and capacity; and (3) an interactive Tableau workbook of hospital demand variables. Identifying code efficiencies reduced a single model runtime from ~100 seconds to 28 seconds. The use of cloud computing reduced simulation runtime from ~20 hours to 15 minutes. Across forecasts, the average MAPEs were 21.6% and 7.1% for ICU and non-ICU bed demand, respectively. By incorporating hospital-level occupancy data, we reduced the average MAPE to 6.5% for ICU bed demand and 3.9% for non-ICU bed demand, indicating improved accuracy. Conclusions: We adapted an ABM and continually improved it during COVID-19 forecasting by optimizing code and computing resources and including real-time hospital-level occupancy data. Planned SEIR model updates for enhanced forecasts include the addition of compartments for undocumented infections and recoveries as well as permission of reinfection from recovered compartments.
Funding: No
Disclosures: None
Retrospective Study on Personal Protective Equipment During Pandemic Link to Outbreak of Carbpenem-Resistant Enterobacteriace
- Kenisha Evans, Jennifer LeRose, Angela Beatriz Cruz, Lavina Jabbo, Teena Chopra
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s9-s10
-
- Article
-
- You have access Access
- Open access
- Export citation
-
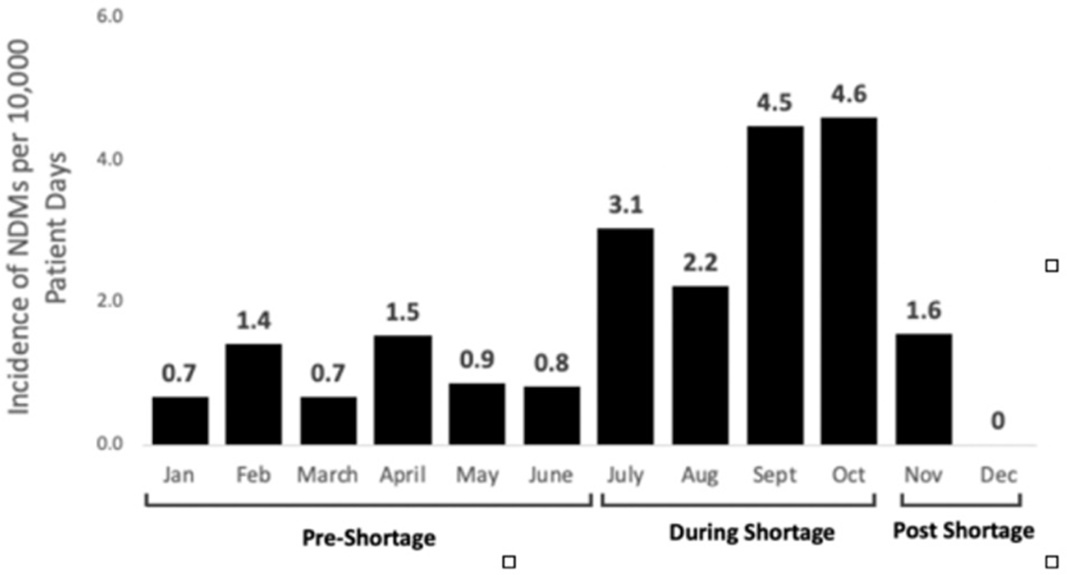
Background: In 2019, according to the Centers for Disease Control and Prevention, carbapenem-resistant Enterobacteriaceae (CRE), had cost the lives of >35,000 patients, particularly the most virulent plasmid-mediated New Delhi metallo-β-lactamase (NDM). Although healthcare systems normally have strict surveillance and infection control measures for CRE, the rapid emergence of novel SAR-CoV-2 and COVID-19 led to a shortage of personal protective equipment (PPE) and medical supplies. As a result, routine infection practices, such as contact precautions, were violated. Studies have shown this depletion and shift in resources compromised the control of infections such CRE leading to rising horizontal transmission. Method: A retrospective study was conducted at a tertiary healthcare system in Detroit, Michigan, to determine the impact of PPE shortages during the COVID-19 pandemic on NDM infection rates. The following periods were established during 2020 based on PPE availability: (1) pre-PPE shortage (January–June), (2) PPE shortage (July–October), and (3) post-PPE shortage (November–December). Rates of NDM per 10,000 patient days were compared between periods using the Wilcoxon signed rank-sum test. Isolates were confirmed resistant by NDM by molecular typing performed by the Michigan State Health Department. Patient characteristics were gathered by medical chart review and patient interviews by telephone. Results: Overall, the average rate of NDM infections was 1.82 ±1.5 per 10,000 patient days. Rates during the PPE shortage were significantly higher, averaging 3.6 ±1.1 cases per 10,000 patient days (P = .02). During this time, several infections occurred within patients on the same unit and/or patients with same treating team, suggesting possible horizontal transmission. Once PPE stock was replenished and isolation practices were reinstated, NDM infection rates decreased to 0.77 ±1.1 per 10,000 patient days. Conclusion: Control of CRE requires strategic planning with active surveillance, antimicrobial constructs, and infection control measures. The study illustrates that in times of crisis, such as the COVID-19 pandemic, the burden of effective infection control requires much more multidisciplinary efforts to prevent unintentional lapses in patient safety. A swift response by the state and local health departments at a tertiary-care healthcare center conveyed a positive mitigation of the highest clinical threats and decreased horizontal transmission of disease.
Funding: No
Disclosures: None
Figure 1.
A Cluster of Aspergillosis Associated with SARS-CoV-2
- Kerrie VerLee, Jim Codman, Russell Lampen, Chau Nguyen, Tunisia Peters, Greg Kruse, Derek VanderHorst, Doreen Marcinek, Molly Kane-Carbone
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s10
-
- Article
-
- You have access Access
- Open access
- Export citation
-
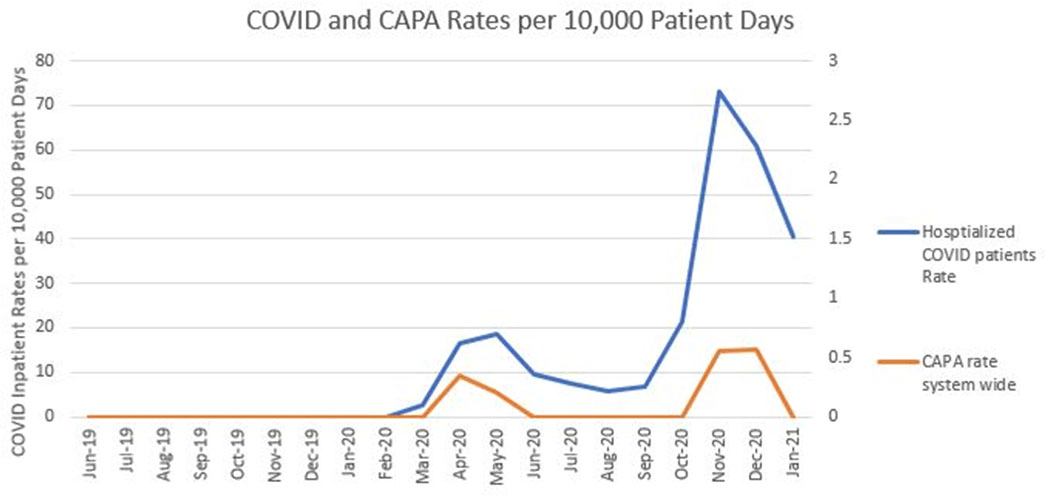
Background: Coronavirus disease 2019 (COVID-19) has demonstrated a variety of presentations and clinical complications, among them coinfection of pneumonia with the mold Aspergillus spp. Patients at risk for invasive disease include transplant recipients and those with prolonged neutropenia, immune disorders, cystic fibrosis, and steroid use. There have been recent descriptions of coronavirus disease–associated pulmonary aspergillosis (CAPA). An outbreak investigation into a cluster of Aspergillus fumigatus infections in a health system intensive care unit uncovered a community-onset (CO) increase in CAPA. Methods: A multidisciplinary outbreak investigation was conducted evaluating sources of contamination, completion of construction projects, and changes in clinical processes. Retrospective chart review was done for the prior 18 months and incidence density rates for Aspergillus infections from June 2019 through December 2020 were calculated per 10,000 patient days, stratified by unit, specimen source, and coinfection with COVID-19. Data were linked with all positive and negative COVID-19 tests performed by the health system’s regional laboratory from March to December 2020. Healthcare-onset (HO) classification was based on infections identified ≥7 days after admission. Statistical analysis was calculated with significance at p < 0.05. Results: Over the last 18 months, 82 patients were identified positive with Aspergillus cultures; of which 10 (12%) met CAPA definitions. Aspergillus fumigatus was the most common species and accounted for 62% of samples, followed by Aspergillus niger (17%). Median rates of HO Aspergillosis were 0.45 cases per 10,000 patient days, whereas the median total rates of infection were 1.97 cases per 10,000 patient days. Rates of CAPA coincided with COVID-19 hospitalization rates. In the spring and fall, surges of COVID-19, the rate ratio of CAPA to COVID hospitalized infections per 10,000 patient days, ranged from 0.006 to 0.015. Once CAPA infections were adjusted for, rates of CO Aspergillus remained high, whereas HO cases suggested baseline acquisition. Conclusion: This study outlines rates of CO aspergillosis as well as CAPA rates coinciding with the healthcare system’s spring and fall surges of COVID-19 hospitalizations. Despite the determination that this was not a hospital-acquired cluster, the investigation revealed some areas for opportunity in construction processes along with maintaining coverage of all patient supplies to reduce the risk of contamination.
Funding: No
Disclosures: None
Figure 1.
Molecular Epidemiology of Large COVID-19 Clusters at an Academic Medical Center, March–October 2020
- Takaaki Kobayashi, Miguel Ortiz, Stephanie Holley, William Etienne, Kyle Jenn, Oluchi Abosi, Holly Meacham, Lorinda Sheeler, Angie Dains, Mary Kukla, Alexandra Trannel, Alexandre Marra, Mohammed Alsuhaibani, Paul McCray, Stanley Perlman, Bradley Ford, Daniel Diekema, Melanie Wellington, Alejandro Pezzulo, Jorge Salinas
-
- Published online by Cambridge University Press:
- 29 July 2021, pp. s10-s11
-
- Article
-
- You have access Access
- Open access
- Export citation
-
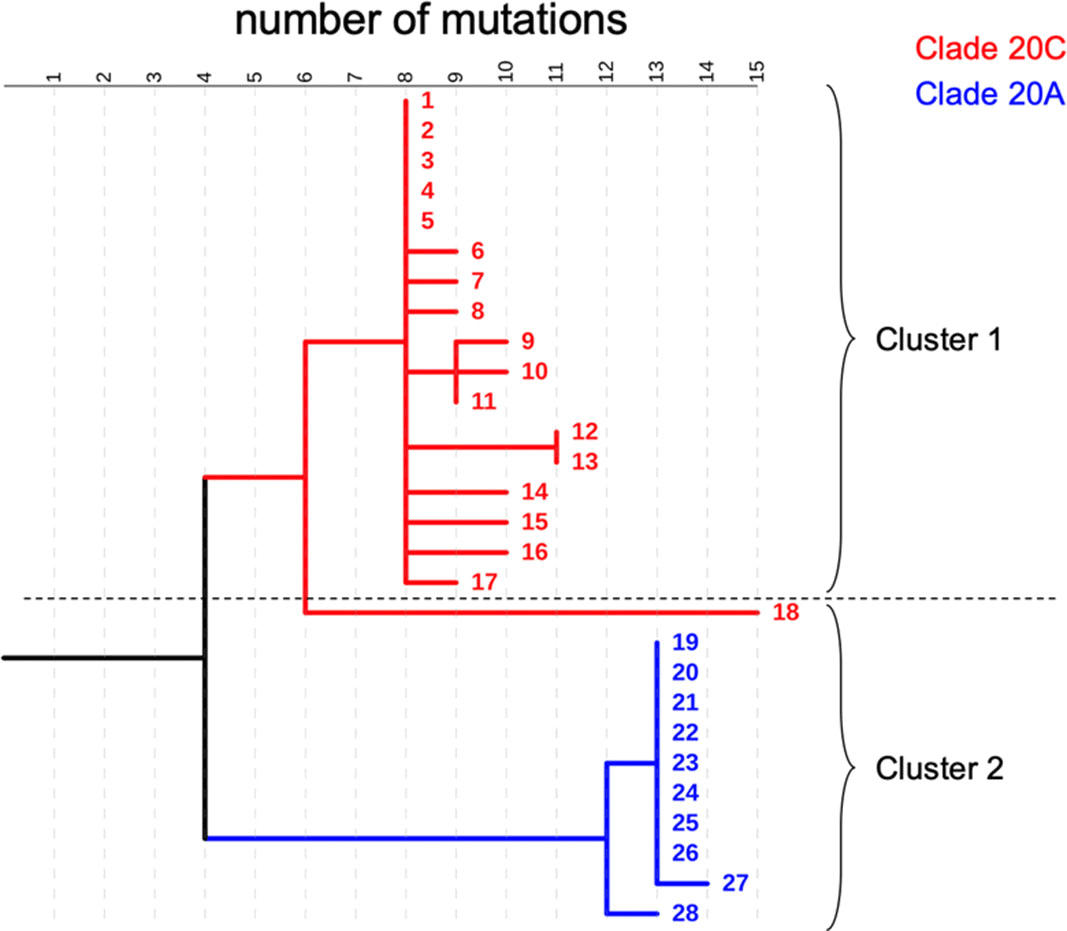
Background: COVID-19 in hospitalized patients may be the result of community acquisition or in-hospital transmission. Molecular epidemiology can help confirm hospital COVID-19 transmission and outbreaks. We describe large COVID-19 clusters identified in our hospital and apply molecular epidemiology to confirm outbreaks. Methods: The University of Iowa Hospitals and Clinics is an 811-bed academic medical center. We identified large clusters involving patients with hospital onset COVID-19 detected during March–October 2020. Large clusters included ≥10 individuals (patients, visitors, or HCWs) with a laboratory confirmed COVID-19 diagnosis (RT-PCR) and an epidemiologic link. Epidemiologic links were defined as hospitalization, work, or visiting in the same unit during the incubation or infectious period for the index case. Hospital onset was defined as a COVID-19 diagnosis ≥14 days from admission date. Admission screening has been conducted since May 2020 and serial testing (every 5 days) since July 2020. Nasopharyngeal swab specimens were retrieved for viral whole-genome sequencing (WGS). Cluster patients with a pairwise difference in ≤5 mutations were considered part of an outbreak. WGS was performed using Oxford Nanopore Technology and protocols from the ARTIC network. Results: We identified 2 large clusters involving patients with hospital-onset COVID-19. Cluster 1: 2 hospital-onset cases were identified in a medical-surgical unit in June 2020. Source and contact tracing revealed 4 additional patients, 1 visitor, and 13 employees with COVID-19. Median age for patients was 62 (range, 38–79), and all were male. In total, 17 samples (6 patients, 1 visitor, and 10 HCWs) were available for WGS. Cluster 2: A hospital-onset case was identified via serial testing in a non–COVID-19 intensive care unit in September 2020. Source investigation, contact tracing, and serial testing revealed 3 additional patients, and 8 HCWs. One HCW also had a community exposure. Patient median age was 60 years (range, 48–68) and all were male. In total, 11 samples (4 patients and 7 HCWs) were sequenced. Using WGS, cluster 1 was confirmed to be an outbreak: WGS showed 0–5 mutations in between samples. Cluster 2 was also an outbreak: WGS showed less diversity (0–3 mutations) and ruled out the HCW with a community exposure (20 mutations of difference). Conclusion: Whole-genome sequencing confirmed the outbreaks identified using classic epidemiologic methods. Serial testing allowed for early outbreak detection. Early outbreak detection and implementation of control measures may decrease outbreak size and genetic diversity.
Funding: No
Disclosures: None
Figure 1.
COVID-19 Vaccination of HCWs in the First Phase of a Large-Scale Mass Vaccination Program within a Healthcare System
- Kimberly Korwek, E. Jackie Blanchard, Julia Moody, Katherine Lange, Ryan Sledge, Maria Benedetti, Kenneth Sands
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s11
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The approval of the first SARS-COV-2 vaccines for COVID-19 were accompanied by unprecedented efforts to provide vaccination to healthcare workers and first responders. More information about vaccine uptake in this group is needed to better refine and target educational messaging. Methods: HCA Healthcare used federal guidance and internal experience to create a systemwide mass vaccination strategy. A closed point-of-dispensing (POD) model was developed and implemented. The previously developed enterprise-wide emergency operations strategy was adapted and implemented, which allowed for rapid development of communications and operational processes. A tiering strategy based on recommendations from the National Academies was used in conjunction with human resources data to determine vaccine eligibility for the first phase of vaccination. A comprehensive data and reporting strategy was built to connect human resources and vaccine consent data for tracking vaccination rates across the system. Results: Vaccination of employed and affiliated colleagues began December 15, 2020, and was made available based on state-level release of tiers. Within the first 6 weeks, in total, 203,544 individuals were eligible for vaccine based on these criteria. Of these, 181,282 (89.1%) consented to and received vaccine, 19,788 (9.7%) declined, and 2,474 (1.2%) indicated that they had already been vaccinated. Of those eligible, the highest acceptance of vaccine was among the job codes of specialists and professionals (n = 7,914 total, 100% consent), providers (n = 23,335, 99.6%,), and physicians (n = 3,218, 98.4%). Vaccine was most likely to be declined among job codes of clerical and other administrative (n = 12,889 total, 80.1% consent), clinical specialists and professionals (n = 22,853, 81.0%,) and aides, orderlies and technicians (n = 17,803, 82.6%,). Registered nurses made up the largest eligible population (n = 56,793), and 89.5% of those eligible consented to receive vaccination. Average age among those who consented was slightly older (48.3 years) than those that declined (44.7 years), as was length of employment tenure (6.9 vs 5.0 years). Conclusion: A large-scale, closed POD, mass vaccination program was able to vaccinate nearly 200,000 healthcare workers for SARS-CoV-2 in 6 weeks. This program was implemented in acute-care sites across 20 different US states, and it was able to meet the various state-level requirements for management of processes, product, and required reporting. The development of a standardized strategy and custom, centralized monitoring and reporting facilitated insight into the characteristics of early vaccine adopters versus those who decline vaccination. These data can aid in the refining and targeting of educational materials and messaging about the SARS-CoV-2 vaccine.
Funding: No
Disclosures: None
Disinfection/Sterilization
High-Level Disinfection in Ambulatory Care: Overcoming the Barriers of a Decentralized System through Auditing and Education
- Sonja Rivera Saenz
-
- Published online by Cambridge University Press:
- 29 July 2021, p. s11
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: High-level disinfection (HLD) of semicritical instruments in a multispecialty ambulatory care network has the potential for increased risk due to the decentralized instrument reprocessing and lack of a sterile processing department. Attention to HLD practices is an important part of device-borne outbreak prevention. Method: An HLD database was developed to identify specific departments and locations where HLD occurred across a 30-medical practice ambulatory care network in eastern Massachusetts, which included otolaryngology, urology, endoscopy, and obstetrics/gynecology departments. Based on qualitative feedback from managers and reprocessing staff, this database centralized information that included the supply inventory including manufacturer and model information, HLD methodology, standard work, and listing of competency evaluations. The infection control team then led audits to directly observe compliance with instrument reprocessing and a monthly-driven HLD calendar was developed to enforce annual competencies. Result: The results of the audits demonstrated variability across departments with gaps in precleaning, transportation of used instruments, the dilution of enzymatic cleaner, and maintenance of quality control logs. Given the uniqueness of shape and size of various ambulatory locations, proper storage and separation between clean and dirty spaces were common pitfalls. Auditing also revealed different levels of staff understanding of standard work and variable inventory management. Centralized education sessions held jointly by the infection control team and various manufacturers for the reprocessing staff helped to create and reinforce best practices. Conclusion: Decentralized HLD that occurs across multiple ambulatory care sites led to gaps in instrument reprocessing and unique challenges due to variable geography of sites, physical space constraints, and an independent approach to procuring medical supplies. Through the auditing and feedback of all areas that perform HLD, an effective and sustainable strategy was created to ensure practice improvement. Streamlining standard work, seeking direct input from frontline staff, and collective educational events were critical to our success in the ambulatory setting.
Funding: No
Disclosures: None















