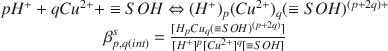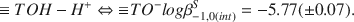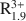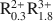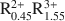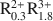Volume 41 - August 1993
Research Article
The Near-Infrared Combination Band Frequencies of Dioctahedral Smectites, Micas, and Illites
- James L. Post, Paul N. Noble
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 639-644
-
- Article
- Export citation
-
The highest frequency near-infrared (NIR) combination bands for specimens of four species of mica—montmorillonite-beidellite, illite, chlorite, and kaolinite—were correlated with respect to Al2O3 content. A direct linear correlation was found between the combination band positions and the Al2O3 contents of the montmorillonite-beidellite series, which may be given as: ν̄ cm−1 = (5.38 ± 0.04) (% Al2O3) + (4412.8 ± 0.9). A similar linear correlation for muscovite is: ν̄ cm−1 = (6.10 ± 0.25) (% Al2O3) + (4434.1 ± 8.3).
Possible NIR band interferences are shown for different mineral mixtures, along with the correlation of different illites with muscovite. No combination bands were found in the frequency region 4425 cm−1 to 4625 cm−1 for specimens in which the Al2O3 content was only in the tetrahedral layer sites.
Introduction
Geothermometry and Geochronology Using Clay Minerals—An Introduction
- Eric Eslinger, J. Reed Glasmann
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 117-118
-
- Article
- Export citation
Research Article
40Ar/39Ar Analyses of Authigenic Muscovite, Timing of Stylolitization, and Implications for Pressure Solution Mechanisms: Jurassic Norphlet Formation, Offshore Alabama
- Andrew R. Thomas, William M. Dahl, Chris M. Hall, Derek York
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 269-279
-
- Article
- Export citation
-
Three authigenic muscovite morphologies are associated with Norphlet Formation stylolitization observed in the Texaco Mobile Area Block 872 #1 well: l)large crystals of 1M muscovite, which grew in the stylolites with their c-axes parallel to the plane of maximum compressive stress; 2) fine-grained bundles of muscovite that occur as pore-fillings near stylolites; and 3) pods of fine-grained muscovite that exist within stylolite insoluble residue and that were precipitated as pore-filling muscovite before the host sandstone pressolved.
The population of large crystals of 1M muscovite grew at 51 ± 9 Ma, pore-filling muscovites precipitated at 77 ± 22 Ma, and muscovite pods have ages of 86 ± 16 Ma, as indicated by 40Ar/39Ar laser fusion. Apparent ages indicate that stylolitization was coincident with the beginning of organic maturation Zone 5 and could be the product of reservoir fluid pressure fluctuations induced by gas leakage. The lower Smackover Formation source/seal rock, acting as a pressure relief valve, could have been compromised by microfractures occurring during hydrocarbon generation and expulsion. Decreases in reservoir fluid pressure would have acted upon the sandstone framework by increasing the effective overburden pressure, thus making the rock more susceptible to pressure solution.
Stylolite frequency and quartz cement volume increase in the finer grained portion of the conventional core. Quartz cement volume correlates inversely to percent sandstone porosity. Apparent muscovite ages indicate that stylolitization occurred after hydrocarbon migration. Silica mobility was limited because pressure solution mineral products were precipitated from within grain films of irreducible water within the sandstone.
Stylolitization of quartz grains accounts for a minimum of 34% of the quartz cement in the upper cored section of the Norphlet Formation and minimum of 17% of the quartz cement in the lower cored Norphlet Formation. Quartz cement volumes are based on stylolite insoluble residue thickness and weight measurements of pyrobitumen within and nearby the insoluble residue seams. Stylolitization of K-feldspar and precipitation of muscovite can release additional silica which may have precipitated as quartz cement.
Analyses of Palygorskites and Associated Clays from the Jbel Rhassoul (Morocco): Chemical Characteristics and Origin of Formation
- Azzedine Chahi, Joelle Duplay, Jacques Lucas
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 401-411
-
- Article
- Export citation
-
The “Formation Rouge” from the Jbel Rhassoul in Morocco is composed of detrital sediments which have a lacustrine origin. The clays contained in the less than 2 µm fraction of the sediments are detrital phengites and illites, illite/smectites or smectites, and palygorskite. Due to the presence of well preserved long fibers, the palygorskite could not have been transported. They are authigenic and must have formed directly by precipitation from solutions rich in Mg and Al. The detrital illites are impoverished in K and tetrahedral Al. The illite/smectites or smectites, on the contrary, are K-rich but have a low tetrahedral charge. They are also richer in Mg and Fe and have a different crystal size, composition, and crystallinity from the illites. They most probably formed by crystallization, similar to the palygorskites, directly from the solution. The Al could have been provided by the detrital illite, which may have been unstable in an alkaline environment and released K and Al to the solutions.
Adsorption and Spectroscopic Studies on the Interactions of Cobalt(III) Chelates with Clays
- Masami Kaneyoshi, Akihiko Yamagishi, Masahiro Tanaguchi, Akiko Aramata
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 1-6
-
- Article
- Export citation
-
The interactions of the following three kinds of racemic and enantiomeric cobalt(III) chelates with montmorillonite and saponite are studied: [Co(en)3]3+ (en = ethylenediamine), [Co(diNOsar)]3+ (diNOsar = (1,8-dinitro-3,6,10,13,16,19-hexaazabicyclo[6,6,6]-eicosane)cobalt(III))and [Co(diAMsar)]3+ (diAMsar = (1,8-diamino-3,6,10,13,16,19-hexaazabicyclo-[6,6,6]eicosane)-cobalt(III)). At neutral pH, these complexes are adsorbed as a trivalent cation up to 90%–100% of the cation exchange capacity of a clay. No difference is observed in the maximum adsorption amount between the racemic and enantiomeric isomers. The basal spacings of the clay-chelate adducts are determined by the X-ray diffraction measurements of non-oriented powder samples: 14.3 Å for [Co(en)3]3+ montmorillonite, 16.5 Å for [Co(diNOsar)]3+ montmorillonite, and 16.9 Å for [Co(diAMsar)]3+ montmorillonite. The results imply that the chelates form a monolayer in the interlayer space. From the one-dimensional Fourier analyses of the diffraction pattern of [Co(diNOsar)]3+ montmorillonite, the chelate is concluded to be adsorbed with its three-fold symmetry axis in parallel with the layer surface. This is in contrast with the previous results of [Ru(phen)3]2+ and [Ru(bpy)3]2+, which are adsorbed with their three-fold symmetry axes perpendicular to the surface. The conclusion is consistent with the angular dependence of the infrared absorption spectrum of the film of the adduct.
Zeolites in Pyroclastic Deposits in Southeastern Tenerife (Canary Islands)
- J. E. Garcia Hernandez, J. S. Notario del Pino, M. M. Gonzalez Martin, F. Hernan Reguera, J. A. Rodriguez Losada
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 521-526
-
- Article
- Export citation
-
The chemical and the mineralogical composition of a group of pumiceous tuffs associated with recent salic volcanic episodes from Tenerife (Canary Islands) have been studied. The investigation focused on the two main types of pyroclastic deposits of the zone: ash-flows and ash-falls. The samples can be classified chemically as trachytic and phonolitic rocks with an intermediate silica content and a high percentage of alkali cations (Na+ and K+). The mineralogical composition, determined by X-ray diffraction, scanning electron microscopy, and optical microscopy, shows the occurrence of zeolites (mainly phillipsite, with lesser chabazite and analcime), associated with the parent glass. K-feldspar (sanidine) and calcite are accessory minerals. Zeolites are significantly more abundant in the ash-flow deposits. Zeolite formation by hydrothermal weathering in closed-system conditions varies according to the nature and the origin of the pyroclastic deposits. Tenerife phillipsites differ from typical diagenetic, lacustrine, and deep-sea phillipsites, both in chemical and mineralogical features. Alkali cations exceed divalent cations in the unit-cell that, assuming a monoclinic symmetry, has the following parameters: a = 8.46–10.55 Å, b = 14.21–14.40 Å, c = 7.80–8.70 Å, and β = 105°–110°.
Considerations and Applications of the Illite/Smectite Geothermometer in Hydrocarbon-Bearing Rocks of Miocene to Mississippian Age
- Richard M. Pollastro
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 119-133
-
- Article
- Export citation
-
Empirical relationships between clay mineral transformations and temperature provide a basis for the use of clay minerals as geothermometers. Clay-mineral geothermometry has been applied mainly to diagenetic, hydrothermal, and contact- and burial-metamorphic settings to better understand the thermal histories of migrating fluids, hydrocarbon source beds, and ore and mineral formation.
Quantitatively, the most important diagenetic clay mineral reaction in sedimentary rocks is the progressive transformation of smectite to illite via mixed-layer illite/smectite (I/S). Changes in both the illite/smectite ratio and ordering of I/S, as determined from X-ray powder diffraction profiles, correlate with changes in temperature due to burial depth. Although the smectite-to-illite reaction may be influenced by several factors, reaction progress appears to be strongly controlled by temperature. Studies show that the model proposed by Hoffman and Hower in 1979 is applicable in burial diagenetic settings from about 5 to 330 Ma, and includes most rocks about Miocene to Mississippian in age. Reliability of the I/S geothermometer is, however, dependent upon a good understanding of the rock's original clay-mineral composition.
Changes in the ordering of I/S are particularly useful in the exploration for hydrocarbons because of the common coincidence between the temperatures for the conversion from random-to-ordered I/S and those for the onset of peak, or main phase, oil generation. Here, the utility of the I/S geothermometer is reviewed in hydrocarbon-bearing rocks of Miocene to Mississippian age. Using three common applications, the I/S geothermometer is compared to other mineral geothermometers, organic maturation indices, and grades of indigenous hydrocarbons. Good agreement between changes in ordering of I/S and calculated maximum burial temperatures or hydrocarbon maturity suggests that I/S is a reliable semiquantitative geothermometer and an excellent measures of thermal maturity.
Origin and Clay-Mineral Genesis of the Cretaceous/Tertiary Boundary Unit, Western Interior of North America
- R. M. Pollastro, B. F. Bohor
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 7-25
-
- Article
- Export citation
-
A 3-cm-thick, two-layered clay unit that records mineralogic and textural evidence of a catastrophic event that occurred at a time now marked as the end of the Cretaceous Period was preserved in ancient peat-forming environments of the Western Interior Basin of North America. The two layers of this unit consist of altered distal ejecta and are easily distinguished by their distinctive texture and impact components from other clay beds, mainly tonsteins and detrital shales, occurring within the sequence of rocks enclosing the Cretaceous/Tertiary (K/T) boundary interval.
The lower claystone layer of the K/T boundary unit represents melted silicic target rock that has altered mainly to kaolin minerals. Impact components and signatures of this lower layer include a relict imbricate fabric of glass fragments, shards, bubbles, hollow spherules (altered microtektites), small amounts of shocked mineral grains, and a subdued iridium anomaly. These components and textures, combined with the layer's restricted areal distribution, indicate that this layer, called the “melt ejecta layer,” is the distal part of an ejecta blanket deposit. We interpret the melt ejecta layer to be an altered deposit of mostly impact-derived, shock-melted, silicic target material that traveled through the atmosphere within a detached ejecta curtain and on other ballistic trajectories.
The upper laminated layer of the K/T boundary unit consists mostly of altered vitric dust and abundant shocked minerals whose size and amounts decrease away from the putative crater site in the Caribbean area. High-nickel magnesioferrite crystals, high iridium content, geochemical signature, and worldwide distribution all suggest this upper layer originated from a cloud of vaporized bolide and entrained target-rock materials ejected above the atmosphere. The components of this layer, called the “fireball layer,” settled slowly by gravitational processes from an Earth-girdling vapor cloud and were deposited immediately on top of the already-emplaced melt ejecta layer.
The clay minerals that formed in the two layers are largely a function of composition and the highly unstable, shock-modified state of the fallout materials altered in acidic, organic-rich waters of ancient peat swamps. The fireball layer is mostly altered to smectitic clay from a mafic glass condensed from the vaporized chondritic bolide, along with some kaolinite formed from blebs of melted silicic target material entrained in the vapor plume cloud during ejection. In contrast, the melt ejecta layer is mainly kaolinitic, derived from silicic glass formed from melted target rocks. In this layer, the glass rapidly altered to mostly disordered, micrometer-sized “cabbage-like” or submicrometer-sized embryonic forms of spherical halloysite, probably from an allophane precursor. These crystallization characteristics of the melt ejecta layer are much different than those which formed coarse vermicular aggregates and platy kaolinite crystals in tonsteins from outside the K/T boundary interval throughout the Western Interior. The contrast in the incipient formation of dominantly kaolinitic clay minerals in the basal melt ejecta layer and of smectitic clay minerals in the overlying fireball layer reflect silicic versus mafic starting materials, respectively, and also supports the proposed two-phased meteorite impact ejection and dispersal model.
During subsequent burial and diagenesis of the K/T boundary unit, the metastable halloysite and smectite aggraded to kaolinite and mixed-layer illite/smectite, respectively. Both the ordering of kaolinite and illitization of smectite varies locally as a function of the degree of diagenetic grade or maturity, probably in response to local variations in temperature due to maximum burial depth (burial diagenesis).
Charge Heterogeneity and Nanostructure of 2:1 Layer Silicates by High-Resolution Transmission Electron Microscopy
- P. B. Malla, M. Robert, L. A. Douglas, D. Tessier, S. Komarneni
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 412-422
-
- Article
- Export citation
-
Several soil and reference smectites and vermiculites and one reference illite were examined by high-resolution transmission electron microscopy (HRTEM) to decipher the nanostructure and layer charge heterogeneity in these minerals. HRTEM results were compared with those obtained from powder X-ray diffraction (XRD) analysis. Samples were either exchanged with Na+ ions followed by equilibration with a very dilute solution of NaCl in a pressure membrane apparatus at 316 hPa (pF = 2.5) to see the effect of hydration and applied pressure on layer organization, or exchanged with dodecylammonium ions to see the expansion behavior. Oriented samples were embedded in a low viscosity resin and cut approximately 500 Å thick perpendicular to d(001) using an ultramicrotome fitted with a diamond knife. In general, Na-saturated soil clays possessed crystallites that were thinner (c-direction) and shorter (ab-direction) as compared with reference clays. In all cases, samples treated with dodecylammonium chloride exhibited nanostructures that were more disintegrated as compared with Na-saturated samples. In a soil vermiculite, dodecylammonium ion exchange showed frayed edges indicating the initiation of mica transformation to vermiculite from edge toward core. In a reference vermiculite (Transvaal) treated with dodecylammonium ions, in addition to completely expanded crystallites, a regular interstratification between expanded vermiculite and mica (phlogopite) layers was clearly observed in some crystallites. Such nanostructural details were not detected by XRD. HRTEM of the Na-treated illite showed thick crystallites having 10 Å layer separations, whereas the dodecylammonium-exchanged illite showed three types of layers with different degrees of expansion indicating charge heterogeneity in illite: 1) unexpanded (10 Å, highest charge) crystallites; 2) expanded high-charge vermiculite-like (24 Å) crystallites; and 3) occasionally expanded high-charge vermiculite-like (24 Å) layers interspersed in the matrix of 10Å crystallites.
The Formation of Illitic Clays from Kaolinite in KOH Solution from 225°C to 350°C
- Wuu-Liang Huang
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 645-654
-
- Article
- Export citation
-
Kaolinite was converted into illitic clays in a 2.58 m KOH solution in gold capsules using cold-seal pressure vessels at 225°, 250°, 300°, and 350°C and 500 bars. The XRD shows that the major reaction products are illitic clays with no interlayer expandability. The TEM shows that the illitic clays appear mainly platelet-like with a K/Si ratio close to that of muscovite/illite. The extent of the conversion was monitored by measuring the XRD peak ratio of muscovite (illite) and kaolinite in quenched run products. The results reveal that kaolinite converts to muscovite/illite in the KOH solution at an initial rate two to three orders of magnitude faster than that of similar reactions at near-neutral conditions.
Potassium Halide-Treated Montmorillonite (KTM) as a Solid Phase in Liquid Chromatography
- Faina Tsvetkov, Lisa Heller-Kallai, Uri Mingelgrin
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 527-536
-
- Article
- Export citation
-
Columns for high pressure liquid chromatography were prepared from spray dried samples of montmorillonite that were heated with potassium halides (KTM) to increase both the cation density on the clay surfaces and the expandability of the interlayers. Some of the clay samples were exchanged with Cu before and/or after the potassium halide treatment.
Retention of nitrobenzene and its chloro and methyl derivatives, of methyl substituted phenols and of nitrophenols on these columns was studied, using eluents ranging in polarity from hexane to isopropanol. The retention of the aromatic molecules depends on their specific interaction with active sites on the clay surfaces and on steric effects which limit access to the clay interlayers. Both penetrability and surface interaction are controlled by the composition and method of preparation of the solid phase and by the polarity of the mobile phase. Very strong adsorption of some eluates and efficient chromatographic separations between related substituted benzenes were achieved. Mixtures of the three isomers of cresol, chloronitrobenzene, nitrotoluene or nitrophenol were completely resolved by a judicious combination of solid phase and eluent.
Nature of the Illitic Phase Associated with Randomly Interstratified Smectite/Illite in Soils
- D. A. Laird, E. A. Nater
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 280-287
-
- Article
- Export citation
-
A dispersion-centrifugation-decantation procedure was used to isolate various particle size fractions from a sample of clay (<2 µm fraction) separated by sedimentation from the Ap horizon of a Webster soil (fine-loamy, mixed, mesic Typic Haplaquoll). The 0.02–0.06 µm size fraction was found to be enriched in an illitic phase associated with randomly interstratified smectite/illite. X-ray powder diffraction, chemical analysis, and high-resolution transmission electron microscopy confirmed that most of the illitic material in the 0.02–0.06 µm size fraction was composed of two-layer elementary illite particles with a layer charge of −0.47 per formula unit. The results demonstrate that this low-charge illitic phase can be physically separated from soil materials and that the low-charge illitic phase has chemical, morphological, and mineralogical properties that are uniquely different from those of smectite and illite.
Three Zones for Illite Formation During Burial Diagenesis and Metamorphism
- D. D. Eberl
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 26-37
-
- Article
- Export citation
-
Reinterpretation of published data for shale cuttings from the Gulf of Mexico sedimentary basin identifies three reaction zones for illite formation with increasing depth for well CWRU6. In a shallow zone (1.85 to 3 km), non-expanding illite-like layers formed primarily by the coalescence of smectite 2:1 layers around interlayer K+. In a middle zone (3 to 4 km), illite crystals neoformed from solution as coarser K-bearing phases and smectite were dissolved by organic acids. In the deepest zone (>4 km), illite recrystallized as less stable illite crystals dissolved, and more stable illite crystals grew during mineral ripening. The progressive loss of radiogenic argon in the deepest zone yielded a constant apparent age for the clays with depth, an effect previously attributed to “punctuated diagenesis.” The above hypothesis for illite formation emphasizes the need to establish the zone (i.e., the reaction mechanism) from which shales were derived before making detailed geologic interpretations based on illite mineralogy.
Synthesis and Paragenesis of Na-Beidellite as a Function of Temperature, Water Pressure, and Sodium Activity
- J. Theo Kloprogge, A. M. J. van der Eerden, J. Ben H. Jansen, John W. Geus, Roelof D. Schuiling
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 423-430
-
- Article
- Export citation
-
In the chemical system Na2O-Al2O3-SiO2-H2O, the stability field of Na-beidellite is presented as a function of pressure, temperature, and Na- and Si-activity. Na0.7-beidellite was hydrothermally synthesized using a stoichiometric gel composition in the temperature range from 275° to 475°C and at pressures from 0.2 to 5 kbar. Below 275°C kaolinite was the only crystalline phase, and above about 500°C paragonite and quartz developed instead of beidellite. An optimum yield of 95% of the Na0.7- beidellite was obtained at 400°C and 1 kbar after 20 days. Gels with a Na-content equivalent to a layer charge lower than 0.3 per O20(OH)4 did not produce beidellite. They yielded kaolinite below 325°C and pyrophyllite above 325°C. With gels of a Na-content equivalent to a layer charge of 1.5, the Na-beidellite field shifted to a minimum between temperatures of 275° and 200°C. This procedure offers the potential to synthesize beidellite at low temperatures. Beidellite synthesized from Na1.0-gel approach a Na1.35 composition and those from Na1.5- and Na2.0-gels a Na1.8 composition.
Modeling of H+ and Cu2+ Adsorption on Calcium-Montmorillonite
- Markus Stadler, Paul W. Schindler
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 288-296
-
- Article
- Export citation
-
The interaction of H+- and Cu2+-ions with Ca-montmorillonite was investigated in 0.1 mol/dm3 solutions of Ca(CIO4)2 at 298.2 K by Potentiometrie titrations using both glass electrodes (for H+) and ion specific electrodes (for Cu2+ ). The experimental data were interpreted on the basis of the surface complexation model. The calculations were performed with the least-squares program FITEQL (Westall, 1982) using the constant capacitance approximation. The best fit was obtained with a set of equilibria of the general form
and the constants log β1,0(int)S = 8.16 (± 0.04), log β-1,0(int)S = −8.71 (± 0.08), log β0,1(int)S = 5.87 (± 0.06), log β−1,1(int)S = −0.57 (± 0.12), log β−2,1(int)S = −6.76 (± 0.02). An appropriate modeling of the H+ adsorption data requires the introduction of a second surface group ≡ TOH with the acidity constant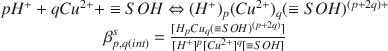 In addition, the ion exchange equilibria Ca2+ − Cu2+ and Ca2+ − H+ had to be taken into account. Arguments are presented to identify the groups ≡ SOH and ≡ TOH as surface aluminol groups =Al(OH)(H2O) and surface silanol groups ≡ Si-OH, respectively.
In addition, the ion exchange equilibria Ca2+ − Cu2+ and Ca2+ − H+ had to be taken into account. Arguments are presented to identify the groups ≡ SOH and ≡ TOH as surface aluminol groups =Al(OH)(H2O) and surface silanol groups ≡ Si-OH, respectively.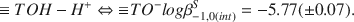
Illite/Smectite Geothermometry of the Proterozoic Oronto Group, Midcontinent Rift System
- Kirsten L. Price, S. Douglas McDowell
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 134-147
-
- Article
- Export citation
-
Characterization of the Nonesuch Formation, middle unit of the Proterozoic Oronto Group, as a potential hydrocarbon source for the Lake Superior basin portion of the Midcontinent Rift system requires an understanding of the thermal maturity of the region and its relationship to the thermal history. Illite/smectite (I/S) expandability data were collected from the Nonesuch Formation and the overlying Freda Sandstone and compared with organic thermal maturity data; both data sets coupled with a thermal and burial history for the White Pine area of Michigan allow regional interpretation of maximum formation temperatures of the Nonesuch Formation and the Freda Sandstone with respect to time. Samples collected from drill holes in northeastern Wisconsin display nearly pure smectite within the lower Freda Sandstone trending abruptly to ordered I/S within the Nonesuch Formation. Regular trends of decreasing expandability with depth occur in four other drill holes to the northeast. Comparison of I/S expandability between similar stratigraphic intervals reveals a significant trend of increasing thermal maturity to the northeast, with the lowest thermal maturities observed in the Iron River Syncline area just west of White Pine, Michigan.
I/S geothermometry suggests maximum temperatures in the Nonesuch Formation of 140°C in Wisconsin, 115°C in the Iron River Syncline area, 160°C at White Pine, and 190°C near the southern portions of the Keweenaw Copper District. The geographic pattern of temperatures determined from I/S geothermometry is identical to that determined from organic thermal maturity indicators in the Nonesuch Formation (Imbus et al., 1988, 1990; Hieshima and Pratt, 1991; Pratt et al, 1991; Mauk and Hieshima, 1992).
Regular variations in I/S expandability with depth occur in the Freda Sandstone and the Nonesuch Formation near the southern limits of the Keweenaw Copper District. These variations suggest a fossil geothermal gradient of 55°C/km and limit the thickness of sediment above the Nonesuch Formation to approximately 3 km. In comparison, 3.6 km of Freda Sandstone are presently exposed near the Wisconsin border, and numerical modeling suggests a range of 4–6 km of sediment overlying the Nonesuch Formation. None of the data indicate the presence of the Bayfield Group sediments above the Nonesuch Formation at the time of clay diagenesis. Samples from White Pine suggest a two-stage burial history: 1) clay reaction, possible hydrocarbon maturation, and copper-sulfide mineralization at maximum temperatures above 100°C during the main rifting and burial event, followed by 2) fracturing, reverse faulting, and fluid circulation during a rift-terminating compressional event that may have allowed petroleum migration and native copper mineralization at temperatures below 100°C. Abrupt changes in I/S expandability with depth and the presence of poorly crystalline I/S (greater than 80% expandable) and kaolinite in the Freda Sandstone in Wisconsin appear to represent later overprinting of the diagenetic assemblage by fluids that were probably cooler and of differing composition than earlier diagenetic fluids. However, the authigenic assemblage from the vicinity of White Pine, Michigan, which includes up to 25% expandable I/S, appears to represent a diagenetic profile formed during the main rifting and burial event. Therefore, these expandable I/S-type clays are essentially 1.0 billion years old.
Compositional End Members and Thermodynamic Components of Illite and Dioctahedral Aluminous Smectite Solid Solutions
- Barbara Ransom, Harold C. Helgeson
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 537-550
-
- Article
- Export citation
-
Consideration of XRD, TEM, AEM, and analytical data reported in the literature indicates that dioctahedral aluminous smectite and illite form two separate solid solutions that differ chemically from one another primarily by the extent of Al substitution for Si, the amount of interlayer K, and the presence of interlayer H2O. The data indicate that limited dioctahedral-trioctahedral and dioctahedral-vacancy compositional variations occur in both minerals. Excluding interlayer H2O and based on a half unit cell [i.e., O10(OH)2], natural dioctahedral smectite and illite solid solutions fall within the compositional limits represented by A0.3${\rm{R}}_{1.9}^{3 + }$
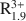 Si4O10(OH)2-AR2+ R3+ Si4O10(OH)2-A0.25${\rm{R}}_{0.3}^{2 + }{\rm{R}}_{1.8}^{3 + }$
Si4O10(OH)2-AR2+ R3+ Si4O10(OH)2-A0.25${\rm{R}}_{0.3}^{2 + }{\rm{R}}_{1.8}^{3 + }$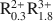 Al0.25Si3.75O10(OH)2 for smectites and A0.8${\rm{R}}_{1.9}^{3 + }$
Al0.25Si3.75O10(OH)2 for smectites and A0.8${\rm{R}}_{1.9}^{3 + }$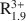 Al0.5Si3.5O10(OH)2-A0.55${\rm{R}}_{0.45}^{2 + }{\rm{R}}_{1.55}^{3 + }$
Al0.5Si3.5O10(OH)2-A0.55${\rm{R}}_{0.45}^{2 + }{\rm{R}}_{1.55}^{3 + }$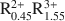 Al0.1Si3.9O10(OH)2-A0.9${\rm{R}}_{0.3}^{2 + }{\rm{R}}_{1.8}^{3 + }$
Al0.1Si3.9O10(OH)2-A0.9${\rm{R}}_{0.3}^{2 + }{\rm{R}}_{1.8}^{3 + }$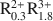 Al0.9Si3.1O10(OH)2 for illites, where A represents either monovalent cations or divalent cations expressed as their monovalent equivalent (e.g., Ca2+/2); R2+ stands for the divalent cations Mg2+ and Fe2+ and R3+ refers to the trivalent cations Al3+ and Fe3+. Taking account of these compositional limits, smectite and illite solid solutions can be described in terms of nine and six thermodynamic components, respectively, all of which are consistent with both the law of definite proportions and the concept of a unit cell. Thermodynamic components that can be used to describe natural smectite solid solutions in terms of a half unit cell [i.e., O10(OH)2] can be expressed as NaAl3Si3O10(OH)2, NaAl3Si3O10(OH)2 ·4.5H2O, Al2Si4O10(OH)2, Fe2Si4O10(OH)2, Mg3Si4O10(OH)2, Fe3Si4O10(OH)2, K3AlSi4O10(OH)2, KAl3Si3O10(OH)2, and Ca0.5Al3Si3O10(OH)2. Of these, NaAl3Si3O10(OH)2 ·4.5H2O provides explicitly for the presence of interlayer H2O in the mineral. Thermodynamic components representing illite solid solutions in natural systems can be written for a half unit cell as KAl3Si3O10(OH)2, KMg3AlSi3O10(OH)2, KFe3AlSi3O10(OH)2, Al2Si4O10(OH)2, KFe2AlSi3O10(OH)2, and K3AlSi4O10(OH)2. The calculations and observations summarized below indicate that neither smectite nor illite occur in nature as stoichiometric phases and that the two minerals do not form a mutual solid solution corresponding to mixed-layered illite/smectite.
Al0.9Si3.1O10(OH)2 for illites, where A represents either monovalent cations or divalent cations expressed as their monovalent equivalent (e.g., Ca2+/2); R2+ stands for the divalent cations Mg2+ and Fe2+ and R3+ refers to the trivalent cations Al3+ and Fe3+. Taking account of these compositional limits, smectite and illite solid solutions can be described in terms of nine and six thermodynamic components, respectively, all of which are consistent with both the law of definite proportions and the concept of a unit cell. Thermodynamic components that can be used to describe natural smectite solid solutions in terms of a half unit cell [i.e., O10(OH)2] can be expressed as NaAl3Si3O10(OH)2, NaAl3Si3O10(OH)2 ·4.5H2O, Al2Si4O10(OH)2, Fe2Si4O10(OH)2, Mg3Si4O10(OH)2, Fe3Si4O10(OH)2, K3AlSi4O10(OH)2, KAl3Si3O10(OH)2, and Ca0.5Al3Si3O10(OH)2. Of these, NaAl3Si3O10(OH)2 ·4.5H2O provides explicitly for the presence of interlayer H2O in the mineral. Thermodynamic components representing illite solid solutions in natural systems can be written for a half unit cell as KAl3Si3O10(OH)2, KMg3AlSi3O10(OH)2, KFe3AlSi3O10(OH)2, Al2Si4O10(OH)2, KFe2AlSi3O10(OH)2, and K3AlSi4O10(OH)2. The calculations and observations summarized below indicate that neither smectite nor illite occur in nature as stoichiometric phases and that the two minerals do not form a mutual solid solution corresponding to mixed-layered illite/smectite.
Synthesis of Smectite from Volcanic Glass at Low Temperature
- Katsutoshi Tomita, Hisanori Yamane, Motoharu Kawano
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 655-661
-
- Article
- Export citation
-
Smectite and zeolites were formed from a volcanic glass as the products of reaction with NaOH solution at 90°C and 100°C under atmospheric pressure. Formation conditions of smectite and various zeolites were determined by the ratio of the amounts of volcanic glass (g) to NaOH (g) in the solution. Smectite was formed under the condition that the values of weight of volcanic glass (g)/(NaOH(g)/40) are between 0.5 and 6. Fe was an important constituent of the octahedral layer of smectite.
Formation of Clay Minerals During Low Temperature Experimental Alteration of Obsidian
- Motoharu Kawano, Katsutoshi Tomita, Yoshitaka Kamino
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 431-441
-
- Article
- Export citation
-
Experimental alteration of obsidian in distilled-deionized water at 150°, 175°, 200°, and 225°C was studied. The alteration products were examined by X-ray powder diffraction, scanning electron microscopy, transmission electron microscopy (TEM), and energy dispersive X-ray analysis (EDX) to evaluate the formation process of clay minerals. The surface composition of obsidian before and after alteration was examined by X-ray photoelectron spectroscopy (XPS), and concentrations of released elements in solution were measured to elucidate alteration and dissolution processes. TEM clearly showed that allophane appeared as the first reaction product in each experiment. With increasing reaction length, noncrystalline straight fibrous material was formed in the aggregates of allophane particles as a metastable transitional phase, and tended to form curled or wavy bundles of fibers with longer reaction. The non-crystalline fibers were transformed into highly curled smectite exhibiting small circular forms less than 1.0 µm in diameter as reaction progressed. EDX confirmed that the smectite consisted mainly of Si, Al, and small amounts of Ca, K, and Fe. XPS revealed the formation of a dealkalized leached layer on the surface of obsidian during the reaction. The concentration of released elements suggested that nonstoichiometric dissolution proceeded during the reaction.
The Chemical Form of Vanadium (IV) in Kaolinite
- A. U. Gehring, I. V. Fry, J. Luster, Garrison Sposito
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 662-667
-
- Article
- Export citation
-
A combined application of electron paramagnetic resonance (EPR) and Fourier-transform infrared (FTIR) spectroscopies with thermal methods was used to determine the chemical form of V(IV) in a Georgia kaolinite (KGa-I). Precise values of the EPR spectroscopic g-values and hyperfine coupling constants were obtained for an untreated sample (g∥ = 1.940, A∦ = 18.71 mT; g⊥ = 1.966, A⊥ = 7.63 mT). Heating the sample to 1000°C in steps while monitoring phase changes with EPR and FTIR spectra led to the following structural interpretations: 1) Vanadium (IV) occurs almost entirely as an isomorphically substituted species in the octahedral sheet of KGa-1 kaolinite; 2) during the dehydroxylation of kaolinite at about 500°C and the subsequent formation of metakaolinite, V(IV)-substituted octahedral sites are readily converted into truncated octahedra exhibiting fourfold coordination; and 3) in these highly distorted four-coordinated sites, V(IV) is metastable, being completely oxidized at about 800°C.