Volume 57 - June 2009
Research Article
Al-Saturated Phlogopite: Charge Considerations and Crystal Chemistry
- Thomas J. Bujnowski, Stephen Guggenheim, Maria Franca Brigatti
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 673-685
-
- Article
-
- You have access Access
- Export citation
-
Factors controlling the crystal structure of phlogopite have been widely investigated; but the role of electrostatic interactions, for example, has received much less attention than other factors. The purpose of the present study was to peform a single-crystal refinement of an Al-saturated phlogopite and to use that refinement to supplement crystal-chemical analyses. The phlogopite investigated was from the Rumford quadrangle, Maine, and has the following chemistry: \$\end{document}
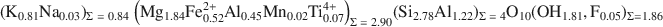 . The sample is a 1M polytype with C2/m symmetry and cell dimensions of a = 5.3220(4), b = 9.2170(7), c = 10.2511(8) Å, and β = 100.081(1)°. Hydrogen atoms were located and the crystal structure was refined to give parameters R1 = 0.0301 and weighted R2 = 0.0887. The octahedral M1 site was larger than the M2 (average M1—O: 2.079 Å, average M2—O: 2.062 Å) and the electron counts were equal (M1 = M2 = 14.8 e−); based on bond distances, which are more accurate than electron counts in determining occupancy; this result is consistent with a slight preference of Mg for M2 and Fe2+ for M1.
. The sample is a 1M polytype with C2/m symmetry and cell dimensions of a = 5.3220(4), b = 9.2170(7), c = 10.2511(8) Å, and β = 100.081(1)°. Hydrogen atoms were located and the crystal structure was refined to give parameters R1 = 0.0301 and weighted R2 = 0.0887. The octahedral M1 site was larger than the M2 (average M1—O: 2.079 Å, average M2—O: 2.062 Å) and the electron counts were equal (M1 = M2 = 14.8 e−); based on bond distances, which are more accurate than electron counts in determining occupancy; this result is consistent with a slight preference of Mg for M2 and Fe2+ for M1.Thirty-five Al-rich, natural phlogopite-1M samples that are of (1) high metamorphic grade, and that have (2) total Al contents ⩾ 1.27 atoms per formula unit (a.p.f.u.), (3) Fe3+ contents ⩽ 0.11 a.p.f.u., and (4) Mn contents ⩽0.10 a.p.f.u. along with the newly described phlogopite, exhibited crystal chemical trends related to increasing Al content. Octahedral substitutions of smaller, high-charge cations (i.e. Al) apparently decrease distortions in the octahedral sites and produce longer M2—O4 distances. In addition, VIFe-F avoidance apparently occurs in high Al-content samples, which are generally high in VIFe. The data set also shows that these samples have limited ordering among M sites (Fe2+ in M1 and Al in M2), an increase in β (99.96° to 100.32°) possibly caused by cation ordering and therefore size differences of M1 and M2, and interlayer (A) sites with A—Oouter distances that increase and A—Oinner distances that decrease with increasing Ti content.
Computer models were used to simulate electrostatic interactions in phlogopite structures with variable Al concentrations utilizing Pauling’s electrostatic valency principle, which considers first-coordination electrostatic interactions. The model results were compared to the maximum Al concentrations in natural and synthetic phlogopite samples. Model results revealed no indications (e.g. a limit reached or a sudden change occurred) that charge saturation/undersaturation of the apical oxygen atoms at Al contents equal to the maximum in natural and/or synthetic samples causes instability that could not be balanced by bond-length variation. However, a cation of higher charge substituting at M1 (or M2) may result in higher electrostatic repulsions between the other octahedral sites. Thus, the Al3+ content in the octahedral sites may reach a maximum, with Fe2+ for Mg substitutions favored.
Article
The transformation of synthetic hectorite in the presence of Cu(II)
- Håkon Fischer, Peter G. Weidler, Bernard Grobéty, Jörg Luster, Andreas U. Gehring
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 139-149
-
- Article
-
- You have access Access
- Export citation
-
The influence of Cu(II) on the hydrothermal and thermal transformations of a synthetic hectorite was investigated by a combined approach using mainly X-ray diffraction, thermal analyses, and electron paramagnetic resonance spectroscopy. The presence of Cu(II) during hydrothermal treatment increased the crystallite size. Copper (II) was both structure-bound and associated with the inner surfaces of the particles. Upon heating, structural destabilization of the hectorite began at ∼400°C as indicated by the formation of free radicals. Between 600 and 700°C, the hectorite converted to enstatite, and in the presence of Cu(II), to enstatite and richterite. The formation of richterite as an additional conversion product is explained by the creation of structural weakness due to structure-bound Cu(II) in F-containing hectorite. Our results suggest that traces of Cu(II), typical of natural environments, may influence the conversion products in high-temperature geochemical systems.
Significance of phyllosilicate mineralogy and mineral chemistry in an epithermal environment. Insights from the palai-islica Au-Cu deposit (Almería, SE Spain)
- Javier Carrillo-Rosúa, Salvador Morales-Ruano, Iñaki Esteban-Arispe, Purificación Fenoll Hach-Alí
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-24
-
- Article
-
- You have access Access
- Export citation
-
Phyllosilicate mineralogy is key to understanding hydrothermal processes within accepted epithermal deposit models but little information has been published about the mineral chemistry of epithermal deposits. X-ray diffraction, optical and electronic microscopy (scanning and transmitted), electron microprobe, and Fourier transform infrared spectroscopy were used in this work to study phyllosilicates in the Palai-Islica Au-Cu epithermal, volcanic-hosted deposit, in order to link phyllosilicate mineralogy and mineral chemistry to ore genesis. Different phyllosilicate assemblages are characteristic of two types of mineralization, and related hydrothermal alteration. Chlorite and mica appear in polymetallic quartz veins with sulfides, and in the related chloritic and sericitic hydrothermal alteration. These minerals have notable textural and chemical differences (i.e. Fe/(Fe+Mg), Si and Al in chlorite and illitic and phengitic components in mica) amongst veins and altered rocks, revealing different genetic conditions. These chemical features also distinguish propylitic and regional, non ore-related, low-temperature alteration. Hot hydrothermal fluids of near-neutral pH are responsible for vein mineralization and alteration. Illite, interstratified illite-smectite, kaolinite, and pyrophyllite are characteristic, with a distribution pattern by zones, of the intermediate argillic and advanced argillic alteration around areas of silicification. In the latter, native gold appears associated with interstratified illite-smectite, suggesting a relatively low-temperature formation. Hot, low-pH fluids are responsible for this mineralization and alteration assemblage. The present study contributes to epithermal models showing the co-existence of two different alteration styles in the same hydrothermal system.
Research Article
Relationship Between Pb2+ Adsorption and Average Mn Oxidation State in Synthetic Birnessites
- Wei Zhao, Haojie Cui, Fan Liu, Wenfeng Tan, Xionghan Feng
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 513-520
-
- Article
-
- You have access Access
- Export citation
-
The relationship between vacant Mn structural sites in birnessites and heavy-metal adsorption is a current and important research topic. In this study, two series of birnessites with different average oxidation states (AOS) of Mn were synthesized. One birnessite series was prepared in acidic media (49.6–53.6 wt.% Mn) and the other in alkaline media (50.0–56.2 wt.% Mn). Correlations between the Pb2+ adsorption capacity and the d110 interlayer spacing, the AOS by titration, and the release of Mn2+, H+, and K+ during adsorption of Pb2+ were investigated. The maximum Pb2+ adsorption by the birnessites synthesized in acidic media ranged from 1320 to 2457 mmol/kg with AOS values that ranged from 3.67 to 3.92. For birnessites synthesized in alkaline media, the maximum Pb2+ adsorption ranged from 524 to 1814 mmol/kg, with AOS values between 3.49 and 3.89. Birnessite AOS values and Pb2+ adsorption increased as the Mn content decreased. The maximum Pb2+ adsorption to the synthetic birnessites calculated from a Langmuir fit of the Pb adsorption data was linearly related to AOS. Birnessite AOS was positively correlated to Pb2+ adsorption, but negatively correlated to the d110 spacing. Vacant Mn structural sites in birnessite increased with AOS and resulted in greater Pb2+ adsorption. Birnessite AOS values apparently reflect the quantity of vacant sites which largely account for Pb2+ adsorption. Therefore, the Pb2+ adsorption capacity of birnessite is mostly determined by the Mn site vacancies, from which Mn2+, H+, and K+ released during adsorption were derived.
Article
Selectivity of Co and Ni by K-depleted micas
- Yunchul Cho, Sridhar Komarneni, Sang-il Choi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 279-289
-
- Article
-
- You have access Access
- Export citation
-
Contamination of the environment with heavy metals, including cationic radionuclides, is a serious problem which has yet to be fully overcome. A class of potentially effective cation exchangers for sequestering heavy metals which has received little attention is K-depleted mica. The purpose of this study was to investigate the heavy-metal cation exchange properties of K-depleted phlogopite and biotite, which were prepared from a natural phlogopite and biotite, respectively, using sodium tetraphenylborate (NaTPB). The X-ray diffraction (XRD) patterns showed that interlayer K+ ions were completely replaced with exchangeable Na+ ions, resulting in the expansion of the d001 spacing of both K-depleted phlogopite and K-depleted biotite. In order to investigate the cation exchange selectivity of K-depleted phlogopite and biotite for Co2+ and Ni2+, cation exchange isotherms and Kielland plots were constructed. The isotherms and Kielland plots indicated that both K-depleted phlogopite and biotite are highly selective for Co2+ as well as Ni2+. The XRD patterns after both 2Na+ → Co2+ and Ni2+ exchange reactions suggest that double sheets of interlayer water are present in the interlayer. These K-depleted micas are potential cation exchange materials for removal of some heavy metals such as Ni and radioactive species such as 60Co from solution.
Research Article
Trans-Vacant and cis-Vacant 2:1 Layer Silicates: Structural Features, Identification, and Occurrence
- Victor A. Drits, Bella B. Zviagina
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 405-415
-
- Article
-
- You have access Access
- Export citation
-
A comprehensive study of clay minerals should include determination of the vacancy pattern of the dioctahedral sheet. The purpose of this report is to consider the advantages and limitations in various diffraction and non-diffraction methods for the determination of the layer types in clay minerals. Identification of trans-vacant (tv) and cis-vacant (cv) clay minerals reported here is based on powder X-ray diffraction (XRD) patterns calculated for different polytypes consisting of either tv or cv layers, on the simulation of experimental XRD patterns corresponding to illite or illite fundamental particles in which tv and cv layers are interstratified, and on the semi-quantitative assessment of the relative content of the layer types in the interstratified structures by generalized Méring’s rules. A simple and effective method for identification of tv and cv layers in dioctahedral 2:1 layer silicates employs thermal analysis and is based on different dehydroxylation temperatures for tv and cv illite and smectite layers.
Crystal chemical analysis of various dioctahedral 2:1 layer silicates consisting of tv and cv layers indicates that compositional control is present in the distribution of octahedral cations over trans- and cis-sites. In dioctahedral smectites the formation of tv and cv layers is related to the layer composition and local order-disorder in the distribution of isomorphous cations. Dioctahedral 1M micas with abundant Fe3+ and Mg occur only as tv varieties. In contrast, 1M-cv illite, as well as cv layers in illite fundamental particles of I-S, can form only as Fe- and Mg-poor varieties. In illites and illite fundamental particles of I-S consisting of tv and cv layers, cv layers prevail when the amounts of Al in octahedra and tetrahedra are >1.55 and >0.35 atoms per O10(OH)2, respectively.
The main factors responsible for the stability of cv and tv illites have been established. Monomineral cv 1M illite, its association with tv 1M illite, and interstratified cv/tv illite occur around ore deposits, in bentonites, and in sandstones mostly as a result of different types of hydrothermal activity. The initial material for their formation should be Al-rich, and hydrothermal fluids should be Mg- and Fe-poor.
Tv and cv smectites of volcanic origin differ in terms of octahedral cation composition and distribution of isomorphous octahedral cations. Mg-rich cv smectites have random distribution of isomorphous octahedral cations, whereas in Mg-bearing tv smectites octahedral Mg cations are dispersed so as to minimize the amount of Mg-OH-Mg arrangements.
Terahertz Time-Domain Spectroscopy of Selected Layered Silicates
- Marián Janek, Ignác Bugár, Dušan Lorenc, Vojtech Szöcs, Dušan Velič, Dušan Chorvát
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 416-424
-
- Article
-
- You have access Access
- Export citation
-
Micaceous layer silicate clay minerals are attractive materials for applications involving non-linear optics because of their low cost and ability to form well ordered, platy aggregates, but such applications require precise knowledge of the dielectric behavior of the clay. The purpose of the present study was to use Terahertz time-domain spectroscopy (THz-TDS) to determine the dielectric properties of certain cleavable layered clay minerals, including muscovite, vermiculite, phlogopite, and biotite. The samples were characterized by X-ray diffraction and Fourier transform infrared spectroscopy as well as chemical analysis by Energy dispersive X-ray spectroscopy. The THz frequency window investigated was the far-infrared region of 3.3 to ∼40.0 cm−1 corresponding to 0.1 and 1.2 THz, respectively. The samples were selected so that the hydrated form of the interlayer cation, e.g. Mg2+ present in the interlayer gallery of vermiculite, could be compared to species such as phlogopite, biotite, and muscovite with the dehydrated form of interlayer cations such as K+ or Na+. The frequency-dependent complex index of refraction of these natural materials was determined to vary between 2.50 and 2.80. The presence of water in the interlayer space of vermiculite was reflected in the detection of increased values of the absorption index in comparison with the muscovite, phlogopite, and biotite.
Article
X-Ray diffraction line-broadening study on two vibrating, dry-milling procedures in kaolinites
- Pablo Pardo, Joaquín Bastida, Francisco J. Serrano, Rafael Ibáñez, Marek A. Kojdecki
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 25-34
-
- Article
-
- You have access Access
- Export citation
-
Due to the great technological importance of the microstructure of kaolinite, characterizing its evolution during dry milling of kaolin and analyzing the microstructural information obtained from different methods were the main aims of this work. The microstructural alteration of kaolinite is evaluated by X-ray diffraction and electron microscopy methods, comparing the results obtained and analyzing the correlations between them. The Warren-Averbach and Voigt-function methods of X-ray diffraction microstructural analysis have been applied successfully to the study of the effects of two different, vibrating-cup dry-milling configurations in the microstructure of kaolinite from the reflections corresponding to (001) diffracting planes. The crystallite-size estimates obtained from the two methods correlate well. Field emission scanning electron microscopy measurements of kaolinite particle thickness are in good agreement with the crystallite size estimated by the two methods. The Warren-Averbach method also provided estimates of the contribution to line broadening. Vibrating-cup milling has been proved to be a more efficient method of strain comminution of kaolinites than other milling techniques, reaching extensive microstructural degradation within seconds.
Research Article
Origin and Mineralogy of Sepiolite and Palygorskite From the Tuluanshan Formation, Eastern Taiwan
- Ming Kuang Wang, Pao Chung Tseng, Shyun Sheng Chang, Dah Tong Ray, Yen Hong Shau, Yun Wei Shen, Ruey Chyong Chen, Po Neng Chiang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 521-530
-
- Article
-
- You have access Access
- Export citation
-
The Tuluanshan Formation of the eastern Coastal Range of Taiwan overlies an andesitic core complex presumed to be the source of hydrothermal fluids responsible for the Si- and Mg-rich mineralization of sepiolite and palygorskite (attapulgite) which are found in veins within fissures and in fracture zones of the volcanic rocks of the region. This study was undertaken in order to understand these relationships better by characterizing sepiolite and palygorskite in this Formation and by examining their occurrence and distribution in the Tungho (TH) and Chunjih (CJ) areas. Samples were analyzed using X-ray diffraction (XRD), thermal analysis, Fourier-transform infrared (FTIR) spectroscopy, and petrographic, scanning (SEM), and transmission (TEM) electron microscopic methods. Sepiolite and palygorskite are blocky and earthy-type materials that display fibrous characteristics when viewed using TEM and SEM and occurred alone or with chalcedony in veins. The fibers of blocky sepiolite are commonly intercalated with smectite but the earthy type of sepiolite and palygorskite observed in this study displayed precipitation from fluid enriched in Si, Al, Mg, and minor Fe and depleted in other ions at an earlier stage of offset of the andesitic veins. Continuation of reverse faulting and high shearing stress caused the precipitation of a significant quantity of interlaminated sepiolite. Sepiolite and palygorskite were formed at an earlier stage of fluid interaction relative to smectite in the Tuluanshan Formation.
Article
Simple synthesis and characterization of nanoporous materials from talc
- Chunfang Du, Huaming Yang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 290-301
-
- Article
-
- You have access Access
- Export citation
-
Synthetic siliceous mesoporous materials are of great value in many different applications, including nanotechnology, biotechnology, information technology, and medical fields, but historically the resource materials used in their synthesis have been expensive. Recent efforts have focused on indirect synthesis methods which utilize less expensive silicate minerals as a resource material. The purpose of the present study was to investigate talc, a natural silicate mineral, as one such resource. It was used as raw material to prepare two advanced materials: porous silica (PS) and ordered mesoporous silica (MCM-41). The PS, with a specific surface area of 260 m2/g and bimodal pore-size distribution of 1.2 nm and 3.7 nm, was prepared by grinding and subsequent acid leaching. The MCM-41, with a large surface area of 974 m2/g and a narrow pore-size distribution of 2.8 nm, was obtained using a surfactant, cetyltrimethylammonium bromide (CTAB), by hydrothermal treatment using the as-prepared PS as a source of Si. The two resultant materials were characterized by small angle X-ray diffraction (SAXRD) and wide-angle X-ray diffraction (WAXRD), high-resolution transmission electron microscopy (HRTEM), solid-state magic-angle-spinning nuclear magnetic resonance (MAS NMR), Fourier transform infrared spectroscopy (FTIR), and N2 adsorption-desorption measurements. Based on these measurements, possible processes of transformation of PS from talc, upon acid treatment, and the formation of MCM-41 were investigated systemically. Acid leaching induced the transformation of a rigid layered structure to a nearly amorphous one, with micropores formed by a residual layered structure and mesopores formed from a condensed framework. The MCM-41 was a mixture of silanol groups (Si(SiO)3(OH)) and a condensed Q4 framework structure (Si(SiO)4), with a small amount of remaining Q3 layered structure (Si(SiO)3OMg). The increased Q4/Q3 value confirmed greater polymerization of MCM-41 than of PS. At the low CTAB concentration used (2 wt.%), the highly charged silicate species controlled the surfactant geometry. Charge-density matching, together with the degree of polymerization of the silicates, determined the resultant mesophase.
Research Article
Variation of Preferred Orientation in Oriented Clay Mounts as a Result of Sample Preparation and Composition
- R. Dohrmann, K. B. Rüping, M. Kleber, K. Ufer, R. Jahn
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 686-694
-
- Article
-
- You have access Access
- Export citation
-
In X-ray diffraction (XRD) analysis, preparation of oriented clay specimens enhances their 00l reflections by arranging basal surfaces parallel to the specimen surface. In one-dimensional modeling of XRD intensities, degree of preferred orientation is one of the variable parameters and a user may choose different σ* values for different minerals. The usual assumption is, however, that the layers of all clay minerals that are present exhibit a similar degree of preferred orientation to that of the clay mineral flakes parallel to the basal plane. If the orientation of individual clay minerals is significantly different, and if this is not taken into account, the relative proportions of the constituent minerals cannot be modeled accurately. The actual or so-called ‘preferred’ orientation is a potentially large source of error in any attempt at quantitative XRD analysis because it cannot be assumed to be constant among different minerals and may also vary as a result of pretreatment. In the present study the influence of sample composition and sample pretreatment on the degree of preferred orientation was determined using the parameter σ*. A statistical parameter was calculated to determine and ensure the reproducibility of σ* measurements. The most important result was that, when mixed together, clay minerals influence each other in terms of the degree of preferred orientation. Among individual samples, the degree of preferred orientation can be different for each clay mineral. The power of sonication used in sample pretreatment of a pure kaolinite and a pure illite had no significant influence on the degree of preferred orientation. The changes in intensities upon variation of the tilting angle (χ) allowed for calculation of σ* of smectites in pure samples, in admixtures, and in samples treated in two different ways (air-dried and glycerol-intercalated), which is reported here for the first time. Smectites are very fine grained with flexible morphology which is believed to be the reason for their tendency to exhibit poor orientation (σ* = 22°); further research is required to establish whether this is a general feature of smectites. After glycerol treatment a soil smectite showed a slightly better orientation compared to the air-dried pattern. The results of the study illustrate the difficulty of predicting changes in preferred orientation of clay mineral admixtures, even if non-platy minerals such as clay-sized quartz are added. In general, σ* decreased when non-platy minerals were added, which is explained by changes in geometry of the specimen. Not all clay minerals, however, showed simultaneous changes in their orientation behavior.
Article
Swelling behavior of Na- and Ca-montmorillonite up to 150°C by in situ X-Ray diffraction experiments
- Shoji Morodome, Katsuyuki Kawamura
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 150-160
-
- Article
-
- You have access Access
- Export citation
-
The effects of temperature on the swelling properties of smectites are important for a variety of different geological conditions, but studies on this topic have been rather limited. The purpose of this study was to investigate the swelling behavior of Na- and Ca-montmorillonite at various temperatures greater than room temperature, up to 150°C, using in situ X-ray diffraction (XRD) analysis. A sample chamber was designed, the temperature and humidity of which were controlled precisely, for environmental in situ measurements. The XRD measurements were performed at small relative humidity (RH) intervals for precise observation of the swelling behavior.
The swelling behavior of Na-montmorillonite showed distinct zero-, one-, and two-layer hydration states. The basal spacings of Na-montmorillonite changed continuously with RH for various temperatures in the transition region between the zero- and one-layer hydration states, and the swelling curves of the transition region moved to greater degrees of RH with increasing temperature. The basal spacings jumped from the one- to two-layer hydration states for all temperatures at almost the same RH.
The basal spacings of Ca-montmorillonite changed continuously from the zero- to the two-layer hydration states at all temperatures. This behavior is remarkably different from that of Na-montmorillonite. At low-RH conditions, the d001 value of Ca-montmorillonite decreased with increasing temperature. The swelling curves of Ca-montmorillonite did not show a plateau at any temperature for the one-layer hydration state. The swelling curves of Ca-montmorillonite moved to greater RH with temperature, similar to the transformation region between the zero- and one-layer hydration states in Na-montmorillonite. These differences between Na- and Ca-montmorillonite are related to the hydration powers of exchangeable cations.
Rapid and direct synthesis of spherical organotalc
- Maguy Jaber, Fabrice O. M. Gaslain, Jocelyne Miehé-Brendlé
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 35-39
-
- Article
-
- You have access Access
- Export citation
-
Organotalcs, in which organic moieties are covalently bonded to Si atoms belonging to the tetrahedral sheets, are usually prepared by a sol-gel process starting from ethanolic solution of Mg nitrate, organo-alcoxysilanes, and aqueous sodium hydroxide solution. In this case, gypsum-like particles are obtained. In this work, evaporation-induced self-assembly within aerosols was used for the first time in order to prepare organotalc spheres. These hybrid lamellar materials can be used as environmental barriers, as polymer fillers, and as catalytic supports. Using octyltriethoxysilane as a source of Si, spherical particles with sizes ranging from 20 nm to 1 µm are obtained. X-ray diffraction and transmission electron microscopy images show that the d001 value equals 2.8 nm meaning that, in this case, organic moities are either alternatively distributed or identically tilted in the interlayer space. Compared to the classical synthesis at room temperature or in autoclaves, and besides being a continuous process, the reaction time is reduced to several minutes instead of hours. Homogeneous forms are obtained by aerosol whereas irregular shapes are obtained in the classical synthesis.
Research Article
The Formation of Layered Double Hydroxides on Alumina Surface in Aqueous Solutions Containing Divalent Metal Cations
- František Kovanda, Petra Mašátová, Petra Novotná, Květa Jirátová
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 425-432
-
- Article
-
- You have access Access
- Export citation
-
Layered double hydroxides (LDHs) are often used as precursors for mixed-oxide catalysts and the deposition of a LDH layer on supporting materials would be advantageous because of better utilization of active components. The purpose of the present study was to investigate the formation of LDHs on Al2O3/Al supports prepared by the anodic oxidation of aluminum foil in dilute aqueous solutions of Co, Mn, and/or Ni nitrates. The LDH deposition was carried out under hydrothermal conditions at 80–180°C for periods ranging from 8 h to 7 days. In the initial stages of the reaction, a surface alumina hydration was observed. The LDH phase was detected after long-term deposition (3–7 days) at 120–160°C and only a small amount of Mn was incorporated in the LDHs deposited. In solutions containing only Co and Mn cations, scanning electron microscopy (SEM) images show a gradual growth of platy crystals resulting in the formation of discrete bulky aggregates with sizes up to several tens of micrometers. The adhesion of aggregates to the support probably decreased with increasing size and oversized aggregates fell away; none was found in SEM images of the samples obtained after prolonged reaction times. When Ni cations were present in the solution, they were incorporated preferentially into the LDH phase; an homogeneous layer was formed, with single platy crystals oriented perpendicular to the support. After calcination at 500°C, the products formed on Al2O3/Al support exhibited a shift of reduction maxima to higher temperatures in comparison with mixed oxides obtained by thermal decomposition of coprecipitated LDHs.
Partial Dissolution of Glauconitic Samples: Implications for the Methodology of K-Ar and Rb-Sr Dating
- Arkadiusz Derkowski, Jan Środoń, Wojciech Franus, Peter Uhlík, Michał Banaś, Grzegorz Zieliński, Maria Čaplovičová, Małgorzata Franus
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 531-554
-
- Article
-
- You have access Access
- Export citation
-
The K-Ar dating of glauconite has been used as an important stratigraphic tool for many decades. The application of this technique is limited to pure glauconites, free of detrital contamination by K-bearing phases, often not easy to detect. This study extends the application of isotope dating to the contaminated glauconites and offers a precise technique for detecting the detrital contamination of glauconites.
The most common K-bearing detrital contaminants have smaller (K-feldspars, Al-rich dioctahedral micas) or greater (trioctahedral micas) dissolution rates than glauconite in extremely low pH solutions. The differences in the dissolution rates can be applied to evaluate the purity of the glauconite and its crystallization age.
The interlaboratory GLO glauconite standard and grain-size fractions separated from glauconitic sandstones of the Paleogene (sample GL) and Jurassic (sample GW8) ages were treated with acid (3M HCl, at 99±2°C) for different reaction times (0.5–7 h) and measured for their apparent isotopic ages.
Microporous amorphous silica with large specific surface area is the solid product of the reaction and its content increases with reaction time. The K-Ar dates (apparent ages) of the solid residues increase significantly with reaction time: from 44.6 to 107 Ma for the GL sample and from 125.7 to 394.7 Ma for GW8. The increase is negligible in the case of the GLO standard. The Rb-Sr data of the GL sample were modeled using initial 87Sr/Sr ratios of 0.707–0.709, which resulted in a 29.9–35.8 Ma date for the untreated portions of GL, and ∼42.6 Ma after 7 h of treatment.
The increase of isotopic K-Ar date with increasing time of dissolution is interpreted to be a result of increasing concentration of detrital, acid-resistant, K-bearing minerals, observed also with the electron microscope and X-ray diffraction. Probabilistic modeling based on single (K-Ar) or double (K-Ar and Rb-Sr) isotopic systems evaluated the isotopic ages of the detrital and authigenic minerals, and their K2O and Rb concentrations. The crystallization ages computed using these two methods are: 24.0, 26.5, and 32.3 Ma for the GL material, and 117.3–121.8 Ma for the GW8 series.
The proposed method based on partial dissolution is a potential tool for evaluating the reliability of glauconite dating.
Article
Application of 29Si and 27Al MAS NMR spectroscopy to the study of the reaction mechanism of kaolinite to illite/muscovite
- Marco Mantovani, Alberto Escudero, Ana Isabel Becerro
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 302-310
-
- Article
-
- You have access Access
- Export citation
-
Understanding the mechanisms for illitization of clay minerals has important applications in reconstructing geologic histories and determining the origins of physical and chemical characteristics of buried sediments. While many studies have been carried out on this topic, few have focused on the mechanism of illite formation from kaolinite. The purpose of this study was to investigate more deeply the illitization of kaolinite in KOH solution at a high solid/liquid ratio (1000 mg/mL). X-ray diffraction (XRD) and infrared spectroscopy were used to follow the formation of new crystalline phases and the composition of the octahedral sheet, while the transformation of the Si and Al local environments was analyzed by 29Si and 27Al magic angle spinning nuclear magnetic resonance spectroscopy (MAS NMR). The results revealed that the first reaction stage consists of the diffusion of Al from the octahedral to the tetrahedral sheet of the kaolinite TO layers, giving rise to the precursors of the illite/muscovite nuclei. Combination of XRD with 27Al MAS NMR measurements indicated that a minimum amount of tetrahedral Al is required in the original TO layer before condensation of a second tetrahedral sheet occurs to complete the formation of the illite/muscovite TOT layers.
Enhancement of dissolution rates of amorphous silica by interaction with amino acids in solution at pH 4
- Motoharu Kawano, Tamao Hatta, Jinyeon Hwang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 161-167
-
- Article
-
- You have access Access
- Export citation
-
Amino acids are present in various geochemical environments and they interact with mineral surfaces. To evaluate the effects of amino acids on mineral dissolution at pH conditions less than their isoelectric points (pI), dissolution experiments of X-ray amorphous silica in solutions containing 10.0 mmol/L of various amino acids (cysteine, asparagine, serine, tryptophan, alanine, threonine, histidine, lysine, and arginine) at pH 4 were performed. The results confirmed that basic amino acids (histidine, lysine, and arginine) produce an 8- to 8.5-fold enhancement of the rate of dissolution of amorphous silica compared with an amino acid-free control. Neutral amino acids (cysteine, asparagine, serine, tryptophan, alanine, and threonine) enhanced rates of dissolution by a factor of ∼3 to 3.5. The rate-enhancement effects of amino acids are controlled by concentrations of the amino acid’s cationic species which interact with the negatively charged >SiO− sites at the surface of the amorphous silica.
Research Article
Removal of a Binary Dye Mixture of Congo Red and Malachite Green From Aqueous Solutions Using a Bentonite Adsorbent
- Abdil Özdemir, Can Serkan Keskin
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 695-705
-
- Article
-
- You have access Access
- Export citation
-
An important application of clay is as a solid adsorbent for industrial dyes. The aim of the present work was to carry out an experimental—theoretical study of the adsorption of dye mixtures, namely malachite green (MG) and Congo red (CR), by bentonite. Adsorption studies were conducted after evaluation of the impact of several parameters, including pH, adsorbate dose, and contact time, on the removal of MG and CR. The pH of the dye solution is strongly affected by the chemistry of both the dye molecules and of the adsorbent in an aqueous solution. Where both dye molecules exist in solution, the optimum pH was found to be 8.2 in order to achieve the maximum adsorption of both MG and CR. Preliminary studies showed that 60 min of contact time is sufficient to reach adsorption equilibrium. The adsorption studies were carried out using 1.0 g samples of bentonite. The amount of dye adsorbed was found by application of classical least squares to the synthetic dye mixtures. Data from equilibrium adsorption on bentonite were analyzed by Freundlich, Langmuir, Redlich-Peterson, and Temkin isotherm equations using regression analysis for non-linear forms of those equations. For binary-mixture analysis, isotherm parameters were determined from single-component adsorption studies and the theoretical amount of dye adsorbed was calculated using an extended Langmuir isotherm. Non-linear error analysis showed that the Temkin and Redlich-Peterson isotherms gave the best fits to the equilibrium data for adsorptive removal of MG and CR by bentonite.
Article
Chemostratigraphic correlation of sediments containing expandable clay minerals based on ion exchange with Cu(II) triethylenetetramine
- Tomáš Grygar, Jaroslav Kadlec, Anna Žigová, Martin Mihaljevič, Tereza Nekutová, Richard Lojka, Ivo Světlík
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 168-182
-
- Article
-
- You have access Access
- Export citation
-
Copper(II) triethylenetetramine [Cu(trien)]2+ is an agent suitable for the 1-step determination of the cation exchange capacity (CEC) of many geomaterials using a procedure much less laborious than other, commonly used methods. It is also suitable for the determination of the composition of original exchangeable cations. In contrast to other common ions used for CEC analysis, the Cu(II) complex with triethylenetetramine, [Cu(trien)]2+, is specific for expandable clay minerals. The robustness of [Cu(trien)]2+ analysis was verified using reference clays, ion-exchanged reference clays, sediments, and soils. The [Cu(trien)]2+-based CEC of expandable clay minerals is not influenced significantly by ferrihydrite, goethite, manganite, birnessite, calcite, and gypsum. Birnessite, calcite, and gypsum admixtures affect the composition of the evolved cations. [Cu(trien)]2+ does not recover the entire CEC of soils (but rather that of the clay minerals only) which contain components other than clays which contribute to the CEC, e.g. soil organic matter. In a series of loess with buried paleosols and recent soils the [Cu(trien)]2+-based CEC ranged from 30 to 110% of total CEC obtained by traditional BaCl2 methods. The relative ratio of Ca to Mg, the prevailing exchangeable cations in soils and sediments in exogenic environments, are similar after [Cu(trien)]2+ and conventional BaCl2 treatments. The Ca/Mg ratio in the exchangeable fraction was used successfully for chemostratigraphic correlation of paleolacustrine sediments from a large lake in the Upper Carboniferous basins of eastern equatorial Pangaea and a series of recent flood plain sediments of the meandering Morava River in the Czech Republic. The Ca/Mg ratio obtained by [Cu(trien)]2+ analysis is proposed as a novel tool for the chemostratigraphic correlation of sediment series containing expandable clay minerals.
Correction
Evidence of Degradation of Triarylmethine Dyes on Texas Vermiculite
- Giora Rytwo, Yotam Gonen, Reuma Huterer-Shveky
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 555-565
-
- Article
-
- You have access Access
- Export citation
-
Synthetic dyes in industrial effluents pose a significant risk to human health and the environment, so much effort has been expended to degrade them using various methods, including the use of clay minerals as catalysts. The purpose of this study was to advance understanding of the mechanisms for clay-catalyzed degradation of crystal violet (CV) and other triarylmethine dyes using three different vermiculite clays (Llano, Texas, VTx-1; Ojen, OV; and Russian, RV), a montmorillonite (SWy-1), and a Spanish sepiolite (SEP). While OV, RV, SWy-1, and SEP showed almost no activity with respect to dye degradation, VTx-1 caused complete removal of the dye from solution up to the equivalent of 200% of the cation exchange capacity of the clay. While large amounts of dye were removed from the solution, no change in basal spacing was observed by X-ray diffraction. The kinetics of removal of CV from solution began after a lag period of >10 days in a process that can be described by pseudo-second order kinetics. By comparison, adsorption of CV onto SWy-1 and SEP was immediate, without any lag period. Sonication treatment of the VTx-1 vermiculite suspension caused the CV removal process to begin immediately. Fourier-transform infrared measurements of adsorption of CV on clays revealed that for the OV and RV vermiculites, SEP sepiolite, and SWy-1 montmorillonite the spectra were similar to the original dye; the spectra of the VTx-1-dye differed considerably, however, exhibiting vibrations of methylene groups (—CH2—) which were not present in the CV molecule. The significant changes in the IR spectrum indicated that CV underwent degradation on the surface of the VTx-1 vermiculite. Carbon-content analysis led to the conclusion that degradation products remained bound to the clay. Similar effects were observed for two other triarylmethine dyes (malachite green and methyl green) added to VTx-1, indicated that it may, therefore, be considered suitable as a sorbent to remove and decompose such dyes from industrial effluents. Pretreatment by sonication would remove the need for long incubation times.