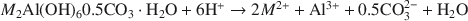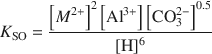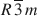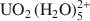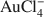Volume 51 - February 2003
Editorial
Editorial
- Derek C. Bain
-
- Published online by Cambridge University Press:
- 01 January 2024, p. 587
-
- Article
-
- You have access Access
- Export citation
Research Article
Geochemical Signatures of Paleodepositional and Diagenetic Environments: A STEM/AEM Study of Authigenic Clay Minerals from an Arid Rift Basin, Olduvai Gorge, Tanzania
- Victoria C. Hover, Gail M. Ashley
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 231-251
-
- Article
- Export citation
-
Olduvai Gorge, Tanzania (East African Rift) exposes a 100 m thick Plio-Pleistocene sequence of dominantly volcaniclastic sediments deposited in a 50 km wide closed basin containing a playa lake. A scanning transmission electron and analytical electron microscopy (STEM/AEM) study of authigenic clay minerals in sediments from representative depositional environments in the basin (pyroclastic fan, fluvial plain, wetland, lake margin and lake basin) was undertaken to determine whether clay compositions and textures could provide unique geochemical fingerprints characteristic of source area (Plio-Pleistocene trachytic volcanics vs. Precambrian quartzose-feldspathic basement) or paleoenvironmental conditions.
Our study shows that compositional signatures obtained by clay minerals during early pedogenesis are inherited from their parent source rocks. Sediments sourced from volcanics contain highly disordered, dioctahedral smectite. Those sourced from Precambrian basement are similar, but are more Al-rich. Subsequent neoformation in the pedogenic (soil) or diagenetic (lake-margin, lake) environments results in the modification of original clay mineralogy, compositions, and textures, and unique paleoenvironmental fingerprints are acquired. Soils developed on the distal pyroclastic fan contain smectite with more Fe(III) and Mg than smectite from the proximal pyroclastic fan sediments. A trend of decreasing Al and increasing Mg content occurs in smectite compositions in samples from the fluvial to lake-margin and lake environments as a result of partial replacement of original dioctahedral Al-rich smectite by neoformed trioctahedral Mg-rich smectite (stevensite). Neoformed celadonite replaces smectite in the most saline lake sediments.
The STEM/AEM data collectively indicate that diagenesis in the saline-alkaline lake results in the replacement of Al-rich dioctahedral smectite by Mg-rich trioctahedral smectite (stevensite) and Mg- and Al-rich celadonite. Thus, determination of clay mineral compositions at a basin-wide scale provides a useful tool for interpreting the spatial distribution of depositional and diagenetic environments.
Hydrotalcite-Like Minerals (M2Al(OH)6(CO3)0.5.XH2O, Where M = Mg, Zn, Co, Ni) in the Environment: Synthesis, Characterization and Thermodynamic Stability
- C. A. Johnson, F. P. Glasser
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-8
-
- Article
- Export citation
-
Hydrotalcite-like layered double hydroxides (LDH), of the formulation M2Al(OH)6(CO3)0.5.H2O, where M = Mg, Zn, Co, Ni, have been prepared, the products characterized and their solubility products measured at ionic strengths of 0.0065 and 0.0128 M and at 25°C. Steady-state solubility was reached after 100 days. The solubility products have been formulated according to the following reaction:
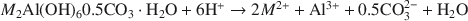 where
where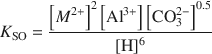
Average values of Kso for I = 0, estimated using the Davies equation, are 25.43, 20.80, 22.88 and 20.03 for Mg, Zn, Co and Ni, respectively. Model calculations reveal that the thermodynamic stability of the LDHs is greater than that of the corresponding divalent hydroxides for Zn, Co and Ni below a pH of ∼10, 9 and 8, respectively, and at least up to pH 12 for Mg.
Removal of Heavy Metals from Waste Waters by Natural and Na-Exchanged Bentonites
- E. Álvarez-Ayuso, A. García-Sánchez
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 475-480
-
- Article
- Export citation
-
Batch sorption studies of Cr(III), Ni(II), Zn(II), Cu(II) and Cd(II) were conducted on Ca-bentonite and Na-bentonite to determine their retention capacities for these metal cations commonly present in the waste waters of galvanic industries. The Langmuir model was found to describe the sorption process well, offering maximum sorption capacities of 44.4 mg Cr/g, 6.32 mg Ni/g, 5.75 mg Zn/g, 7.72 mg Cu/g, 7.28 mg Cd/g on Ca-bentonite and 49.8 mg Cr/g, 24.2 mg Ni/g, 23.1 mg Zn/g, 30.0 mg Cu/g, 26.2 mg Cd/g on Na-bentonite. The use of Na-bentonite in the purification of waste waters from Ni, Cr and acid-Zn electroplating processes was an effective treatment in small-scale and in semi-industrial-scale tests. This proves the worth of this method as an alternative to existing methods. In contrast, Nabentonite was not effective in Zn sorption from cyanide-containing waste waters.
Ab Initio Determination of Edge Surface Structures for Dioctahedral 2:1 Phyllosilicates: Implications for Acid-Base Reactivity
- Barry R. Bickmore, Kevin M. Rosso, Kathryn L. Nagy, Randall T. Cygan, Christopher J. Tadanier
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 359-371
-
- Article
- Export citation
-
The atomic structure of dioctahedral 2:1 phyllosilicate edge surfaces was calculated using pseudopotential planewave density functional theory. Bulk structures of pyrophyllite and ferripyrophyllite were optimized using periodic boundary conditions, after which crystal chemical methods were used to obtain initial terminations for ideal (110)- and (010)-type edge surfaces. The edge surfaces were protonated using various schemes to neutralize the surface charge, and total minimized energies were compared to identify which schemes are the most energetically favorable. The calculations show that significant surface relaxation should occur on the (110)-type faces, as well as in response to different protonation schemes on both surface types. This result is consistent with atomic force microscopy observations of phyllosilicate dissolution behavior. Bond-valence methods incorporating bond lengths from calculated structures can be used to predict intrinsic acidity constants for surface functional groups on (110)- and (010)-type edge surfaces. However, the occurrence of surface relaxation poses problems for applying current bond-valence methods. An alternative method is proposed that considers bond relaxation, and accounts for the energetics of various protonation schemes on phyllosilicate edges.
A Crystal-Chemical Study of Natural and Synthetic Anionic Clays
- Cristina de la Calle, Charles-Henri Pons, Jacques Roux, Vicente Rives
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 121-132
-
- Article
- Export citation
-
A comparative crystallochemical study was performed on natural and synthetic hydrotalcite-like compounds with similar compositions. The nature of the brucite-like sheet stacking was addressed by means of powder X-ray diffraction. From the resulting electron diffraction patterns it was possible to establish the order-disorder of the cations in the brucite-like sheet. The results show that a natural sample from Snarum is an intergrowth of hydrotalcite (3R1 polytype) and manasseite (2H1 polytype) at a ratio of 77:23 (wt.%). An aluminian serpentine is associated with the hydrotalcite and manasseite minerals. The structure of a synthetic sample, Mg:Al = 2:1, was determined as space group
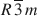 . For a few crystals in this sample, the octahedral cation distribution is compatible with the observed supercell (a = a′ √3). A second synthetic sample showed the presence of stacking faults and was described as a random layer sequence of two polytypes (3R and 2H).
. For a few crystals in this sample, the octahedral cation distribution is compatible with the observed supercell (a = a′ √3). A second synthetic sample showed the presence of stacking faults and was described as a random layer sequence of two polytypes (3R and 2H).
Acid Dissolution of Reduced-Charge Li- and Ni-Montmorillonites
- Helena Pálková, Jana Madejová, Dominique Righi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 133-142
-
- Article
- Export citation
-
Reduced-charge samples were prepared from Li+- and Ni2+-saturated SAz-1 montmorillonite by heating at 150 and 300°C for 24 h. X-ray diffraction analysis showed interstratification of non-swelling and swelling interlayers in LiS150, while fully expandable interlayers were found in NiS150. Heating at 300°C caused collapse of the interlayers in LiS300 in contrast to NiS300, for which some expandable layers were interstratified in the pseudo-pyrophyllite structure. The infrared (IR) spectra of heated Li-SAz showed new OH-stretching and overtone bands near 3670 cm−1 and 7170 cm−1 (1395 nm), respectively, indicating creation of local trioctahedral domains containing Li(I) in the previously vacant octahedra. No evidence of OH groups in trioctahedral coordination was found in the spectra of heated Ni-SAz. Nickel is supposed to be trapped in the hexagonal holes of the tetrahedral sheets. Reduction of the layer charge substantially affected the extent of the dissolution of SAz-1 montmorillonite in HCl. The MIR and NIR spectra of unheated Li- and Ni-SAz showed a substantial degradation of their structure after acid dissolution. New bands observed at 3744 cm−1 and 7314 cm−1 (1367 nm) were assigned to the vibrations of Si-OH groups formed upon acid treatment. These bands are a means of checking the extent of acid attack on smectites, even in cases when no differences are observed in the 1300−400 cm−1 spectral region (traditionally used to monitor this process). Both the IR spectra and solution analysis revealed that development of non-swelling interlayers in heated montmorillonites substantially reduced dissolution of these samples. The results obtained confirmed that acid attack of the smectite structure occurred at both interlayer surfaces and edges. If the accessibility of the layers for protons is low due to non-swelling interlayers, the dissolution was slower and took place mainly from the particle edges.
Geological and Mineralogical Characterization of Zeolites in Lacustrine Tuffs, Ngakuru, Taupo Volcanic Zone, New Zealand
- R. L. Brathwaite
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 589-598
-
- Article
- Export citation
-
Mordenite and clinoptilolite have replaced glass shards and pumice in vitric tuffs that are the products of ash fall-out into lake basins of late Quaternary age in the Taupo Volcanic Zone. The vitric tuffs are intercalated with siltstone and diatomite and overlie pumice-rich, rhyolitic ignimbrite. A Zr/TiO2-Nb/Y immobile element ratio plot indicates that the vitric tuffs, like the ignimbrite, are of rhyolitic composition. X-ray diffraction and scanning electron microscopy studies indicate that the mordenite and clinoptilolite are accompanied by authigenic K-feldspar and Opal-CT. The zeolites and other authigenic minerals are very fine grained (<10 µm), with open meshes of acicular mordenite crystals that result in low densities (0.7–1.0 g cm−3) in mordenite-rich tuffs. From Pearce element ratio analysis of whole-rock chemical analyses, only Na and K appear to have been mobilized during alteration. The zeolite deposits are associated with sinter, hydrothermal eruption breccias and silicified fault breccias that represent surface or near-surface manifestations of geothermal activity. Plant material extracted from a sinter overlying one of the deposits has a 14C age of 8498±60 BP, which is interpreted to be the age of zeolite deposition for this deposit. Mordenite and clinoptilolite occur in the lower-T (60–110°C) parts of some active or recently active geothermal systems elsewhere in the Taupo Volcanic Zone. The main fluid in these systems is weakly saline (alkali-chloride) water heated by geothermal activity. The Ngakuru zeolite deposits are interpreted as the products of the reaction of vitric tuffs with this type of water in the near-surface part of recently active geothermal systems.
The 10 Å to 7 Å Halloysite Transition in a Tropical Soil Sequence, Costa Rica
- Christopher Q. Kautz, Peter C. Ryan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 252-263
-
- Article
- Export citation
-
Soils developed on Pleistocene andesitic lava flows and fluvial detritus in the Atlantic coastal plain of Costa Rica display a clay mineral assemblage that includes 10 Å and 7 Å halloysite and lesser amounts of kaolinite and dioctahedral vermiculite. Other secondary minerals include gibbsite, goethite, hematite, maghemite, allophane and amorphous Al hydroxides. Active floodplain soils are dominated by 10 Å halloysite and contain less allophane, while soil clays from Pleistocene terraces consist of a mixture of 10 Å and 7 Å halloysite as well as less dioctahedral vermiculite, kaolinite, and amorphous Al hydroxides. Residual soils formed on Pleistocene lava flows are dominated by 7 Å halloysite with less abundant kaolinite, dioctahedral vermiculite, 10 Å halloysite and amorphous Al hydroxides. This sequence suggests transformations of 10 Å halloysite to 7 Å halloysite and allophane to amorphous Al hydroxides with time. The presence of 10 Å halloysite in Pleistocene terrace soils implies slow reaction rates or metastability.
Quantitative X-ray diffraction (QXRD) analysis indicates a decrease in the amount of plagioclase feldspar from 34 wt.% in the 1–2 year floodplain to 0–1.6% in terrace and residual soils. Plagioclase weathering is paralleled by the formation of dioctahedral clay, allophane and Al hydroxides. Analysis by QXRD also indicates that crystalline minerals comprise 70–95% of the soil fraction, implying 5–30% X-ray-amorphous material. These data are verified by selective extraction using ammonium oxalate, which indicates 8–30% amorphous material. Chemical analysis of the extractant by inductively coupled plasmaatomic emission spectrometry indicates that allophane (Al:Si ratios of 0.92–3.82) occurs in floodplain and some terrace soils while amorphous Al hydroxides appear to coexist with allophane in Pleistocene terrace and residual soils with Al:Si ratios of 6.53–8.53. Retention of Mg to a greater extent than Na, Ca and K suggests Mg incorporation into hydroxide sheets in dioctahedral vermiculite as well as substitution into hydroxides.
Diffusion of H2O in Smectite Gels: Obstruction Effects of Bound H2O Layers
- Yoshito Nakashima
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 9-22
-
- Article
- Export citation
-
In water-rich smectite gels, bound or less mobile H2O layers exist near negatively-charged clay platelets. These bound H2O layers are obstacles to the diffusion of unbound H2O molecules in the porespace, and therefore reduce the H2O self-diffusion coefficient, D, in the gel system as a whole. In this study, the self-diffusion coefficients of H2O molecules in water-rich gels of Na-rich smectites (montmorillonite, stevensite and hectorite) were measured by pulsed-gradient spin-echo proton nuclear magnetic resonance (NMR) to evaluate the effects of obstruction on D. The NMR results were interpreted using random-walk computer simulations which show that unbound H2O diffuses in the gels while avoiding randomly-placed obstacles (clay platelets sandwiched in immobilized bound H2O layers). A ratio (volume of the clay platelets and immobilized H2O layers)/(volume of clay platelets) was estimated for each water-rich gel. The results showed that the ratio was 8.92, 16.9, 3.32, 3.73 and 3.92 for Wyoming montmorillonite (⩽ 5.74 wt.% clay), Tsukinuno montmorillonite (⩽ 3.73 wt.% clay), synthetic stevensite (⩽ 8.97 wt.% clay), and two synthetic hectorite samples (⩽ 11.0 wt.% clay), respectively. The ratios suggest that the thickness of the immobilized H2O layers in the gels is 4.0, 8.0, 1.2, 1.4 and 1.5 nm, respectively, assuming that each clay particle in the gels consists of a single 1 nm-thick platelet. The present study confirmed that the obstruction effects of immobilized H2O layers near the clay surfaces are important in restricting the self-diffusion of unbound H2O in water-rich smectite gels.
Monte Carlo and Molecular Dynamics Simulation of Uranyl Adsorption on Montmorillonite Clay
- Omar F. Zaidan, Jeffery A. Greathouse, Roberto T. Pabalan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 372-381
-
- Article
- Export citation
-
We performed Monte Carlo and molecular dynamics simulations to investigate the interlayer structure of a uranyl-substituted smectite clay. Our clay model is a dioctahedral montmorillonite with negative charge sites in the octahedral sheet only. We simulated a wide range of interlayer water content (0 mg H2O/g clay — 260 mg H2O/g clay), but we were particularly interested in the two-layer hydrate that has been the focus of recent X-ray absorption experiments. Our simulation results for the two-layer hydrate of uranyl-montmorillonite yield a water content of 160 mg H2O/g clay and a layer spacing of 14.66 Å. Except at extremely low water content, uranyl cations are oriented nearly parallel to the surface normal in an outer-sphere complex. The first coordination shell consists of five water molecules with an average U-O distance of 2.45 Å, in good agreement with experimental data. At low water content, the cations can assume a perpendicular orientation to include surface oxygen atoms in the first coordination shell. Our molecular dynamics results show that
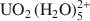 complexes translate within the clay pore through a jump diffusion process, and that first-shell water molecules are exchangeable and interchangeable.
complexes translate within the clay pore through a jump diffusion process, and that first-shell water molecules are exchangeable and interchangeable.
Desorption Behavior of Cd, Zn and Pb Sorbed on Hydroxyaluminum- and Hydroxyaluminosilicate-Montmorillonite Complexes
- Uttam Kumar Saha, Kōzō Iwasaki, Katsutoshi Sakurai
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 481-492
-
- Article
- Export citation
-
There is a clear gap in the understanding of the desorption patterns of metals sorbed on soils and clays, despite their importance in the mobility, transport and fate of metals in natural environments. In this study, we investigated the desorption behavior of Cd, Zn and Pb sorbed on montmorillonite (Mt) and on hydroxyaluminum (HyA)- and hydroxyaluminosilicate (HAS)-Mt complexes. At pH 6.5, 2.5 g L–1 of HyA-Mt and HAS-Mt sorbed almost all of the 10–6 M Cd, Zn or Pb, while Mt under the same condition sorbed ~48, 49 and 55% of the added Cd, Zn and Pb, respectively. Based on pH50 values, the selectivity of metal sorption on Mt was Pb > Zn > Cd, and on the complexes, it was Pb ≫ Zn = Cd. In general, larger fractions of sorbed metals were remobilized from Mt than from the complexes. Again, in comparison with Pb, larger fractions of sorbed Cd and Zn were remobilized from different clays. Reducing the pHs of the equilibrium sorption systems from a fixed point (6.5) to different points (6.0, 5.5, 5.0, 4.5, and 4.0) and from different points (6.5, 6.0, 5.5, 5.0, and 4.5) to a fixed point (4.0) both yielded hysteretic metal desorption patterns. The fractions of Cd and Zn desorbed through Na and Cu exchange from the clays, especially from the complexes, were very different, indicating the existence of cation exchangeable metal sorption sites of weak and strong affinities on the complexes. Based on the EDTA-extractable fractions of Cd and Zn from HAS–Mt and HyA-Mt, it appeared that HyA–metal bonds are stronger than the HAS–metal bonds. Compared with other agents, acetic acid remobilized the highest fractions of all metals irrespective of the type of clays, with a concomitant release of Al or Al + Si. The Pb-HyA/HAS-Mt bonds were, however, still much too strong to be broken substantially by this mechanism. The results accomplished in this study suggest further attention to the fundamental understanding of the mobility, fate, bioavailability and toxicity of the concerned metals in soils and related environments.
Structural Transformations of Clay Minerals in Soils of a Climosequence in an Italian Alpine Environment
- Aldo Mirabella, Markus Egli
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 264-278
-
- Article
- Export citation
-
Clays of a soil sequence with five profiles in the Val Genova (northern Italy) along an elevation gradient with climate ranging from moderate to Alpine were investigated with XRD using several diagnostic treatments. Smectites developed in the surface horizons of podzolic soils either from chlorite through the removal of hydroxy interlayers or from mica, which weathers in a first step to regularly or irregularly interstratified clay minerals. Citrate treatment allowed the detection of low-charge expandable minerals in the Bhs or Bs horizons. Therefore, the reduction of the charge of 2:1 clay minerals occurred before the removal of hydroxy polymers by fulvic acids and low-weight organic acids. Due to the more intense podzolization process near the tree line, the d060 region showed a temporal evolution of trioctahedral to dioctahedral mineral structures in the well developed Podzols. The pedogenic smectites of the E or Bhs horizons generally included one or several populations with various charges. In most cases, smectite was a heterogeneous mixture of montmorillonite and interstratified beidellite-montmorillonite. A pure beidellite phase could not be detected. The soils near the tree line, where weathering processes were most intense, had two main components: one with a charge >0.75, representing vermiculite-like minerals, and the other with a charge near 0.25, representing smectite. The charges of the beidellitic component and montmorillonite were almost equal. The higher the weathering state of the investigated soils, the lower was the layer charge of smectites.
Adsorption of AUCL4- by Kaolinites: Effect of pH, Temperature and Kaolinite Crystallinity
- Hanlie Hong, Zhenya Sun, Zhengyi Fu, Xinmin Min
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 493-501
-
- Article
- Export citation
-
Adsorption of the
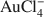 complex by kaolinites of different crystallinities (Kao-1 and Kao-2) from 100 mL of AuCl3·HCl·4H2O solutions containing 10,000, 500 and 50 µg/L Au and 1 g of kaolinite was measured at pH 3 to 9 at ambient temperature and 120°C. Adsorption from the 50, 500 and 10,000 µg Au/L solutions ranged from 64 to 100% at ambient temperature and from 68 to 100% at 120°C for both kaolinites. Adsorption was pH dependent with a maximum at pH <5 and a minimum at neutral and alkaline pHs. Up to 1 mg Au/g kaolinite was adsorbed by the kaolinites at both ambient temperature and 120°C. In a separate Au adsorption experiment using 100 mL of 4000 µg Au/L solutions and 0.02 to 1.0 g of Kao-1, up to 8.55 mg Au/ g of kaolinite was adsorbed. The pH dependence of Au adsorption suggests that surface complexation of Au to alumina sites at the edges of kaolinite particles might be involved. Protonation of kaolinite surface sites might facilitate adsorption of the anionic Au complex. Both kaolinites adsorbed ~100% of added Au at low pH values, but the less crystalline kaolinite (Kao-2) adsorbed more Au at high pH. Greater Au adsorption would be expected for the less crystalline Kao-2 sample if adsorption occurred at the edges of kaolinite particles.
complex by kaolinites of different crystallinities (Kao-1 and Kao-2) from 100 mL of AuCl3·HCl·4H2O solutions containing 10,000, 500 and 50 µg/L Au and 1 g of kaolinite was measured at pH 3 to 9 at ambient temperature and 120°C. Adsorption from the 50, 500 and 10,000 µg Au/L solutions ranged from 64 to 100% at ambient temperature and from 68 to 100% at 120°C for both kaolinites. Adsorption was pH dependent with a maximum at pH <5 and a minimum at neutral and alkaline pHs. Up to 1 mg Au/g kaolinite was adsorbed by the kaolinites at both ambient temperature and 120°C. In a separate Au adsorption experiment using 100 mL of 4000 µg Au/L solutions and 0.02 to 1.0 g of Kao-1, up to 8.55 mg Au/ g of kaolinite was adsorbed. The pH dependence of Au adsorption suggests that surface complexation of Au to alumina sites at the edges of kaolinite particles might be involved. Protonation of kaolinite surface sites might facilitate adsorption of the anionic Au complex. Both kaolinites adsorbed ~100% of added Au at low pH values, but the less crystalline kaolinite (Kao-2) adsorbed more Au at high pH. Greater Au adsorption would be expected for the less crystalline Kao-2 sample if adsorption occurred at the edges of kaolinite particles.
Laser Shadow Analysis of Particle-Size Distribution of Montmorillonites in Aqueous Suspensions
- S. Yariv, I. Lapides
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 23-32
-
- Article
- Export citation
-
Particle-size analysis (obtained by Galai CIS 1) was used to determine the following statistical parameters of aqueous suspensions of Na-, Ca- and Mn-montmorillonite: the size distributions, the diameters of the largest and of the median and mean particles, and the percentage of particles with diameters <1.5 µm. Dilution or shaking had almost no effect on the particle-size distribution curves and statistical parameters of Ca- and Mn-montmorillonites but the curves and the statistical parameters of Na-montmorillonite were very much affected by these treatments. The median and mean diameters of Na-montmorillonite range from 0.8 to 10.5 µm and 0.8 to 11.6 µm, respectively, the median and mean diameters of Ca-montmorillonite range from 1.5 to 3.6 µm and 2.12 to 4.2 µm, respectively, and those for Mn-montmorillonite from 1.2 to 2.3 µm and 1.5 to 2.5 µm, respectively. The presence of large particles of Na-montmorillonite was attributed to the extensive swelling of this clay in aqueous suspensions by osmotic water adsorption. The median and mean diameters of aged Na-montmorillonite suspensions indicate that swelling increases with dilution. The swelling of Ca- and Mn-montmorillonite, on the other hand, is limited and their particle size does not increase with dilution.
Adsorption of Two Quinolinecarboxylic Acid Herbicides on Homoionic Montmorillonites
- Alba Pusino, Antonio Gelsomino, Maria G. Fiori, Carlo Gessa
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 143-149
-
- Article
- Export citation
-
The adsorption of the herbicides quinmerac 7-chloro-3-methylquinoline-8-carboxylic acid (QMe) and quinclorac 3,7-dichloroquinoline-8-carboxylic acid (QCl) on homoionic Fe3+-, Al3+-, Cu2+-, Ca2+-, K+- and Na+-exchanged montmorillonite was studied in aqueous solution. Adsorption data were fitted to the logarithmic form of the Freundlich equation. Ca- and Na-exchanged montmorillonites were ineffective in the adsorption of QMe. On the other hand, the QMe adsorption on Fe-exchanged montmorillonite was rapid and the equilibrium was attained after 15 min. An H-type isotherm was observed for the QMe adsorption on Fe-clay, indicating a high affinity of the solute for the sorption sites and almost complete adsorption from dilute solution. On the other hand, the adsorption isotherm of QMe on Al- and K-clay was of the S-type. This shape suggests that the solvent molecules may compete for the sorption sites. A Fourier transform infrared (FTIR) study suggested that the adsorption mechanism of QMe on Fe-, Al- and K-clay involves the protonation of QMe molecule due to the acidic water surrounding the saturating cations. The greater acidity of Fe-clay compared with Al- and K-clay explains both the lower QMe adsorption observed on Al and K systems and the lack of adsorption on Na and Ca systems. In contrast, the formation of a Cu complex permitted QMe to be adsorbed to a large extent to Cu-clay as shown by FTIR analysis. The QCl was adsorbed only by Fe-clay and the adsorption isotherm of QCl on Fe clay was of the S-type. This finding is consistent with the lower basic character of the QCl molecule nitrogen. In fact, the replacement of the electron-releasing methyl group in QMe with an electron-withdrawing Cl atom to form QCl makes the nitrogen lone-pair electrons of the quinoline ring unavailable for either protonation or complexation.
The Mineralogy and Distribution of Zeolitic Tuffs in the Maramures Basin, Romania
- Jean-jacques Cochemé, Peter J. Leggo, Gheorghe Damian, Alexandrina Fulop, Béatrice Ledésert, Olivier Grauby
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 599-608
-
- Article
- Export citation
-
The Maramures Basin, in the Carpathian mountain belt of northern Romania on the border with the Ukraine, belongs to the eastern part of the Pannonian Basin. In the study area, extensional tectonic movements during the Miocene were coeval with silicic and intermediate volcanism in the inner part of the Eastern Carpathians. Throughout this region, explosive events have resulted in the deposition of pyroclastic flows and ash-fall deposits interbedded with marine sediments.
Several tuff units of Badenian (15–13.6 Ma) age occurring throughout the area are extensively zeolitized. These rocks occur as massive homogeneous beds, white to pale greenish-blue, and are commonly extensively jointed. In the Bârsana-Calinesti area and along the Morii Valley, two conspicuous tuff units that can be traced over many km are separated by a calcareous sandstone bed. Most tuffs have a vitroclastic texture in which former glass shards are pseudomorphed by clay minerals and clinoptilolite. Opal-CT commonly occurs as clumps of radiating rods that produce a spherical morphology. Also, rare celadonite is found in the lower greenish tuffs. Pyrogenic crystal fragments are quartz, plagioclase and biotite. Folded muscovite plates and fragments of basement rocks are dominant among the lithic clasts. Above the Bârsana Formation, a second series of white zeolitized tuff, the Ocna Sugatag Formation, is represented by at least two different units overlying an evaporite salt deposit. A large outcrop of a massive white tuff at this locality contains abundant fine-grained clinoptilolite and cation-exchange capacity values of >160 meq/100 g. Clinoptilolite-Ca is also present in the Sighetu tuff unit in the northern part of the Maramures Basin where a distinctive horizon contains plant remains preserved in spherical concretions. Plant material and algal limestones in the same succession strongly suggest that the marine depositional environment was close inshore, and shallow-rather than deep-water conditions are inferred. A mineralogically similar, unaltered, volcanic tuff found in the Coas area suggests that the precursor glass was rhyolitic (72–74% SiO2) with a high-K calc-alkaline affinity. We conclude that pervasive zeolitization is due to the interaction between seawater and vitroclasts at an early stage after deposition.
Characterization of Microbially Fe(III)-Reduced Nontronite: Environmental Cell-Transmission Electron Microscopy Study
- Jin-wook Kim, Yoko Furukawa, Tyrone L. Daulton, Dawn Lavoie, Steven W. Newell
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 382-389
-
- Article
- Export citation
-
Microstructural changes induced by the microbial reduction of Fe(III) in nontronite by Shewanella oneidensis were studied using environmental cell (EC)-transmission electron microscopy (TEM), conventional TEM, and X-ray powder diffraction (XRD). Direct observations of clays by EC-TEM in their hydrated state allowed for the first time an accurate and unambiguous TEM measurement of basal layer spacings and the contraction of layer spacing caused by microbial effects, most likely those of Fe(III) reduction. Non-reduced and Fe(III)-reduced nontronite, observed by EC-TEM, exhibited fringes with mean d001 spacings of 1.50 nm (standard deviation, σ = 0.08 nm) and 1.26 nm (σ = 0.10 nm), respectively. In comparison, the same samples embedded with Nanoplast resin, sectioned by microtome, and observed using conventional TEM, displayed layer spacings of 1.0–1.1 nm (non-reduced) and 1.0 nm (reduced). The results from Nanoplast-embedded samples are typical of conventional TEM studies, which have measured nearly identical layer spacings regardless of Fe oxidation state. Following Fe(III) reduction, both EC- and conventional TEM showed an increase in the order of nontronite selected area electron diffraction patterns while the images exhibited fewer wavy fringes and fewer layer terminations. An increase in stacking order in reduced nontronite was also suggested by XRD measurements. In particular, the ratio of the valley to peak intensity (v/p) of the 1.7 nm basal 001 peak of ethylene glycolated nontronite was measured at 0.65 (non-reduced) and 0.85 (microbially reduced).
Intercalation and Surface Modification of Smectite by Two Non-Ionic Surfactants
- Youjun Deng, Joe B. Dixon, G. Norman White
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 150-161
-
- Article
- Export citation
-
Non-ionic surfactants Brij 56 and Igepal CO 720, containing hydrophilic poly(ethylene oxide) (PEO) segments, expanded smectite from 1.5 nm to 1.7 nm at room temperature. The surfactant-smectite composites had larger layer spacings than Ca-smectite after heat treatment. The surfactant-smectite composites were solvated and expanded to 1.8–1.9 nm by polar solvents, glycerol and water, but were not affected by the non-polar or weakly polar solvents, toluene, hexane or octanol. The hydrophilic PEO segments of non-ionic surfactants would logically access the interlayer spaces of smectite whereas the hydrophobic segments extend away from the mineral. The molecular structure and solvation properties suggest that the surfactant molecules are probably concentrated in the margin area of the interlayer galleries forming an annular ring structure between two neighboring silicate sheets. Only two layers or less of the surfactants could access the interlayer galleries of smectite and layer spacings did not exceed 1.8 nm even where excess surfactant was introduced into the composites. The layer spacings of the surfactant-smectite composites were well preserved during water or electrolyte solution washings, indicating stability of most non-ionic surfactant molecules in the interlayer galleries even though ∼30% of the adsorbed Igepal CO 720 was desorbed by exhaustive washing. The non-ionic surfactant treatment preserved >80% of the CEC of the smectite. The interlayer cations of the resulting surfactant-smectite were exchangeable as in the untreated smectite. Therefore, the non-ionic surfactant-smectite was much more efficient at removing heavy metal ions than activated carbon or cationic surfactant-treated smectite. The surfactant-smectite composites effectively removed aromatic chlorophenols from a pH 4.9 acetate buffer solution while untreated smectite did not adsorb these molecules. The enhanced adsorption of the aromatic compounds is attributed to the aliphatic segments of the two surfactants.
Exchangeable Ion and Thermal Treatment Effects on Basal Spacings of Al-Hydroxy Pillared Montmorillonites
- Aluísio Sousa Reis, Jr., José Domingos Ardisson
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 33-40
-
- Article
- Export citation
-
Al-hydroxy intercalated clays (ALHICs) obtained from different parent clays were used to investigate the interactions between oligomers and clay surface layers.
The thermal stability of ALHICs obtained from natural, Cs-, Ca-, Ba-, Fe-, Cu- or Ce-montmorillonites has been investigated by studying the relationship between basal spacing and calcination temperature. X-ray diffraction has shown that the basal spacing of ALHICs obtained from Cu-montmorillonite calcined at 550°C is 13.4 Å, different from basal spacings of ALHICs obtained from Fe-montmorillonite (16.0 Å) and other parent clays (16.7 Å). Thermograms for AlHICs obtained from natural, Fe- and Ce-montmorillonites displayed distinct steps at 225 and 650°C, attributed to the dehydration of Al13 oligomers, and the dehydroxylation of the surface layer, respectively. By contrast, thermograms of ALHICs obtained from Cu-montmorillonite displayed one step between 250 and 700°C for both dehydration and dehydroxylation. Mössbauer parameters showed that Fe3+ octahedra in octahedral sheets are distorted in pillared interlayered clay (PILC) obtained from Cu-montmorillonite and undistorted in that obtained from Fe-montmorillonite. The difference in thermal stability for the various ALHICs is attributed to the retention of some of the original cations after intercalation with Al13 oligomers, which induces several interactions between the oligomers and the clay surface layers.