Velo-cardio-facial syndrome (VCFS) is a genetic disorder associated with characteristic dysmorphology, cleft palate, cardiac anomalies and major psychiatric disorders, including schizophrenia (Reference Shprintzen, Goldberg and LewinShprintzen et al, 1978; Reference Murphy, Jones and OwenMurphy et al, 1999). The frequencies of the disorder have been reported to be 1/4000 (Reference Wilson, Cross and WrenWilson et al, 1994) and 1/9700 live births (Reference du Montcel, Mendizabal and Aymedu Montcel et al, 1996), but both of these frequencies were only minimum estimates. The disorder is associated with interstitial deletions of chromosome 22q11; 87% of cases share a 3 Mb deletion, 8% have a smaller nested 1.5 Mb deletion, 4% have one of two other nested deletions and the remainder have unique deletions (Reference Shaikh, Kurahashi and SaittaShaikh et al, 2000) (Fig. 1). It has been proposed that deletions in this region are facilitated by the presence of low copy repeats (LCR22) at the boundaries of the various deletions (Reference Carlson, Sirotkin and PanditaCarlson et al, 1997). Most cases occur sporadically in the population, indicating that the 22q11 region is highly prone to rearrangement.
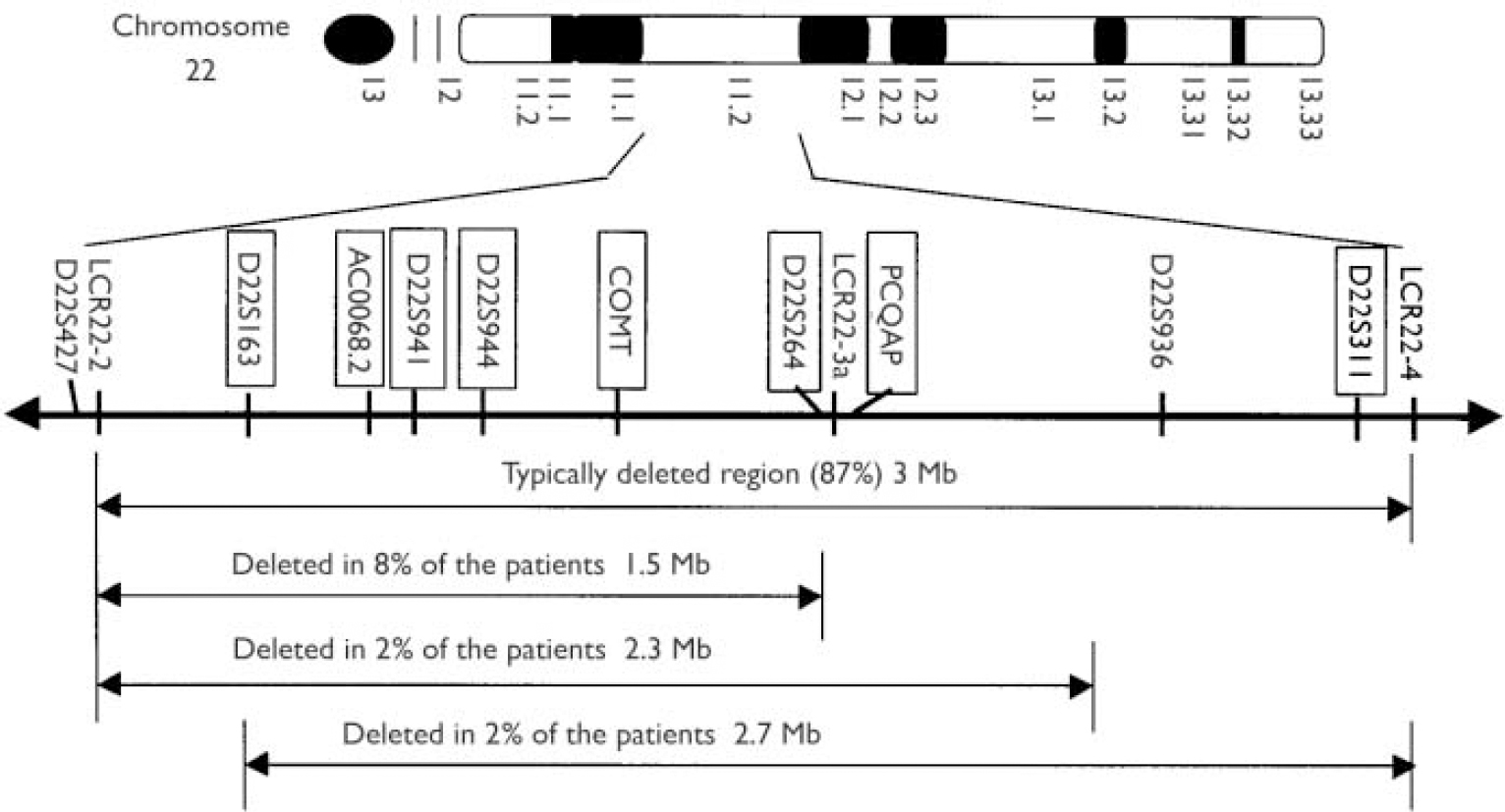
Fig. 1 Distribution of the genotyped markers (in blocks) in the velo-cardio-facial syndrome (VCFS) region on chromosome 22q11.2. LCR, low copy repeat.
Background
Several studies of individuals with VCFS have reported an increased incidence of psychosis, particularly schizophrenia (Reference Shprintzen, Goldberg and Golding-KushnerShprintzen et al, 1992; Reference Karayiorgou, Morris and MorrowKarayiorgou et al, 1995; Reference Papolos, Faedda and VeitPapolos et al, 1996; Reference Gothelf, Frisch and MunitzGothelf et al, 1997; Reference Bassett, Hodgkinson and ChowBassett et al, 1998; Reference Murphy, Jones and OwenMurphy et al, 1999; Reference Usiskin, Nicolson and KrasnewichUsiskin et al, 1999). The rate of VCFS in patients with schizophrenia is reported to be 0.33–2% (Reference Chow, Merce and WingChow et al, 1997; Reference Karayiorgou, Galke and BudarfKarayiorgou et al, 1997; Reference Arinami, Ohtsuki and TakaseArinami et al, 2001), compared with the frequency in the general population of only ∼0.025% (Reference Wilson, Cross and WrenWilson et al, 1994). When individuals with schizophrenia have been selected for the presence of two or more clinical features consistent with VCFS (palatal, cardiac, facial or other congenital anomalies, and/or learning difficulties), 22q11 deletions have been identified in 20–59% of cases (Reference Gothelf, Frisch and MunitzGothelf et al, 1997; Reference Bassett, Hodgkinson and ChowBassett et al, 1998).
Recently, it has been suggested that VCFS might be especially common in individuals with psychosis of early onset. Papolos et al (Reference Papolos, Faedda and Veit1996) reported that bipolar illness associated with VCFS usually has a childhood onset, whereas Usiskin et al (Reference Usiskin, Nicolson and Krasnewich1999) reported that 3/47 (6.4%) patients with a very-early-onset diagnosis of schizophrenia (<13 years) had VCFS deletions.
Aim
To test the hypothesis that there is an increased rate of VCFS among patients with early-onset psychosis, we screened 192 patients whose first psychotic episode had occurred before their 18th birthday (referred to as juvenile-onset cases) for 22q11 deletions. We postulated that more than 1% of patients would have such deletions. We also screened 329 patients with schizophrenia whose age at onset had been after their 18th birthday, to estimate the frequency of VCFS deletions in a more typical sample of cases.
METHOD
Subjects
All patients were recruited as part of our ongoing genetic association studies. The juvenile-onset subjects were recruited in the UK and Bulgaria and had an age at onset before their 18th birthday. Age-at-onset of illness was defined as the age of first occurrence of delusions, hallucinations or mania. In order to determine age at onset reliably, we only included information that was present in concurrent hospital or out-patient records. The Bulgarian sample consisted of 55 patients with schizophrenia (age at onset=9–17 years, mean=15.6 years, s.d.=1.6; 25 males and 30 females) and 32 patients with bipolar I affective disorder or schizoaffective disorder, either manic or bipolar type (age at onset=13–17 years, mean=15.8 years, s.d.=1.2; 11 males and 21 females). The UK sample consisted of 19 cases of bipolar I disorder (White British origin, age at onset=8–17 years, mean=15.6 years, s.d.=2.2; 4 males and 15 females) and 86 unrelated schizophrenia cases (age at onset=7–17 years, mean=15.6 years, s.d.=1.9; 56 males and 30 females). The age distribution of the juvenile cases is given in Table 1.
Table 1 Age distribution of the juvenile-onset cases of psychosis

| Age at onset (years) | Schizophrenia, Bulgaria | Bipolar 1, Bulgaria | Schizophrenia, UK | Bipolar 1, UK | Total cases |
|---|---|---|---|---|---|
| 7 | - | - | 1 | - | 1 (0.5%) |
| 8 | - | - | - | 1 | 1 (0.5%) |
| 9 | 1 | - | - | - | 1 (0.5%) |
| 10 | - | - | 2 | - | 2 (1.0%) |
| 12 | 1 | - | 2 | - | 3 (1.6%) |
| 13 | 3 | 1 | 4 | 1 | 9 (4.7%) |
| 14 | 4 | 5 | 11 | 2 | 22 (11.5%) |
| 15 | 14 | 5 | 16 | - | 35 (18.2%) |
| 16 | 13 | 10 | 12 | 8 | 43 (22.4%) |
| 17 | 19 | 11 | 38 | 7 | 75 (39.1%) |
| Total | 55 | 32 | 86 | 19 | 192 |
The remaining subjects were 329 unrelated UK patients with schizophrenia who did not satisfy our criteria for juvenile onset, either because their age at onset was clearly more than 18 years or because there was insufficient information available to define onset precisely. Of these, 244 were male and 85 were female. Subjects with an IQ <70 were excluded. In Bulgaria, patients were recruited by their own psychiatrists from both in- and out-patient settings in the capital Sofia and two major towns and their surrounding districts in the south and south-west of the country. In the UK, patients were recruited via psychiatrists from in-patient and out-patient services from a number of centres in Wales and England.
All patients were interviewed with an abbreviated version of the Schedules for Clinical Assessment in Neuropsychiatry (SCAN; Reference Wing, Babor and BrughaWing et al, 1990) and a best-estimate consensus diagnosis was reached on the basis of the interview and psychiatric hospital notes by two experienced researchers. The DSM–IV (American Psychiatric Association, 1994) diagnostic criteria were used in the study. The teams in Bulgaria and the UK used the same clinical instruments. The consensus diagnoses in Bulgaria were made in each case by G.K. and I.N. The UK sample was collected over a period of several years by a number of different fieldworkers. A consensus diagnosis had to be reached by two of them for a patient to be included in the study. All patients were interviewed when they were adults.
We also genotyped a sample of 444 healthy blood donors who had been broadly matched for gender and age with the patients from the general and juvenile samples from the UK population. The blood donors were recruited from the UK blood transfusion service in Cardiff, Wales. All patients and controls signed an informed consent form for participation in genetic association studies.
Genotyping
We screened for the presence of deletions by genotyping five microsatellite markers and one single nucleotide polymorphism in the deleted VCFS region (Fig. 1). Individuals who have a deletion are expected to have only one allele for each of these markers because they have only one copy of the chromosome 22 for this region. The chances of an individual having only one allele of any one marker but having two copies of that marker (i.e. being homozygous for that marker) depend on the allele frequencies and number of alleles of the marker. The heterozygosity (the frequency of observing individuals with two different alleles) of the markers that we chose ranged from ∼30% for PCQAP to 89% for D22S264. The probability that an individual is homozygous for each of the six markers in the absence of deletions is low (∼0.1%), therefore we expected that this method would allow us to end up with only a small number of individuals who were potential candidates to have deletions. We intended to confirm the presence of a deletion by fluorescence in situ hybridisation (FISH) if we had available cell lines from that individual, or by semi-quantitative polymerase chain reaction (PCR; see below). Our screening method is much more cost-effective compared with performing FISH on all the samples.
For the juvenile cases we used three known microsatellite markers (D22S264, D22S941, D22S944), one CA repeat that we identified from the genomic sequence of the clone AC000068.2 (indicated as AC0068.2 in Fig. 1) and one CAG repeat within the gene PCQAP (described by Reference De Luca, Conti and GrifoneDe Luca et al, 2003). This ensured that even some of the smaller, less common deletions were covered with a sufficient number of polymorphic markers. All five markers are in the typically deleted region (3 Mb) and, with the exception of PCQAP, are also within the less common region (1.5 Mb) deleted in 8% of the patients with VCFS (Fig. 1). The single nucleotide polymorphism that we used is a well-known marker within the catechol-O-methyltransferase (COMT) gene (Val158Met) that has been reported in numerous studies in the past (e.g. Reference Kirov, Murphy and ArranzKirov et al, 1998). It has a heterozygosity of ∼50% because the two alleles have an almost equal frequency in the UK population.
For the patients with schizophrenia having an age at onset of more than 18 years we used five microsatellite markers (D22S1638, D22S941, D22S944, D22S264, D22S311) and the Val158Met single nucleotide polymorphism at COMT. All the microsatellites are within the common (3 Mb) deleted region.
The microsatellites were amplified using standard PCRs with forward primers 5′-labelled with fluorescent dyes (FAM, HEX or TET). The fragments were loaded together and resolved on an AB1373 automated sequencer.
Confirmation of VCFS deletion
To confirm the presence of VCFS deletion we used semi-quantitative PCR. For this assay we simultaneously amplified two fragments in the same reaction. One fragment was chosen within exon 5 of COMT (which should be deleted in VCFS cases) and the other fragment was chosen from exon 11 of the ABCG1 gene on chromosome 21. Both genes have been screened extensively in the past for genetic variants: the ABCG1 gene by us (Reference Kirov, Lowry and StephensKirov et al, 2001) and the COMT gene by a number of teams. This ensured that there would be no common polymorphisms within these fragments that can interfere with the reaction. Primer sequences for the COMT fragment were:
forward: CTGTTCCAGGTCACCCTTGT reverse: CAGGTACCGGTCCTTCCAGT
For ABCG1 we used the following primers:
forward: TCATTGGCCTGCTGTACTTG reverse: CAGCGGTCTGTCACTCACAT
The forward primer of each pair was 5′-labelled with a fluorescent dye (FAM). The fragment from COMT amplified a product 120 bp in size, whereas the ABCG1 amplified a product of 141 bp. All four primers were added to the same PCR reaction, thus ensuring identical conditions for the amplification of both fragments. We optimised the reaction by reducing the number of cycles so that it was terminated during the exponential phase of the PCR, during which the amount of PCR product is dependent upon the starting number of DNA template molecules. The PCR products were quantitated on an ABI373 automated sequencer. If a person has a single copy of COMT, then the ratio of the amounts of the PCR products representing COMT and ABCG1 is ∼50% that of the ratio obtained from an individual with two copies of COMT (Fig. 2).

Fig. 2 Overlaid images of the products of the COMT (left) and ABCG1 (right) fragments of three individuals. The lower two traces (almost identical) of the COMT fragment are from one known velo-cardio-facial syndrome (VCFS) case and the patient with schizophrenia that we tested. The size of the peaks is ∼ 50% smaller than the peak of the control (top trace).
RESULTS
To confirm that our approach detects VCFS deletions, we genotyped a sample of 45 patients with known VCFS deletions previously diagnosed using FISH (the sample is fully described by Reference Murphy, Jones and OwenMurphy et al, 1999). We used five microsatellites (D22S944, D22S1638, D22S941, D22S264, D22S311) and the COMT Val158Met single nucleotide polymorphism. Our approach correctly identified all 45 known cases of VCFS deletions by showing that they had only one allele for each marker within the deleted region.
In the test sample, only one individual (a patient with schizophrenia) gave a pattern of homozygosity for all markers tested. This effectively excludes the presence of deletions in the rest of the population. The homozygous individual had been genotyped also for a number of single nucleotide polymorphisms in three genes from the VCFS region as part of our ongoing genetic research projects. We observed that he was homozygous for every single nucleotide polymorphism genotyped in the COMT, ARVCF and TBX1 genes (a total of 20 single nucleotide polymorphisms), thus adding further evidence that he had a VCFS deletion. We did not have a cell line for this individual and were unable to confirm the deletion by FISH. We therefore performed the semi-quantitative PCR described above. To provide positive and negative controls for the assay, we also genotyped known individuals with VCFS and those without VCFS, respectively. The overlaid traces of one control, one known VCFS case and the suspected VCFS case are shown in Fig. 2. This experiment confirmed that the individual had a VCFS deletion because his trace was similar to that of the known VCFS case and about half the size of the COMT fragment of the non-deleted case.
DISCUSSION
Prevalence of VCFS in patients with schizophrenia
The frequency of 22q11 deletions is estimated to be at least 1:4000 in the general population and approximately 30% of people with this deletion develop symptoms of psychosis, especially schizophrenia (Reference Pulver, Nestadt and GoldbergPulver et al, 1994; Reference Murphy, Jones and OwenMurphy et al, 1999). Karayiorgou et al (Reference Karayiorgou, Galke and Budarf1997) reported that 3/207 randomly selected patients with schizophrenia had a 22q11 deletion and on retrospective assessment had facial features of VCFS (this report includes the 100 patients reported earlier by this group (Reference Karayiorgou, Morris and MorrowKarayiorgou et al, 1995)). Chow et al (Reference Chow, Merce and Wing1997) reported that 2/100 randomly selected Chinese patients with schizophrenia had 22q11.2 deletions. Arinami et al (Reference Arinami, Ohtsuki and Takase2001) identified one patient with a 22q11.2 deletion among 300 randomly selected patients with schizophrenia. In the present study, we did not identify a single patient with a 22q11 deletion among 192 patients with early-onset psychosis and therefore the present study does not provide support for the hypothesis that there is a higher incidence of 22q11 deletions among patients with early-onset psychosis. We identified only one such case among an additional sample of 329 schizophrenia cases with a nonjuvenile age at onset of illness, which is a lower rate than in the previous reports.
The patient with VCFS deletion that we identified did not have a recognised low IQ but had left school with no qualifications. He did not have children. According to his own account, he developed signs of illness at around the age of 17 years, when he thought that his father was the Devil (there was no hospital documentation to support this, so he did not satisfy our strict criteria for inclusion in the juvenile psychosis sample). His first hospitalisation was at the age of 21 years. Over the years he had exhibited auditory and olfactory hallucinations, thought withdrawal, bizarre delusions, paranoid ideas and suicidal ideation.
Statistical consideration
If the frequency of 22q11 deletions was 2% in juvenile-onset samples ascertained according to our criteria then we would have had 98% power to detect at least one observation of a deletion. If the true rate is 1%, the corresponding power was 85%. Our data suggest that if people with psychosis, including juvenile-onset cases, have an increased risk of 22q11 deletions it is only a modest increase. On combining our data with the three previous studies that estimated the frequency of VCFS in subjects ascertained for schizophrenia without major bias towards co-ascertaining subjects with VCFS, there are seven observations of 22q11 deletions out of 1077 individuals with schizophrenia, which is a prevalence of ∼0.65% (we exclude individuals with bipolar disorder in our sample from the overall figure).
If the prevalence of VCFS is 1:4000 in the general population and 24% of these people develop schizophrenia (Reference Murphy, Jones and OwenMurphy et al, 1999), then 1:16 666 of the general population should have both schizophrenia and VCFS. Assuming that the prevalence of schizophrenia is 1% in the general population, we would expect that 1:166 or 0.6% of randomly ascertained subjects with schizophrenia would have VCFS. The frequently quoted figure of 1/4000 for VCFS in the general population is only an approximation based on the rate of congenital heart defects (Reference Wilson, Cross and WrenWilson et al, 1994). The only other large study (Reference du Montcel, Mendizabal and Aymedu Montcel et al, 1996) reported a frequency of 1:9700 based on symptomatic cases from a Birth Defects Registry in France. Both studies stressed that the rate of milder cases is likely to be higher but the precise incidence is not known, therefore our estimate that an unselected sample of 0.6% of patients with schizophrenia should also have VCFS can be only an approximation. However, it concurs well with the figure of 0.65% that represents the combined world literature. Further large-scale screens are required on samples ascertained using epidemiological principles to obtain a precise estimate of the risk for this deletion in typical schizophrenia.
Age at onset and IQ
One possible characteristic of our sample that may have reduced the prevalence of deletions is that individuals with an IQ of <70 were excluded. In the study of Arinami et al (Reference Arinami, Ohtsuki and Takase2001), the patient with schizophrenia and VCFS deletion had an IQ of 61. In the study of Karayiorgou et al (Reference Karayiorgou, Galke and Budarf1997), two patients with schizophrenia had a history of learning problems as children. One of the two patients found by Chow et al (Reference Chow, Merce and Wing1997) also had ‘mental retardation’. Thus, a stricter interpretation of our data is that a VCFS deletion is an uncommon cause for schizophrenia in patients with a normal IQ. It is also important to note that all three individuals with childhood-onset schizophrenia who were found to have VCFS in the study of Usiskin et al (Reference Usiskin, Nicolson and Krasnewich1999) developed signs of psychosis before their 13th birthday. Only eight of our patients had an onset this young (Table 1). It is therefore possible that the prevalence of 22q11 deletions is higher in cases with very early onset, although further work is needed in this unusual group of cases. The patient that we identified with VCFS had an illness onset at the age of 17 years, but, even if we were to classify this patient as having a juvenile-onset psychosis, the rate of VCFS among this population is still lower than previously proposed.
Possible sources of bias
It is not known whether publication bias has influenced the previous estimates of VCFS rates in patients with schizophrenia but it is more likely, in principle, that a small study that detected at least one case of VCFS will be published than a small study that did not. This bias will act to inflate the reported prevalence of VCFS in the population with schizophrenia. On the other hand, our genetic study might be biased against finding VCFS cases because we did not include patients with known low IQ, as discussed above. Another factor that will reduce the observed prevalence of VCFS is the increased mortality in these patients, due largely to congenital heart disease. This would have been even greater 30 or 40 years ago, when many potential current individuals with VCFS would have been born, so that they would be less likely to reach the average age of ascertainment.
Criteria for screening for VCFS
Our data indicate that screening for VCFS is not justified in the general population of patients with psychosis, even in those with an early onset, at least when the first episode of psychosis occurs after the age of 13 years. The main criteria for screening in psychosis remain the presence of cleft lip and/or palate, characteristic dysmorphology, learning disability, congenital heart disease or hypocalcaemia (Reference Murphy and OwenMurphy & Owen, 1996; Reference Gothelf, Frisch and MunitzGothelf et al, 1997; Reference Bassett, Hodgkinson and ChowBassett et al, 1998).
Clinical Implications and Limitations
CLINICAL IMPLICATIONS
-
▪ The rate of velo-cardio-facial syndrome (VCFS) deletions in patients with psychosis but without learning difficulties is lower than previously reported and is in the region of 0.6%.
-
▪ The rate does not appear to be increased substantially in cases with onset between 13 and 18 years.
-
▪ Patients with psychosis should not be screened routinely for VCFS deletions unless they have a history of learning difficulties, specific clinical features of VCFS or possibly onset below the age of 13 years.
LIMITATIONS
-
▪ We did not include patients with learning difficulties or specific clinical features of VCFS deletions.
-
▪ We cannot estimate the exact prevalence of this disorder in patients with psychosis. Much larger samples would be required for this purpose.






eLetters
No eLetters have been published for this article.