Volume 68 - Issue 1 - February 2020
Article
Direct Synthesis of 5A Zeolite From Palygorskite: The Influence of Crystallization Directing Agent on the Separation Performance for Hexane Isomers
- Le Chen, Jun-Yan Qian, Chuang Yang, Ping-Ping Xu, Dan-Dan Zhu, Jing Zhong, Ming-Yang He, Qun Chen, Zhi-Hui Zhang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-8
-
- Article
- Export citation
-
The efficient separation of hexane isomers from the light naphtha fraction is a significant challenge in the petrochemical industry. 5A zeolite adsorbent is used commercially to sieve alkane isomers. In this study, 5A zeolites were synthesized using a low-cost natural clay mineral precursor, i.e. palygorskite (PAL), with the addition of crystallization directing agent (CDA). By varying the mass ratio of CDA/deionized water, 5A zeolites were obtained as CDA-5%, CDA-7.5%, and CDA-10%. All products were submicron particles with an average particle size of 400–800 nm. A sieving test of CDA-induced 5A zeolites was carried out on hexane adsorbates including n-hexane (nHEX), 2-methylpentane (2MP), and 3-methylpentane (3MP). According to vapor-phase batch adsorption experiments, a significant equilibrium amount (0.149 g/g) of nHEX and only 0.0321 g/g 2MP and 0.0416 g/g 3MP were adsorbed on the 5A zeolite product with CDA-5%. The dynamic adsorption performance of 5A zeolite (CDA-5%) was evaluated by breakthrough curves of binary mixtures of nHEX/2MP and nHEX/3MP. Palygorskite 5A (PAL 5A) zeolite achieved maximum dynamic adsorption capacities of nHEX (0.16 g/g in both cases) at 200°C and 1.2 MPa total pressure. This work provided an economic alternative for the synthesis of 5A zeolites using natural clay minerals instead of chemical raw materials.
Original Paper
Surface Chemisty, Microstructure, and Rheology of Thixotropic 1-D Sepiolite Gels
- Pengfei Liu, Mingyong Du, Peta Clode, Hualong Li, Jishan Liu, Yee-Kwong Leong
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 9-22
-
- Article
- Export citation
-
The rheological properties of sepiolite gels in relation to solution chemistry, fiber charge, and microstructure are poorly understood. The purpose of this study was to bring more clarity to this topic by quantifying the effects of solution pH, ionic strength, and adsorbed tetrasodium pyrophosphate (TSPP) additive on rheological properties. The electrical charge on sepiolite fibers was investigated to explain the fiber interaction configuration observed in the microstructure. Fiber interaction forces and dynamics explained the ageing behavior of the gel. Sepiolite gels of only a few percent solids displayed long-time ageing behavior, which was manifested by an increasing yield stress with wait time and continued for weeks. The gel microstructure showed randomly orientated rigid fibers with cross configuration attraction. Each fiber experiences both attractive (van der Waals and heterogeneous charge) and repulsive (electric double layer) forces, and initially a net force. The repulsive force causes these fibers to orientate or move continually to achieve a state of force equilibrium and this process takes a long time. The Leong model describes this ageing behavior. For good fiber separation, high intensity probe sonication of the suspension was required. The yield stress increased with sonication time, solids loading, and temperature. The yield stress was absent at pH > 11 and increased to a maximum value at pH < 8. This maximum was insensitive to pH between 4 to 8, and ionic strength up to 1 M KCl. TSPP reduced this maximum and shifted the zero yield stress region to a lower pH, ~7. The zero yield stress state corresponded to a zeta potential with a minimum magnitude of 30 mV.
Acid-treated Clay Minerals as Catalysts for Dehydration of Methanol and Ethanol
- Monika Marosz, Andrzej Kowalczyk, Barbara Gil, Lucjan Chmielarz
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 23-37
-
- Article
- Export citation
-
The purification of clay minerals prior to their use as catalysts can escalate processing costs so methods are needed whereby less purification is necessary. One such potential method is acid treatment of the unpurified clay minerals. The main objectives of the present study were to develop the optimal acid-treatment conditions and to determine how the acidic properties of the modified clay samples influenced their catalytic capability toward the dehydration of ethanol and methanol. Clay mineral samples – allophane, palygorskite, and sepiolite, without purification – were acid treated (0.8 M HNO3; 95°C; 2, 8, or 24 h) and after calcination (500°C; 6 h) tested as catalysts for the conversion of methanol to dimethyl ether and of ethanol to diethyl ether and ethene. The changes in chemical and structural compositions as well as surface acidity of the mineral samples were analyzed and correlated with their catalytic performance. Among the samples studied, allophane was the most catalytically active in the dehydration of methanol to dimethyl ether. Acid treatment of this mineral sample decreased methanol conversion slightly. An opposite effect was found for ethanol dehydration to diethyl ether, where acid treatment increased catalytic activity of allophane. The differences in catalytic performance of the mineral samples were discussed with respect to the nature and concentration of acid sites.
Liquid-Phase Xylene Adsorption in Unary, Binary, and Ternary Solute Systems Using Raw and Ni2+ Ion-Exchanged Clinoptilolite: Experimental Study and Thermodynamic Assessment
- Atefeh Salarvand, Cavus Falamaki
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 38-49
-
- Article
- Export citation
-
The adsorptive behavior of clinoptilolite (Cpt) zeolite in its raw and nickel-exchanged form towards m-xylene/p-xylene/ethylbenzene unary, binary, and ternary mixtures was investigated. The motivation behind the research was to elucidate whether Cpt in its raw or ion-exchanged form could exhibit distinctive selective adsorption behavior. The natural Cpt (Si/Al atomic ratio = 4.59) was ion-exchanged twice with 0.5 M Ni(NO3)2 solution at 80°C for 6 h. Adsorption experiments were done at 40°C, using a 4 vol.% solution of the aromatic materials in iso-octane. The maximum amount of the total specific adsorption capacity for binary solute systems was ~0.8 and ~2.0 mmol g–1 for the raw and ion-exchanged Cpt, respectively. For the ternary solute systems, unexpectedly, this capacity increased to ~2.0 and ~3.0 mmol g–1, respectively. For binary mixtures, both forms of Cpt were selective for p-xylene. For ternary mixtures, both forms exhibited a clear selectivity for m-xylene but the Ni-exchanged Cpt was significantly higher over the concentration range studied. The unexpected increase in the adsorption capacity in ternary systems was attributed to the expulsion of tightly bound trace water molecules hindering the access of xylene molecules to the 10-member ring channels of the zeolite framework. Substitution of Mg2+ by Ni2+ in the 10-member ring channels enhanced the adsorption capacity by providing more space and stronger electrostatic interactions between the new cation and the polar m-xylene molecule.
Trace and Rare Earth Element Distribution and Mobility During Diagenetic Alteration of Volcanic Ash to Bentonite in Eastern Iranian Bentonite Deposits
- Alireza Namayandeh, Soroush Modabberi, Alberto López-Galindo
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 50-66
-
- Article
- Export citation
-
The chemical composition of the source magma along with the physiochemical conditions of the depositional environment are the main controlling factors in determining the behavior and mobility of trace elements and rare earth elements (REEs) during the transformation of volcanic ash to bentonite. The purpose of the present study was to determine the distribution pattern and mobility of trace elements and REEs in several bentonite deposits formed by diagenetic alteration of volcanic ash in shallow alkaline water in eastern Iran. Using geochemical and statistical data, the degree of weathering in the parent rocks and the distribution and mobility of trace elements and REEs during the alteration process at seven deposits (Chah-Taleb, Chah-Keshmir, Chah-Golestan, Chah-Pirouz, Gholeh-Gelia, Kharman-Sar, and Khal-Kooh) were studied. None of the parent rocks showed an advanced degree of depositional reworking and, therefore, their chemical composition is representative of the volcanic ash from which the bentonites were formed. In the chondrite-normalized REE patterns of both parent rocks and bentonites, the light rare earth elements (LREEs) were found to be enriched relative to the heavy rare earth elements (HREEs). The variation in δEu and δCe values suggested a high-temperature (<200°C), suboxic, aquatic environment in which the conversion of volcanic ash to bentonite occurred. In the bivariate correlation analysis, Si showed a strong inverse relationship with Al and LREEs, while large ion lithophile elements (LILEs), high field strength elements (HFSEs), HREEs, and LREEs displayed a positive correlation between the elements of their respective groups. The R2 values in the binary diagram of potential immobile elements against Al suggest a qualitative classification in which Ti, Gd, Ga, Pr, Tb, Nd, Sm, Ce, and Nb are considered immobile, and U, Dy, In, Sc, Hf, Zr, La, and Eu are considered to have had poor mobility during formation of the Eastern Iranian bentonite deposits.
Article
Quantification of Mixed-Layer Clays in Multiple Saturation States Using NEWMOD2: Implications for the Potassium Uplift Hypothesis in the SE United States
- Jason C. Austin, Daniel D. Richter, Paul A. Schroeder
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 67-80
-
- Article
- Export citation
-
Quantification of mineral assemblages in near-surface Earth materials is a challenge because of the often abundant and highly variable crystalline and chemical nature of discrete clay minerals. Further adding to this challenge is the occurrence of mixed-layer clay minerals, which is complicated because of the numerous possible combinations of clay layer types, as defined by their relative proportions and the ordering schemes. The problem of ensuring accurate quantification is important to understanding landscape evolution because mineral abundances have a large influence on ecosystem function. X-ray diffraction analysis of the variable cation-saturated clay fraction in soil and regolith from the Calhoun Critical Zone observatory near Clinton, South Carolina, USA, was coupled with modeling using NEWMOD2 to show that mixed-layer clays are often dominant components in the mineral assemblages. Deep samples in the profile (>6.5 m) contain mixed-layer kaolinite/smectite, kaolinite/illite-like, kaolinite-vermiculite, illite-like/biotite, and illite-like/vermiculite species (with ‘illite-like’ defined herein as Fe-oxidized 2:1 layer structure with a negative layer charge of ~0.75 per unit formula, i.e. weathered biotite). The 2:1 layers in the mixed-layer structures are proposed to serve as exchange sites for K+, which is known to cycle seasonally between plant biomass and subsurface weathering horizons. Forested landscapes have a greater number of 2:1 layer types than cultivated landscapes. Of two nearby cultivated sites, the one higher in landscape position has fewer 2:1 layer types. Bulk potassium concentrations for the forested and two cultivated sites show the greatest abundances in the surface forested site and lowest abundance in the surface upland cultivated site. These observations suggest that landscape use and landscape position are factors controlling the mixed-layer mineral assemblages in Kanhapludults typical of the S.E. United States Piedmont. These mixed-layer clays are key components of the proposed mechanism for K+ uplift concepts, whereby subsurface cation storage may occur in the interlayer sites (with increased negative 2:1 layer charge) during wetter reduced conditions of the winter season and as biomass decay releases cation nutrients. Cation release from the mixed-layer clays (by decreased 2:1 layer charge) occurs under drier oxidized conditions during the growing seasons as biota utilize cation nutrients. The types and abundances of mixed layers also reflect long-term geologic factors including dissolution/alteration of primary feldspar and biotite and the subsequent transformation and dissolution/precipitation reactions that operate within the soil horizons. Thus, the resulting mixed-layer clay mineral assemblages are often complex and heterogeneous at every depth within a profile and across landscapes. X-ray diffraction (XRD) assessment, using multiple cation saturation state and modeling, is essential for quantifying the clay mineral assemblage and pools for cation nutrients, such as potassium, in the critical zone.
Extremely Al-Depleted Chlorites From Dolomite Carbonatites of the Kovdor Ultramafic-Alkaline Complex, Kola Peninsula, Russia
- Nikita V. Chukanov, Maria G. Krzhizhanovskaya, Igor V. Pekov, Dmitry A. Varlamov, Konstantin V. Van, Vera N. Ermolaeva, Svetlana A. Vozchikova
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 81-88
-
- Article
- Export citation
-
The problem to be solved is whether Al is a necessary component of Fe-Mg chlorites. Very unusual Al-depleted and Fe-enriched trioctahedral chlorites with the empirical formulae Na0.05Ca0.05(Fe2+3.01Mg2.01Ti0.14Fe3+0.04)Σ6.00[(Si3.53Fe3+0.41Al0.06)Σ4.00O10](OH)8·nH2O (Sample 1) and Na0.05Ca0.01(Fe2+3.26Mg1.97Fe3+0.75Mn0.01Ti0.01)Σ6.00[(Si3.16Fe3+0.75Al0.09)Σ4.00O10](OH)8 (Sample 2) have been discovered in Al-depleted dolomite carbonatites of the Kovdor complex of ultramafic, alkaline rocks and carbonatites, Kola Peninsula, Russia. The presence of substantial amounts of Ti in Sample 1 is another unusual feature of this mineral. In both samples, chlorites are intimately intergrown with cronstedtite-1T which is an indication of a low stability of chlorite structure in the absence of aluminum in the tetrahedral sheet. The crystal structure of chlorite in Sample 1 was solved by the Rietveld method. The mineral is triclinic (IIb-4-module), space group C-
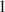 , a = 5.4153(4), b = 9.3805(7), c = 14.5743(12) Å, α = 90.137(5)°, β = 96.928(5)°, γ = 90.043(6)°, V = 734.95(10) Å3, and Z = 2. A problem to be solved is how stable are Al-free chlorites belonging to the clinochlore–chamosite solid-solution series and whether their existence in natural mineral assemblages is possible. The results obtained indicate that even though Al-depleted chlorites belonging to the clinochlore–chamosite solid-solution series exist in Nature as metastable phases, these minerals are extremely rare and much less stable than Al-poor serpentines.
, a = 5.4153(4), b = 9.3805(7), c = 14.5743(12) Å, α = 90.137(5)°, β = 96.928(5)°, γ = 90.043(6)°, V = 734.95(10) Å3, and Z = 2. A problem to be solved is how stable are Al-free chlorites belonging to the clinochlore–chamosite solid-solution series and whether their existence in natural mineral assemblages is possible. The results obtained indicate that even though Al-depleted chlorites belonging to the clinochlore–chamosite solid-solution series exist in Nature as metastable phases, these minerals are extremely rare and much less stable than Al-poor serpentines.