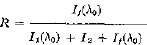Advances in X-ray Analysis, Sixteenth Annual Conference on Applications of X-ray Analysis, August 9-11, 1967
- This volume was published under a former title. See this journal's title history.
Other
Foreword
- H. G. Pfeiffer
-
- Published online by Cambridge University Press:
- 06 March 2019, p. vi
-
- Article
- Export citation
Preface
- G. R. Mallett, J. B. Newkirk
-
- Published online by Cambridge University Press:
- 06 March 2019, p. vii
-
- Article
- Export citation
Research Article
Recent Advances in Quantitative X-Ray Spectrometric Analysis by Solution Techniques
- Eugene P. Bertin
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 1-22
-
- Article
- Export citation
-
Usually X-ray spectrometric analyse? of samples in their original forms—solid, powder, small fabricated parts, liquid, etc.—are more rapid and convenient than analyses by any other method. Thus, the analyst is well advised to strive to analyze samples in their original form whenever practical. However, it is often necessary to reduce nonliquid samples to some other form, for example, when standards are unavailable in the original form, when the same substance is received for analysis in a variety of forms, when homogeneity or matrix effects are severe, or when an internal standard must be added. In such cases, samples and standards are often reduced to a powder, a fusion product, or a solution. If the decision is made to put the sample into solution, the many well-known advantages of solution techniques are realised, including: (1) homogeneity; (2) easy preparation of standards and blanks; (3) easy concentration, dilution, separation, and other treatment; (4) reduced matrix effects and wide choice of ways to deal with matrix effects; (5) wide choice of ways to present the specimen to the spectrometer; and (6) applicability of internal-standard, standard-addition or -dilution, indirect, absorption, and scatter methods. This paper reviews work reported since 1960 in which the specimen is presented to the spectrometer in liquid form. The review is not particularly critical and stresses experimental techniques and treatment of data rather than specific materials and results. The work is discussed in the following categories: (1) sensitivity ; (2) liquid-specimen cells; (3) interaction of primary beam and liquid specimens; (4) matrix effects; (5) indirect (association) analysis; (6) X-ray scatter methods; and (7) X-ray absorption-edge spectrometry.
X-Ray Fluorescence Spectroscopy in the Analysis of Ores, Minerals, and Waters*
- Harry J. Rose, Jr., Frank Cuttitta
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 23-39
-
- Article
- Export citation
-
X-ray fluorescence spectroscopy has been used in solving a wide variety of geologic problems involving mineral, ore, and water analysis. The technique has been a powerful analytical tool in the survey of mineral deposits, as a control to monitor ore flotation processes, for the semimicroanalysis of mineral separates and of rare new mineral species, and for the determination of trace elements in lake and saline waters. Many preparation techniques have been developed for the analysis of complex mineral systems, some combining X-Ray fluorescence with other analytical techniques to provide a complete analysis. These, coupled with improvements in instrumentation, have given the X-ray analyst a means of extending analytical ranges to the microgram level and to include elements that were previously not detectable. Significant advances in sample preparation and methods development have been made in the analysis of milligram quantities of complex geologic materials. The fusion and the solution of specimens appear to be the preferred methods of sample preparation. For samples that vary markedly in composition, the slope-ratio technique offers a new approach to solving matrix problems.
Common Sources of Error in Electron Probe Microanalysis
- Kurt F. J. Heinrich
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 40-55
-
- Article
- Export citation
-
In order to reduce the error of quantitative electron probe microanalysis, error sources in the preparation and measurement of specimens and standards must be minimized. These sources of error are described, and literature references for detailed study are given. A critical analysis is made of 150 analytical measurements of binary specimens previously discussed by Poole and Thomas. It is shown that the cases of serious errors reported by these authors are mainly due to poorly characterized or measured specimens and, in. some cases, to the omission of characteristic fluorescence corrections. If these sources of error are eliminated, a much more favorable error distribution can be obtained through relatively simple correction calculations. Further progress in quantitative micro probe analysis is dependent upon measurements under well-controlled conditions and standard materials of experimentally proven microhomogeneity and reliably determined composition.
X-Ray Spectrographs Analysis of Traces in Metals by Preconcentration Techniques
- C. M. Davis, Keith E. Burke, M. M. Yanak
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 56-62
-
- Article
- Export citation
-
Chemical separation techniques with their roots in classical analysis have been highly developed since the turn of the century. During the last two decades, X-ray spectrography has proven to be a very acceptable method of analysis because of the relative ease and rapidity of measurement of the intensity of characteristic wavelengths, the ready knowledge of the precision of the measurement, the facility of automating the analysis, and the nondestructive nature of the method. When chemical separation techniques are combined with X-ray spectrography, the problem of matrix effects is eliminated and the element being analyzed is substantially concentrated, which affords a means of performing trace element analyses. Published examples of preconcentration followed by X-ray measurement both outside and in the field of metallurgy are cited.
Theoretical Correction for Coexistent Elements in Fluorescent X-Ray Analysis of Alloy Steel
- Toshio Shiraiwa, Nobukatsu Fujino
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 63-94
-
- Article
- Export citation
-
With fluorescent X-ray analysis as routine work, the correction term for the coexistent element can be expressed as linear terms of the weight fraction of the element because the compositions of samples are limited to a small range. Usually those correction factors which require a great deal of work are obtained experimentally. The authors have obtained theoretical equations of fluorescent X-ray intensity which are in good agreement with experimental values. The linear correction factors are obtained from derivatives of those equations, and their values can be easily calculated with a computer. The experimental X-ray intensity versus the weight fractions is usually expressed as a line. However, the linear approximation is not correct over a wide range of the composition. The second derivative of the theoretical equation explains the deviation from the linear approximation and gives the range where the linear approximation is allowed. The calculations are applied to the analysis of stainless steels, several low-alloy steels, and iron ores, and experimental results are corrected by the calculated results. Correction factors for Ni Kα, Fe Kα, Cr Kα, Mn Kα, and Cu Kα in stainless steels and Cr Kα and Mn Kα in low-alloy steels are calculated for coexistent elements such as carbon, silicon, titanium, chromium, manganese, copper, niobium, and molybdenum. For example, standard deviations of chromium and manganese analyzed results in lowalloy steels decrease from 0.169 and 0.044% to0.030 and0.023%, respectively, with theoretical corrections. In the analysis of iron ore, the fluorescent X-ray intensity of iron is affected by combined oxygen, which is different for the various compounds of iron oxides, and other impurities such as alumina, silica, and lime. The correction factors of these are obtained by calculation, and the standard deviation decreases from 1.70 to 0.44% for 55.1 to 68.5% iron. It is found by experiment that the theoretical values have about 1 or 2% of relative errors, and their derivatives also have relative errors of the same order of magnitude. But the ranges of coexistent elements are usually small, a few percent at most in routine work, and the theoretical values can be used in practical analyses.
Micro Fluorescent X-Ray Analyzer
- Toshio Shiraiwa, Nobukatsu Fujino
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 95-104
-
- Article
- Export citation
-
A micro fluorescent X-ray analyzer with a focusing type of spectrometer has been developed to analyze samples of small amounts such as extracted inclusions or precipitates from metals or small areas in samples from 0.1 to 2.0 mm in diameter. This instrument is expected especially to analyze powder samples of small quantity because average values from such samples can be obtained and because surface conditions of the samples scarcely affect the results compared with their effect in electron probs microanalysis. A commercial X-ray tube is combined with a device of slits limiting incident X-rays, a focusing spectrometer with a Rowland circle of 4-in. radius, and a microscope of low magnification for observing the analyzing point on the samples. The wavelength range of the spectrometer with LiF and ADP analyzing crystals is from 1.20 to 9.94 Å, and, therefore, higher elements than aluminum in atomic number can be analyzed. The authors exerted their efforts to obtain the higher X-ray intensities in order to analyze smaller areas. The X-ray intensities obtained are satisfactory, except for light elements. For example, the detected X-ray intensity of pure nickel is 1650 cps with the use of a 0.1-mm diameter specimen, and that of pure sulfur is 52 cps with the use of a 0.1-ramdiameter specimen; however, with a 1-mm-diameter specimen, the intensity of pure nickel is over 5000 cps and that of pure sulfur is 1650 cps. These correspond to the intensities from 20-mm-diameter specimens of those elements when a flat-crystal spectrometer is used. The calibration curve for quantitative analysis generally varies with the sample area under analysis, but the same curves are obtained if the sample area is larger than 1 mm in diameter. Then, powder samples are analyzed quantitatively by using a plastic sample holder of 1-mm diameter and 0.3-mm depth. This instrument has good ability for microanalyzing trace elements by, for example, the ion-exchenge membrane method. The sensitivity represented is nearly 5000 cps/μg for Ni Kα from NiSO4 that is soaked and dried in thin rice paper. Some applications of the micro fluorescent X-ray analyzer to precipitates in steels and corrosion products are reported.
Use of Primary Filters in X-Ray Spectrography: A New Method for Trace Analysis*
- S. Caticha-Ellis, Ariel Ramos, Luis Saravia
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 105-113
-
- Article
- Export citation
-
A method to improve the detectability of trace elements by X-ray fluorescent spectrography is described. The method consists of using appropriate filters in the primary, or exciting, beam. The effects of using filters in the primary beam on the peak-to-background ratio R of a fluorescent line have been analyzed on theoretical grounds. In fact,
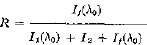
where I1(λ0) is the intensity of the analytic fluorescent line, I1(λ0) is the background intensity due to coherent and Compton scattering of the primary radiation by the specimen, and I2 is the background intensity due to scattering of the fluorescent radiation by the analyzing crystal. Analytical expressions were derived for I1(λ0), I2, and I1(λ0), from which it has been concluded:
1. The ratio I1(λ0)/I1(λ0) decreases when the filter used has its absorption edges at wavelengths longer than λ0.
2. The ratio I2/I1(λ0) can be separated into two parts which vary in opposite ways. The influence of these two parts on the value of R is discussed in the text.
It is then shown that the method should work well at short wavelengths and less well at longer wavelengths. The method was tested in the difficult case where overlapping of the analytical line with a characteristic line of the tube occurred, i.e., in the determination of traces of selenium by using tungsten radiation. The analytic line Se Kα. has a wavelength of 1.106 Å, while W Lγ1occurs at 1.098 Å. There is a marked effect of the filter thickness on the detectability; an optimum thickness appears to exist for each case. In the analysis of selenium, the best filter thickness (which can be selected by mere inspection of the diagrams reproduced in the text) increased the detectability of selenium traces by an order of magnitude. Finally, from statistical considerations, the quantity tσ2 is proposed as an index of the effectiveness of the filter: the smaller tσ2 is, the better the filter is. Here σ is the standard deviation of the intensity of the analytic line and t is the total counting time spent on the measure of the analytic line arid background. In order to study the dependence of the index tσ2 on the filter thickness, measurements were made on samples of sugar containing known concentrations of strontium. Then tσ2 was plotted against the thickness of the filter for each concentration; these curves do show a minimum. Thus, an optimum filter thickness exists in each case.
Precision and Accuracy of Silicate Analyses by X-Ray Fluorescence
- A. K. Baird, E. E. Welday
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 114-128
-
- Article
- Export citation
-
The relative merits of different methods of sample preparation, of different instrumental operating procedures, and of different calibration techniques for major and minor elemental analyses of silicates must be judged by adequate estimates of precision and accuracy. Sources of imprecision in the X-ray method include electronic deviations, counting statistics, and sample collection and preparation. The variations attributable to these sources set limiting values to any X-ray method. Because most X-ray methods are comparative and thus dependent upon standards, the accuracy is more difficult to establish. Sources of inaccuracy in calibration methods include biases introduced by instrumental design or operation, matrix and particle size effects reflected as scatter or calibration line curvature, and an inadequate knowledge of the chemistry of the standard or standards used. With an adequate estimate of the precision, a practical measure of the accuracy of the X-ray fluorescence method can be obtained by the predictions of the compositions of standard silicates—standards whose compositions are assumed to be established by repeated wet-chemical analyses. This practical approach to an assessment of errors suggests to us that the comparative X-ray methods are highly precise. Within the limits of precision and with appropriate standards, accurate predictions of the compositions of silicate rocks can be made without applying empirically or theoretically derived correction factors. In this paper tests of precision and accuracy will be described which have been used to assess variabilities in quantitative analysis from the sample collection, through the sample preparation, to the calibration with standards.
Applications of Computerized Statistical Techniques in Quantitative X-Ray Analysis
- Betty J. Mitchell
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 129-149
-
- Article
- Export citation
-
The place of statistical techniques and the digital computer in analytical chemistry is clearly exemplified in the X-ray spectrograph laboratory. As the statistician is obsolete who uses only a desk calculator for his calculations in this era of rapidly changing technology and the need for increased productivity, so the X-ray man is severely handicapped who ignores the place of statistical techniques and associated digital computation in his work. For his calculations, the statistical X-ray analyst may choose from among the following: a small specialized computer designed as part of his X-ray equipment, a medium-size computer located in the laboratory building with the capacity to handle all the problems generated by the various analytical specialists, a time-sharing computer station which he shares with other analytical or testing sections of his main laboratory, or batch processing of his data by his company's computer center. Advanced types of desk calculators are now available which will permit rapid completion of some types of complex calculations, especially if they are set up in a systematic fashion. Careful study of an individual laboratory's problems will provide economic justification for the most appropriate type of computer calculation; the choice is dependent on many factors. This paper describes the advantages and disadvantages of the various computer systems for the X-ray spectrographer and the numerous statistical techniques which he may employ as an aid in hi? work. Statistics provide him with the tools to design his experiments, interpret his data, control the process which supplies his samples, improve sampling procedures, calibrate his X-ray equipment, perform theoretical studies, and calculate, correct, average, and evaluate his analyses. Digital computation provides him with an extraordinarily rapid means of making these calculations; in some cases, they are virtually impossible without computerization. Examples of analyses are described which are made practicable only by this combined statistical computerized approach. The accuracy and precision of computerized analyses are higher than for results obtained by the usual X-ray methods; analyst error is reduced to a minimum. The economics of present-day industrial life, especially applied to the analytical laboratory as a service organization, demand the most efficient possible operation of all analytical equipment. The X-ray laboratory presents an outstanding example of the need for statistical analysis and its associated digital-computer computation.
X-Ray Fluorescence Analysis of a Manganese Ore
- W. D. Egan, F. A. Achey
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 150-157
-
- Article
- Export citation
-
To replace time-consuming chemical analysis, a procedure was developed for applying X-ray fluorescence to the analysis of a manganese ore. This X-ray fluorescence method is rapid; the total time for both sample preparation and analysis is ½ hr. The method is also simple enough for routine laboratory use. The components determined, the concentration ranges, and the agreement between chemical and X-ray analysis in terms of standard deviation are:
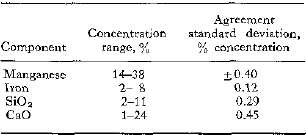
The agreements between chemical and X-ray analysis noted for manganese and iron are obtained by correcting for CaO concentration when determining the manganese and for manganese and SiO2 concentrations when determining the iron. The corrections employ empirical equations developed by a multiple regression technique.
Sample preparation is reduced to a minimum because it consists of only two steps, pulverization of the manganese ore followed by briquet ring. Four minutes of grinding with Boraxo as the grinding aid gives sufficient uniformity to minimize the effect of particle size variation. The ground mix of manganese ore and Boraxo is pressed into 1.25-in. briquettes for analysis.
Total Nondestructive Analysis of Caas Syenite
- A. Volborth, B. P. Fabbi, H. A. Vincent
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 158-163
-
- Article
- Export citation
-
Determination of 35 trace and 8 major elements in a granitic rock by nondestructive X-ray emission and fast-neutron activation methods has been accomplished. Trace elements arsenic, barium, cadmium, cerium, cobalt, chromium, cesium, copper, gallium, gadolinium, germanium, hafnium, mercury, indium, lanthanum, molybdenum, manganese, niobium, neodymium, nickel, rubidium, antimony, scandium, samarium, tin, strontium, tantalum, thorium, titanium, vanadium, yttrium, ytterbium, zinc, and zirconium and major elements aluminum, calcium, iron, potassium, and silicon have been estimated in the vacuum X-ray spectrograph. Phosphorus has been excited by a chromium-target tube. Major elements magnesium, sodium, and oxygen have been estimated by the Henke aluminumtarget X-ray spectrograph. Major elements oxygen and silicon have also been determined by fast-neutron activation. Detection limits for traces are in cur system in the range of 1 to 100 ppm with standard deviations of about 1 to 10 ppm. Precision in the determination of major constituents is better than 1% in relative standard deviation. The data are reported in parts per million for the trace elements and for the major elements as well. It is suggested that this type of repotting of geochemical data be preferred to the conventional reporting of major constituents as oxides.
X-Ray Fluorescence of Suspended Particles in a Liquid Hydrocarbon
- E. L. Gunn
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 164-176
-
- Article
- Export citation
-
In the application of X-ray fluorescence to the inspection of petroleum or petrochemical products, the measured element may not be ideally dispersed in the organic substrate but may exist as suspended particles. A question naturally arising is how small the particles must be to approximate the fluorescent intensity level of the true solution. The fluorescent emissions of suspensions of particles in a viscous hydrocarbon were measured and the intensities compared with true solutions of zinc, iron, and silicon, respectively. Particle segregates were classified by raicrosieve and photomiorographic inspection. Iron or zinc particles of 8-μ size yield 65% of the intensity of the solution state. Increase of particle size to 70 μ reduces intensity by several fold. Silicon, dispersed as porous silicon dioxide particles of 28-μ size, yields 80% of the intensity of the solution form. Reliable measurement of a suspension can be made provided it is stable and a standard closely simulating it is available for reference or if proper intensity corrections are applied for a known particle size in suspension. Comparisonsof experiment with theory for the particle size-intensity effect agree only moderately well. This is attributed to uncertainties in the parameters relating the two. Theory indicates that zinc or iron in suspended form should be less than 1 μ in size to approximate the intensity of the elements in solution. Additional research aspects of particles in suspension are suggested.
The Effect of Surface Roughness in Polymers on X-Ray Fluorescence Intensity Measurements
- J. Gianelos, C. E. Wilkes
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 177-184
-
- Article
- Export citation
-
We sought to determine how seriously surface roughness affects X-ray intensity measurements in polymers. Fourteen elements ranging from lead to silicon were added singly to fourteen batches of trans-1,4-polyisoprene. Smooth pressings of each batch were made, and intensity readings were taken (I0). Reproducibly rough surfaces were made from these by molding a square wire-mesh pattern into them, with the use of Tyler standard sieve screens. The amount of roughness was controlled by using screens of very fine to very coarse mesh. We studied the change in the X-ray intensity of the rough surfaces versus the smooth [(I/I0) × 100] with respect to: (1) the degree of roughness, (2) concentration of the added element, (3) emitted wavelength of the added element, (4) X-ray tube target material, and (5) correction for matrix effects on the intensity. We found that, at wavelengths emitted below 1 Å, intensity differences are small, regardless of which factors were varied. At wavelengths emitted above 1 Å, however, we found large differences. The intensity changes are highly dependent on roughness. Also, they become greater at the longer emitted wavelengths and with increasing concentration of added elements. Beginning with Ti Kα, losses are much higher with the use of chromium primary radiation than with tungsten. A technique of milling polyethylene into polymers with rough surfaces to provide a smooth surface is discussed.
Production Control of Gold and Rhodium Plating Thickness on Very Small Samples by X-Ray Spectroscopy
- George H. Glade
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 185-190
-
- Article
- Export citation
-
Manufacture of reed switches, critical components in present-day dataprocessing support devices, requires a means of accurate, rapid analysis of elements used in plating tlie levers of the switch. Because of gready reduced feedback time, X-ray spectroscopy has replaced metallographic sectioning and optical measurement as a plating-thickness control method. While 6 hr were required to obtain thickness data for a given sample size by sectioning, X-ray spectroscopy requires only 2 hr, which permits better control of the plating operating. X-ray spectroscopy is now used routinely to control both gold and rhodium plating thicknesses in the 20- to 100μin. (1 × 10−6) thickness range. The large number of samples prevents long count duration, while the small sample size (0.110 by 0.033 in.) reduces the precision of the analysis. However, the precision of the X-ray and optical methods is approximately the same, 8% variance. X-ray accuracy is comparable to that of sectioning since the standards are obtained by sectioning. Simplicity of operation is required since relatively untrained operators are used. An aperture system is used to reduce background. The rhodium thickness measurement is obtained from gross rhodium intensity. Attenuation of gross nickel intensity from the base material was found to be a better measure of gold thickness intensity. Calibration for hoth gold and rhodium is performed by using the same wide detector conditions. The choice of analysis is made by changing only the 2θ angle, thus avoiding the time required for recalibration when changing analysis.
A Study of X-Ray Fluorescence Method with Vacuum and Air-Path Spectrographs for the Determination of Film Thicknesses of II-VI Compounds
- Frank L. Chan
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 191-203
-
- Article
- Export citation
-
Films of II-VI compounds have been prepared by vacuum technique for the determination of their thicknesses. Some difficulties have been experienced in preparing good quality films- The positron of the substrate in the vacuum chamber, the rate of deposition, and the temperature of sublimation are some of the factors influencing the quality of these films. For instance, films of cadmium sulfide could be prepared in the usual yellow or orange color or in colors ranging from brown to black, depending on the conditions enumerated. For energy conversion, a film of proper thickness is one of the requirements for aerospace application. Among the various methods used for the determination of the thickness of these films, X-ray fluorescence can be performed rapidly and nondestructively. After determination of thickness by the X-ray fluorescence method, the samples can be used for other determinations and for energy conversion without their efficiency's having been affected. Other physical and chemical methods have been worked out. Comparison of these methods with the X-ray fluorescence method is made. Procedures and results are presented.
Rare-Earth Analyses by X-Ray-Excited Optical Fluorescence
- W. E. Burke, D. L. Wood
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 204-213
-
- Article
- Export citation
-
X-ray excitation causes rare-earth impurities in yttrium oxide and gadolinium oxide to emit intense and highly characteristic optical line fluorescence, which enables their analyses at low concentrations. The limits of detection for praseodymium, ncodymium, samarium, europium, gadolinium, terbium, dysprosium. holmium, erbium, thulium, and ytterbium in these two oxides range from 1 to 100 parts per billion (ppb). In other rare-earth oxides which have been investigated. the fluorescent intensities arc greatly reduced. Successful analyses can be made only by dilution in high-purity Y2O3. This dilution raises the detection limits for rare earths in these other oxides to the part per million range. X-rays from the chromium target of a cual-target X-ray tube are about two times more efficient in exciting rareeaith optical fluorescence than are the tungsten-target X-rays, even though the total energy output of the chromium target is only about one-third that of the tungsten target. With either target material, the rare-earth intensities vary linearly with the X-ray tube current, but a plot of intensity versus the square of the accelerating potential is not linear; it drops off at higher voltages.
Nondispersive X-Ray Fluorescent Spectrometer
- W. Barclay Jones, Robert A. Carpenter
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 214-229
-
- Article
- Export citation
-
Recent advances in semiconductor particle detector resolutions along with new electronic circuitry associated with these detectors make possible their application in nondispersive elemental analysis. The use of radioactive sources for exciting the characteristic X-rays provides highly stable systems which can be used to accumulate data for prolonged periods. Due to the inherent stability of the detector and the excitation source, the only limitation in sensitivity is the ability to accumulate statistics above the background of scattered counts. Since this method of analysis is nondispersive, it has the capacity to determine many elements simultaneously. Solutions composed of mixtures of three or four elements were studied. The elements selected were bromine, rubidium, and strontium. These elements exhibit wide variations in mass absorption coefficients for the various characteristic X-rays emitted. The concentrations of the elements in solution varied from 10 ppm to 5% by weight. The relative intensities of the characteristic X-ray lines were compared with the concentration of the solutions to establish sensitivity curves and to study linearity of response as well. The interelemental interference was studied and the effect was evaluated for the particular elements under study. Means were developed for predicting and correcting for matrix effects.
The Influence of Sample Self-Absorption on Wavelength Shifts and Shape Changes in the Soft X-Ray Region: The Rare-Earth M Series
- David W. Fischer, William L. Baun
-
- Published online by Cambridge University Press:
- 06 March 2019, pp. 230-240
-
- Article
- Export citation
-
The Mα and Mβ emission spectra and the Mjv and My absorption spectra have been studied for the entire series of rare-earth elements. It is conclusively shown that the complicated multiplet structure observed in the emission spectra is not real emission structure but is, instead, produced by sample self-absorption. This is demonstrated by observing the emission spectra over wide variations in take-off angle and bombarding electron energies and finally by comparing the detailed structure of both the emission and absorption spectra. The MIV and MV absorption structure completely overlaps the Mα and Mβ emission lines, which are each found to have but one intensity maximum when obtained under conditions of minimum. self-absorption. Some of these spectra have never been shown previously, while others have been studied in detail by several investigators. Points of agreement and disagreement with previous work are mentioned, and the wavelengths of the emission lines and absorption edges are listed for all of the rare-earth elements. It is concluded that the 4f → 3d electron transitions are reversible in these elements.