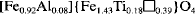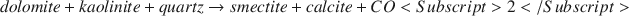Volume 46 - August 1998
Research Article
Iron Influence in the Aluminosilicate Zeolites Synthesis
- E. I. Basaldella, R. M. Torres Sánchez, J. C. Tara
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 481-486
-
- Article
- Export citation
-
The level of Fe impurities in 2 well-crystallized kaolinites was modified (by addition or chemical removal treatment) to analyze the Fe influence in the aluminosilicate zeolite synthesis.
The original and modified clays were heat-treated in order to change their reactivity for zeolite A synthesis, and their thermal transformations were studied by X-ray diffraction (XRD), determination of point of zero charge (PZC) and infrared (IR) techniques. It was established that many structural changes took place, regardless of the Fe clay content. Furthermore, the presence of Fe species in alkaline solution or in the solid phase did not seem to greatly influence the zeolite crystallization, because only small differences in the conversion values among samples with different Fe contents were registered. The crystallization process seemed to be related mainly to AI coordination changes produced by the thermal and Fe removal treatments used.
Modification of Sepiolite by Treatment with Fluorides: Structural and Textural Changes
- María Jesús Belzunce, Sagrario Mendioroz, Jerzy Haber
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 603-614
-
- Article
- Export citation
-
In the search for new applications of natural silicates, various F− treatments have been applied to sepiolite to increase its acidic properties and for use as a catalyst in reactions occurring via carbonium ions. Two types of treatments including hydrofluoric acid (HF) at different concentrations and 2 N NH4F have been utilized and the physicochemical characteristics of the resulting materials studied using standard techniques. The X-ray diffractogram (XRD) patterns indicate a decrease in crystallinity of the original material as well as the appearance of amorphous silica. SEM micrographs showed a shortening and aggregation of the sepiolitic fibers. X-ray photoelectron spectroscopy (XPS), Fourier transform infrared (FTIR), thermogravimetric analysis/differential thermal analysis (TGA/DTA), N2 adsorption-desorption isotherms and Hg intrusion were used to study the changes occurring in the structure, surface area and pore distribution of samples and acidity was evaluated by IR and thermoprogrammed desorption (TPD) of adsorbed ammonia and pyridine. It was found that acidity increased in most of the samples after anionic and cationic interchange between the activating agents and the surface sites, or extralattice cations. Additionally, structural changes induced by treatments modified the Brönsted and Lewis acidity. Mild treatments with ammonium fluoride are more effective than HF treatments in acidity generation.
Kaolin Mineralogy of Clays in Paleosol Profiles on the Late-Miocene Sediments in Penghu Islands (Pescadores), Taiwan
- Wei-Min Wang, Hsueh-Wen Yeh, Pei-Yuan Chen, Ming-Kuang Wang
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 1-9
-
- Article
- Export citation
-
Clay mineral compositions from 2 paleosol profiles (Chu-Wan, CW, and Shiao-Men Yu, SMY, profiles) on the late-Miocene sediments in Penghu Islands (Pescadores), Taiwan, are characterized by random X-ray powder diffraction (XRD) and transmission electron microscopy (TEM). By the clay assemblage of the paleosol profile, we want to explore the probable formation mode of the Penghu paleosols.
The paleosol profiles in study are overlain by a layer of basalt flow. However, the clay mineralogy of the 2 paleosols was not altered metasomatically after burial. Results show that 3 distinctive zones of different dominating kaolin-group minerals are apparent in the profiles. In descending order, they are: 1) spheroidal, hollow 7Å-halloysite, 2) platy, irregular-shaped and disordered kaolinite, and 3) platy, irregular-shaped, disordered kaolinite. The relative crystallinity of kaolin minerals of the 3 layers is: layer 2 > layer 3 > layer 1. On the basis of the XRD, TEM analyses and the crystallinity calculations, the distribution of kaolin in Penghu paleosol profiles appears to be unique. Penghu paleosol profiles show systematic change in kaolin crystallinity and polymorphs with depth. Because the clay type is heterogeneous within the profile, this represents that Penghu paleosol profiles were polypedogenic.
The contact between the upper basalt and the paleosol is the erosion surface, so we do not know exactly what the thickness of the original paleosol was. The first layer (about 20 cm) of the profiles appears to be constituents of the original paleosol. It contains high contents of pedogenic (in situ weathering) hematites and 7Å-halloysites, which implies that the local climate of the Penghu Islands at late Miocene was warm and humid. Intense leaching and dry/wet cycle should be the reason for high contents of halloysite (>60%) in the Penghu paleosols. Laterization was the probable pedogenic process for the formation of the paleosols.
State and Location of Water Adsorbed on Clay Minerals: Consequences of the Hydration and Swelling-Shrinkage Phenomena
- R. Prost, T. Koutit, A. Benchara, E. Huard
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 117-131
-
- Article
- Export citation
-
The application of the Frenkel-Halsey-Hill (FHH) formalism to the water desorption isotherms obtained for the whole range of the activity of water with the pressure membrane device (0.98 < aw < 1) and with the desiccator (0 < aw < 0.98) gives information concerning the nature and the relative importance of the 2 mechanisms involved in the dehydration—hydration processes: adsorption and capillary condensation. The state and location of water are described in each domain. An equation that gives the thickness t of the film of water adsorbed on the walls of pores versus the activity of water is developed. This t-curve is used to get, from the desorption isotherm, the pore size distribution curve of the studied hydrated materials. Then concepts of surface and fabric of clay pastes are discussed as a function of hydration and a mechanism is proposed to explain swelling and shrinkage of finely divided materials. Three kinds of surfaces, related to the aggregate fabric, are defined as a function of their capacity to adsorb water. Each kind of surface is determined by a specific technique: the total surface area (St) by ethylene glycol adsorption, the external surface area of particles (Ss) by nitrogen adsorption and the external surface area of aggregates (Se) by hydraulic conductivity measurements. As a consequence it is only with completely dispersed clays that swelling is a function of St. With unwell-dispersed clays, water adsorption, which induces swelling, successively occurs on St, Ss and Se surfaces.
Oxygen Isotope Compositions of Mixed-Layer Serpentine-Chlorite and Illite-Smectite in the Tuscaloosa Formation (U.S. Gulf Coast): Implications for Pore Fluids and Mineralogic Reactions
- P. C. Ryan, M. E. Conrad, K. Brown, C. P. Chamberlain, R. C. Reynolds, Jr.
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 357-368
-
- Article
- Export citation
-
Oxygen isotopic compositions were determined for coexisting mixed-layer serpentine-chlorite (Sp-Ch) and illite-smectite (I-S) from 5 Tuscaloosa Formation sandstone cores sampled between 1937 and 5470 m burial depth. High gradient magnetic separation (HGMS) was used to concentrate Sp-Ch and I-S from the <0.5 μm fraction of each core sample into fractions with a range in the Sp-Ch: I-S ratio, and end-member δ18O compositions were determined by extrapolation. The Sp-Ch δ18O values range from + 10.4 to 13.7% and increase with burial between 3509 and 5470 m. The only exception is Sp-Ch from 1937 m, which has an anomalously high δ18O value of +12.6‰ The I-S δ18O values range from +16.1 to 17.3% and do not change significantly between 3509 and 5470 m burial depth.
Pore water δ18O compositions calculated from Sp-Ch and I-S values and measured borehole temperatures range from −2.6 to +10.3‰ The isotopically light values indicate that Sp-Ch formed at shallow burial depths in the presence of brackish to marine water and/or meteoric water. The depth-related increase in δ18O of Sp-Ch is attributed to oxygen exchange between mineral and pore water during diagenetic mineral reactions. Increasing δ18O values, in conjunction with XRD and SEM data, indicate that transformation of serpentine layers to chlorite layers and Ibb polytype layers to Iaa polytype layers occurred on a layer-by-layer basis when individual layers dissolved and recrystallized within the confines of coherent crystals. Possible explanations for the variation in I-S δ18O values include depth-related differences in pore water δ18O values present at the time of I-S crystallization, contamination by detrital 2M, mica and 1M polytype rotations that facilitated oxygen exchange.
Structural and Textural Modifications of Palygorskite and Sepiolite Under Acid Treatment
- M. Myriam, M. Suárez, J. M. Martín-Pozas
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 225-231
-
- Article
- Export citation
-
Sepiolite from Mara (Zaragoza, Spain) and palygorskite from Attapulgus (Georgia, USA) were activated by treatment at different concentrations with solutions of boiling HCl under reflux conditions. The natural and treated solids were characterized by mineralogical, chemical and textural analyses.
Acid attack resulted in a progressive dissolution of the octahedral layer of these silicates. Silica contents increased and octahedral cations (Al, Mg and Fe) decreased with the intensity of the acid attack. In both cases, fibrous free silica was obtained.
The sepiolite was destroyed more rapidly than palygorskite under the conditions used because of its magnesic composition and the larger size of its structural microchannels. The maximum increase in specific surface area was obtained for sepiolite at 3 N and for palygorskite at 9 N. Cleaning and disaggregation of the particles and the increase in the number of micropores were responsible for this increase in specific surface area.
Relation of Water and Neutral Organic Compounds in the Interlayers of Mixed Ca/Trimethylphenylammonium-Smectites
- Guangyao Sheng, Stephen A. Boyd
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 10-17
-
- Article
- Export citation
-
Organoclays were prepared by exchanging Ca2+ in a Ca2+-saturated smectite partially or fully with trimethylphenylammonium (TMPA) cations. The mechanistic function of these organoclays as adsorbents for neutral organic compounds in aqueous solution was examined. TMPA cations were found to take a random distribution on the surfaces of mixed Ca/TMPA-smectites. The presence of TMPA, and its random distribution, resulted in water associated with the clay surfaces being held more weakly. Apparently, the interspersing of TMPA and Ca2+ ions prohibits the formation of a stable network of water molecules around Ca2+. Water molecules associated with the siloxane surface in mixed Ca/TMPA-clays are removed during the adsorption of neutral organic compounds from bulk water, leaving only ∼11 strongly held water molecules around each Ca2+, as opposed to ∼58 water molecules in homoionic Ca2+-smectite. These results demonstrate that the amount of water associated with the clay surfaces and interlayers depends on the nature of the exchange cation(s), and not on the amount of available siloxane surface area by itself. We conclude that in TMPA-smectites the TMPA cations function as nonhydrated pillars, and sorption of organic solutes occurs predominantly on the adjacent siloxane surfaces, which are hydrophobic in nature. The water molecules around Ca2+ in mixed Ca/TMPA-smectites obscures some of the siloxane surfaces. This diminishes sorption capacity, in an amount roughly equivalent to the fraction of the CEC occupied by Ca2+, because organic solutes cannot displace the waters of hydration of Ca2+.
Swelling and Texture of Iron-Bearing Smectites Reduced by Bacteria
- Will P. Gates, Anne-Marie Jaunet, Daniel Tessier, Michael A. Cole, Henry T. Wilkinson, Joseph W. Stucki
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 487-497
-
- Article
- Export citation
-
Microbial reduction of clay mineral structural Fe(III) decreases the swelling of nontronite gels, most importantly at intermediate oxidation states (40 to 80 cmol Fe(II) kg−1 clay). The purpose of this study was to establish whether microbial reduction of structural Fe(III) decreased the swelling of other Fe-bearing smectites and to discern the influence that organic compounds of microbial origin (bacterial cells, cell fragments and/or exudates) may have on clay swelling and texture. Structural Fe(III) was reduced by incubating smectite suspensions with either a combination of Pseudomonas bacteria or a mixture of anaerobic bacteria. The influence of organics on clay swelling was estimated on smectites suspended in either organic or inorganic media in the absence of bacteria. The gravimetric water content of the reduced clay gels equilibrated at various applied pressures was recorded as a function of Fe oxidation state. Transmission electron microscopy (TEM) was employed to determine the influence of bacteria and type of media on the texture of reduced smectite gels. Reduction of structural Fe(III) by bacteria decreased the swelling pressure of all Fe-bearing smectites. Increased clay swelling, due to the presence of organics (organic medium, exudates or cell fragments), was correlated to the total Fe content, the extent of structural Fe reduction, as well as the initial swelling characteristics of the Fe-bearing smectites. High structural Fe(II) contents (>50 cmol Fe(II) kg−1) resulted in increased attractive forces between clay platelets that decreased clay swelling, even in organic medium suspensions. Microbial reduction resulted in increased face-face association of individual clay layers, forming larger and more distinct crystallite subunits than in nonreduced clay gels. But, perhaps more importantly, microbial reduction of structural Fe(III) resulted in an increased association between crystallite subunits and, thus, an overall larger particle size and pore size distribution, due to the interaction of bacteria ceils, cell fragments and organic exudates.
Molecular Simulations of Montmorillonite Intercalated with Aluminum Complex Cations. Part I: Intercalation with [Al13O4(OH)24+x(H2O)12−x](7−x)+
- P. Čapková, R. A. J. Driessen, M. Numan, H. Schenk, Z. Weiss, Z. Klika
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 232-239
-
- Article
- Export citation
-
The structure of montmorillonite intercalated with [Al13O4(OH)24+x(H2O)12−x](7−x)+ cations (
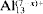 for short), where x = 0, 2 and 4, has been studied using the Cerius2 modeling environment. The Crystal Packer module used in the present study takes into account only the nonbonded interactions between the silicate layer and the Keggin cations. Minimization of the total sublimation energy led to the following conclusions: the structure of the interlayer (that is, the orientation of Keggin cations and the basal spacing) depends on the charge of cations (that is, on the degree of hydrolysis, x). The values of basal spacings in the range 19.38–20.27 Å have been obtained, depending on the charge and arrangement of cations in the interlayer. The dominating contribution to the total sublimation energy comes from the electrostatic interactions. Translations of
for short), where x = 0, 2 and 4, has been studied using the Cerius2 modeling environment. The Crystal Packer module used in the present study takes into account only the nonbonded interactions between the silicate layer and the Keggin cations. Minimization of the total sublimation energy led to the following conclusions: the structure of the interlayer (that is, the orientation of Keggin cations and the basal spacing) depends on the charge of cations (that is, on the degree of hydrolysis, x). The values of basal spacings in the range 19.38–20.27 Å have been obtained, depending on the charge and arrangement of cations in the interlayer. The dominating contribution to the total sublimation energy comes from the electrostatic interactions. Translations of 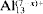 cations along the 2:1 layers give only small fluctuations of the total sublimation energy and basal spacings. No preference for the position of
cations along the 2:1 layers give only small fluctuations of the total sublimation energy and basal spacings. No preference for the position of 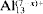 cations in the interlayer of montmorillonite was found during translation along the 2:1 layers. This result confirmed the inhomogeneous distribution of cations in the interlayer and turbostratic stacking of layers.
cations in the interlayer of montmorillonite was found during translation along the 2:1 layers. This result confirmed the inhomogeneous distribution of cations in the interlayer and turbostratic stacking of layers.
Iron Oxides in a Soil Developed from Basalt
- A. T. Goulart, J. D. Fabris, M. F. de Jesus Filho, J. M. D. Coey, G. M. da Costa, E. de Grave
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 369-378
-
- Article
- Export citation
-
A dusky red Oxisol forming on a tholeiitic basalt is found to contain varying proportion of aluminous hematite (Hm) and titanoaluminous maghemite (Mh) in the different size fractions. Maghemite is the main iron oxide in the sand and silt fractions whereas Hm is dominant in the clay fraction, together with gibbsite (Gb), kaolinite (Ka), rutile (Rt) (and probably anatase, An) and Mh. Maghemite is also the major oxide mineral in the magnetic separates of soil fractions (sand, about 65% of the relative Mössbauer spectral area; silt, 60%). Hematite (sand, 30%; silt, 15%) and ilmenite (Im) (sand, 5%; silt, 16%) are also significantly present in the magnetic extract. Accessory minerals are Rt and An. No magnetite (Mt) was detected in any soil fraction. Sand- and silt-size Mh have similar nature (a 0= 0.8319 ± 0.0005 nm; about 8 mol% of Al substitution; saturation magnetization of 49 J T−1 kg−1), and certainly a common origin. Lattice parameters of clay-Mh are more difficult to deduce, as magnetic separation was ineffective in removing nonmagnetic phases. Al content in Hm varies from 14 mol% (clay and silt) to 20 mol% (sand). The proposed cation distribution on the spinel sites of the sand-size Mh is:
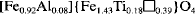
(◻ = vacancy, [ ] = tetrahedral sites and { } = octahedral sites), with a corresponding molar mass of 208.8 g mol−1. The predicted magnetization based on this formula is σ ≅ 68 J T−1 kg−1, assuming collinear spin arrangement. The large discrepancy with the experimentally determined magnetization is discussed.
Dehydration, Diffusion and Entrapment of Zinc in Bentonite
- Y. B. Ma, N. C. Uren
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 132-138
-
- Article
- Export citation
-
Interactions with bentonite are important in the chemical speciation and fate of heavy metals in soils and other ecosystems. The interactions of Zn with bentonite were studied using X-ray diffraction (XRD), dehydration, kinetic and sequential extraction procedures. The species and activity of Zn retained by bentonite were affected markedly by pH. The Zn(OH)+ was retained by bentonite prepared at pH ≥ 6.9. The extent of dehydration of Zn(OH)+-bentonite was higher than that for Zn-bentonite. At a relative humidity of 55.5%, the basal spacing of the Zn(OH)+-bentonite was from 1.21 to 1.26 nm with 1 water sheet and that of the Zn-bentonite was 1.51 nm with 2 water sheets. The greater affinity of Zn(OH)+ for bentonite than Zn was associated with a lower degree of hydration. When an aqueous suspension of Ca-bentonite was incubated with soluble Zn, the concentration of Zn retained by the Ca-bentonite was linearly related to the square root of time. The rate of the interaction was controlled probably by the interlayer diffusion and subsequently by the diffusion into the ditrigonal cavities in bentonite. The Zn retained by bentonite was dehydrated in situ so as to increase the bonding of Zn with surfaces of bentonite. With hydrothermal treatment the retained Zn could diffuse easily into the cavities and transform increasingly to the residual forms that are associated with the entrapped form.
Chemical Variations in Chlorite at the Los Humeros Geothermal System, Mexico
- Raymundo G. Martínez-Serrano, Michel Dubois
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 615-628
-
- Article
- Export citation
-
The chemical composition of hydrothermal chlorite was determined by means of more than 200 electron microprobe analyses (EMPA) in almost all of the 70 chlorite-bearing samples taken from 5 boreholes in a study of the active geothermal system of Los Humeros, Mexico. Bulk rock composition of 6 different volcanic lithologies, as well as available in situ temperatures and chemical compositions of chlorite, were analyzed by principal component analysis (PCA) in order to test the dependence of chlorite composition on physicochemical parameters. The results show that chlorite minerals display a wide range of chemical compositions in this hydrothermal system that reflect the particular conditions of crystallization episodes: The Na + K + 2Ca values are low (from 0 to 0.6) and they show no correlation patterns with octahedral vacancies (¤) in chlorite, indicating that compositional variations are not due to the intergrowth of smectite and/or illite. The octahedral occupancy of most chlorite is relatively high (from 11.3 to 11.95), especially that from a high-temperature range, as is the case of metamorphic chlorite. The octahedral occupancy seems not to be related to other chemical variables of chlorite from the G3, G4 and G5 lithologic units, suggesting that the lack of complete occupancy is not dependent on “contamination” by other silicates (such as quartz). Cationic substitution in tetrahedral sites in chlorite is small and via a Tschermak exchange (MgVISiIV ↔ AlVIAlIV). To preserve a charge balance in the structure, an octahedral substitution of R2+ by Al3+ accompanies the Tschermak exchange. The chemical composition of hydrothermal chlorite is very similar to that of metamorphic chlorite but slightly different from equivalent phases found in diagenetic environments. In hydrothermal chlorite the SiVI, AlVI and ¤ decrease, whereas the AlIV and Fe2+ contents increase with the degree of alteration and depth, the same way as in chlorite formed in diagenetic high-temperature environments. The ferrous iron content, in general, increases with depth and temperature; however, whole-rock chemistry affects the iron distribution in chlorite of Los Humeros. Changes in the oxygen fugacity of fluids at depth also affect the iron distribution in chlorite, XFe = Fe/(Fe + Mg), which ranges from 0.30 to 0.38 in oxidizing conditions and from 0.39 to 0.60 in reducing conditions. Finally, the chemical composition of chlorite in Los Humeros appears to change with temperature, but the correlations of ¤ and AlIV with temperature are more variable than in another nearby active geothermal system located in Los Azufres, Mexico. This implies that geothermometers based on chlorite composition and empirically calibrated in some geothermal systems cannot be generalized and it is necessary to consider other physicochemical variables.
Gallium and Chromium Substitution for Aluminum in Synthesized Beidellite
- Kerstin B. Brandt, Ronald A. Kydd
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 139-144
-
- Article
- Export citation
-
The hydrothermal synthesis of Mg-beidellite in which limited amounts of Ga and Cr are clearly shown to be incorporated into nonexchangeable positions in the clay layer framework is reported for the first time. Elemental analyses indicate that, up to at least 15 mole percent, Ga can replace Al virtually completely; Cr incorporation is more difficult. It appears that the Ga that is incorporated into the beidellite is located primarily in the octahedral sheet. Pillaring of the synthesized beidellite by replacing the Mg2+ with large AlO4Al12(OH)24(H2O)127+ ions showed that approximately 5.5 meq Al3+ was required per g of beidellite to achieve complete pillaring. This amount is very similar to that required for montmorillonite, and suggests that the cation exchange capacity (CEC) of the beidellite is about 98 meq/g.
An Experimental Study of the Effects of Diagenetic Clay Minerals on Reservoir Sands
- Paul H. Nadeau
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 18-26
-
- Article
- Export citation
-
Despite substantial scientific research efforts, accurate determination of the petrophysical effects of clay minerals on reservoir sands remains problematic. Diagenetic clays such as smectite and illite are of particular interest because of the pronounced effects these clays can have on reservoir quality. Here, results are reported from an experimental study based on the hydrothermal growth of smectite in synthetic sands. The sands contained quartz, dolomite and kaolinite, and were reacted at 175–200°C, for 19–45 d. The hydrothermal reaction can be written as follows:
$$dolomite + kaolinite + quartz \to smectite + calcite + CO<Subscript>2</Subscript>$$X-ray diffraction (XRD), electron microprobe (EMP) and electron diffraction (ED) analysis show that the synthetic Mg-rich smectite formed is saponite, with a cation exchange capacity (CEC) of about 100 meq/100 g. After reaction, brine permeability reductions of up to 98% were observed from the growth of less than 5% smectite. Scanning electron microscopy (SEM) observations of critical-point-dried reacted samples show that the clay behaves as a pervasive microporous cement with a complex pore-bridging texture affecting most of the available pore space. Morphologically, the clay is similar to naturally occurring diagenetic smectite from Gulf Coast sandstone reservoirs. The delicate clay texture collapses during air-drying and forms pore-lining masses. This phenomenon is similar to that observed for air-dried reservoir samples which contain dispersed diagenetic clays. An air-dried sample, then resaturated with brine, showed a marked increase in permeability. This increase is associated with the irreversible collapse of the clay texture. The experimental results indicate that the growth of diagenetic clay can severely reduce formation permeability, even at very low clay contents. The results also demonstrate the utility of hydrothermal experimental petrophysics for investigating the effects of diagenesis on rock properties.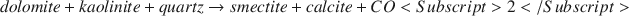
Evaluation of Gas Transport Properties of Backfill Materials for Waste Disposal: H2 Migration Experiments in Compacted Fo-Ca Clay
- C. Gallé, K. Tanai
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 498-508
-
- Article
- Export citation
-
Swelling clays may play a major role in the underground disposal of high-level nuclear waste (HLW) in deep geological formations. A multibarrier concept including a waste container, a steel overpack, an engineered barrier consisting of compacted clay (buffer) and the host rock is a potential technique for such HLW disposal. It is anticipated that H2 will be the main gas generated, mainly by anaerobic metal corrosion during disposal lifetime. After complete resaturation, the clay barrier will have a very low permeability and H2 may accumulate in the space between the overpack and the clay barrier. This could result in pressures exceeding the resistance of the clay and damage to the entire engineered barrier system (EBS).
The French Atomic Energy Commission (CEA) has performed an experimental program on H2 gas migration in a French clay referenced Fo-Ca. The tests were conducted with a specific odometer-type cell developed by the Power Reactor and Nuclear Fuel Corporation (PNC) in Japan. Permeability tests on compacted Fo-Ca clay samples of specific dry densities between 1.6 and 1.9 and for water saturation degrees between 70 and 100% provided significant H2 permeability data (ranging from 10−15 to 10−21 m2). Gas migration experiments were also performed to study the behavior of Fo-Ca clay under high gas pressure. Two kinds of gas transport threshold pressures were detected in unsaturated compacted clay. The first one, called “critical pressure”, is the pressure over which a gas outflow migrates into the clay. This pressure probably exceeds the capillary pressure of the largest pores within the clay. The second one, called “breakthrough pressure”, is detected with increasing gas injection pressure. A sudden rise of gas outflow is subsequently observed. The occurrence of the gas breakthrough is associated with the aperture and propagation of preferential gas transport pathways in the clay. This is apparently linked to the hydromechanical properties and stress state of the clay. When clay samples are not fully watersaturated, the critical pressure is always lower than the breakthrough pressure. However, when the clay is saturated, the 2 pressures appear to be very close. Finally, this study showed that breakthrough pressure—a key parameter for the long-term stability of the clay buffer—varies considerably with the gas injection increments and with the size of the sample. The results of these experiments are consistent with those reported previously by other investigators.
Comparative Study of the Mobility of Major and Trace Elements During Alteration of an Andesite and a Rhyolite to Bentonite, in the Islands of Milos and Kimolos, Aegean, Greece
- George E. Christidis
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 379-399
-
- Article
- Export citation
-
Progressive alteration by seawater of an andesite in the Aegean Island of Milos and an ignimbrite in the Aegean Island of Kimolos, Greece, formed bentonites with or without zeolites. Both profiles are dominated by migration of alkalis and uptake of Mg, Fe and H2O, while Al and Ti are immobile. The relative removal of alkalis controls the formation of either smectite or zeolites. The behavior of Ca and Si depends on the chemistry of the parent rock. In the rhyolitic profile, alteration is controlled by gain of Mg, Fe2+ and Ca and loss of Na, K and Si, while in the andesitic profile by gain of Mg and Fe2+ and loss of Na, K and Ca. In both profiles, significant uptake of SO4- was not observed. Moreover Zr, Nb, V and Ni are immobile and have been enriched residually, while Sr, Rb and Y are lost in both profiles. Thorium is immobile in the rhyolitic profile but is leached in the andesitic profile. Also, the rare earth elements (REE) display fractionation in both profiles; the degree of fractionation increases with the degree of alteration to bentonite. Fractionation of the REE in both profiles and mobility of Th in the andesitic profile are related to the existence of monazite (rhyolitic profile) and apatite (andesite profile). The REE and Th appear to partition into phosphates rather than smectite.
The mobility of Y coupled with the immobility of Nb increases the Nb: Y ratio with advancing alteration, rendering discrimination diagrams that use this ratio to determine the nature of the protoliths misleading. Mass balance calculations showed that in the smectite-rich zones the water: rock (WR) ratio might be as high as 13:1 in both profiles, while in the zeolite-bearing zones it is about 5.5:1. Such WR ratios explain the observed extensive mass transfer and suggest that the pore fluid chemistry might overprint the chemical characteristics of the parent rocks controlling smectite and bentonite chemistry.
Molecular Simulations of Montmorillonite Intercalated with Aluminum Complex Cations. Part II: Intercalation with Al(OH)3-Fragment Polymers
- P. Čapková, R. A. J. Driessen, M. Numan, H. Schenk, Z. Weiss, Z. Klika
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 240-244
-
- Article
- Export citation
-
The Crystal Packer module in the Cerius2 modeling environment has been used to study the structure of montmorillonite intercalated with Al(OH)3-fragment (gibbsite-like) polymers. Basal spacings in gibbsite-like polymers arranged in 2 layers in the interlayer of montmorillonite varied in the range 19.54–20.13 Å, depending on the type and arrangement of Al(OH)3 fragments. The inhomogeneous distribution of intercalating species in the interlayer and, consequently, the turbostratic stacking of layers has been found for gibbsite-like polymers as well as in the case of Keggin cations (Čapková et al. 1998). The dominating contribution to the total sublimation energy comes from electrostatic interactions for both intercalating species, gibbsite-like polymers and Keggin cations.
The Interlayer Structure of La-Vermiculite
- Phillip G. Slade, Peter G. Self, James P. Quirk
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 629-635
-
- Article
- Export citation
-
The structure of the interlayer cation-water system in La-vermiculite with a unit cell of a = 5.33(5), b = 9.18(6), c = 15.13(9) Å and β = 96.82(7)° has been determined in space group C2/m. Under ambient conditions, the interlayer La cations are distributed on a 3a × b superlattice which disappears on dehydration but returns on rehydration. The basal spacing does not change during the dehydration/rehydration process. The character of the superlattice spots indicate that the cation-water system, at ambient conditions, is ordered over relatively large domains. The La cations are surrounded by 8 neighboring water molecules in a distorted cubic arrangement. The spaces between the La-water clusters are occupied by triads of water molecules that are relatively mobile.
Low-Temperature Alteration in Tuffs from Yucca Mountain, Nevada
- Jillian F. Banfield, William W. Barker
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 27-37
-
- Article
- Export citation
-
The structure, chemistry and distribution of hydrothermal alteration and weathering products of feldspars and glass in 3 samples of Yucca Mountain tuffs (GSW G4 borehole at a depth of 1531 ft (464 m) and USW GU3 borehole at 1406 ft (426 m) from the Calico Hills Formation and USW G4 at a depth of 272 ft (82.4 m) from the Topobah Springs Member) were examined by high-resolution transmission electron microscopy (HRTEM) and analytical electron microscopy (AEM). Alteration products are of interest because they may influence the form and distribution of contaminants released from the proposed high-level nuclear waste repository. Samples from the Calico Hills Formation contain alkali-bearing aluminosilicate glass and its alteration products. Zeolites appear to have formed from compositionally similar glass by recrystallization, probably under hydrothermal conditions. Crystals are fibrous and frequently no more than a few tens of nanometers in diameter. Porous aggregates of few-nanometer-diameter, poorly crystalline silica spheres (probably opal C-T) develop adjacent to corroded glass surfaces and zeolite crystals. Finely crystalline Fe-rich smectites coat etched glass surfaces, zeolites and feldspar crystals and occur within opal-like silica aggregates. Microstructures in the clay-dominated coatings and details of smectite-glass interfaces suggest that clays grow in orientations controlled by heterogeneously retreating surfaces and from constituents released at associated glass dissolution sites. The alteration assemblage also includes finely crystalline hematite, goethite, Mn-oxide films and illite formed by alteration of muscovite. The zeolitized sample contains abundant opal-like silica whereas glass in the unzeolitized sample is weathered to smectite-like clays. These differences may be attributed to hydrological and consequent geochemical factors resulting from the higher porosity of zeolitized samples. Exsolved alkali feldspar, which occurs as micron-sized crystals in the Calico Hills Formation and as phenocrysts and in the groundmass of the devitrified Topobah Springs Member, are almost unaltered. Feldspar alteration is confined to cracks and grain boundaries, where minor, poorly crystalline, Fe-bearing aluminosilicate alteration products are developed. In these tuffs, most of the porosity, permeability, high surface area and capacity to affect solution chemistry are associated with products of glass alteration.
Influence of Pore Fluid Composition on Volume of Sediments in Kaolinite Suspensions
- J. Chen, A. Anandarajah
-
- Published online by Cambridge University Press:
- 28 February 2024, pp. 145-152
-
- Article
- Export citation
-
Reported in this paper is a study of the influence of pore fluid composition on sediment volume of kaolinite suspensions. Laboratory tests have been conducted with kaolinite in water with NaCl, CaCl2 and A1C13 of different concentrations and in 10 types of organic liquids of varying values of static dielectric constant. The types of tests performed include regular suspension tests and leaching and cyclic leaching tests on kaolinite sediments. In the leaching tests, sediments formed during the regular suspension tests in water of low salt concentration were subsequently leached with water of high salt concentration. In the cyclic leaching tests, the salt concentration was increased and then decreased. The purpose of the leaching and cyclic leaching tests was to study the change in existing equilibrium fabric caused by subsequent changes in the concentration of salt in pore fluid. Results of the suspension tests indicate that sediment volume of a water suspension decreases with increase in ion concentration and increase in valence of cation. Leaching and cyclic leaching tests indicate that substantial change in salt concentration is required to change the existing fabric. The effect of dielectric constant of pore fluid on sediment volume is somewhat complex. As the dielectric constant increases from 1.9 for heptane to 110 for formamide, sediment volume first decreases, assuming a minimum at 24 for ethanol, increases with a maximum at 80 for water, and decreases again until 110 for formamide. An approximate physico-chemical analysis model is used to interpret some of the data in a quantitative manner. In the analysis model, recently developed theories of double-layer repulsive and van der Waals attractive forces are combined to simulate the behavior of suspensions.

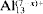 for short), where
for short), where